Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Why primary malignancies of the heart and pericardium are so rare
Naomi Shaw1*; Shiby Stephens2
1 Department of Medicine, St George’s University, London.
2 School of Biosciences, Cardiff University, UK.
*Corresponding Author: Naomi Shaw
Department of Medicine, St George’s University, London.
Email: naomishawuk@live.co.uk
Received : May 26, 2021
Accepted : Jul 02, 2021
Published : Jul 06, 2021
Archived : www.jcimcr.org
Copyright : © Shaw N (2021).
Abstract
Introduction: Primary Malignant Cardiac Tumours (PMCTs) of the heart and pericardium are extremely rare. The incidence rate of both malignant and benign primary tumours is 0.02%, however only one quarter of these are malignant. The reason why these specific malignancies are so uncommon is not yet well understood. This review aims to determine why the heart and pericardium are such inhospitable hosts to PMCTs.
Materials and methods: Literature was systematically analysed using a three-stage methodology; scientific databases were selected; a literature search was conducted and finally a shortlist of six publications was created using a criteria based selection process.
Results: The hearts of 1-day-old mice were able to regenerate after partial resection, however this capacity was lost at seven days old. The terminal differentiation of cardiomyocytes is a result of early cell cycle exit, regulated by Meis1. Myocyte-specific Meis1 deletion resulted in the extension of the postnatal regeneration period and caused reactivation of cardiomyocyte mitosis. Overexpression of Meis1 inhibited cardiac regeneration and decreased neonatal myocyte proliferation. Cardiomyocytes exiting the cell cycle after seven days have less chance of being exposed to mutations. This lack of mutation accumulation results in low rates of PMCTs.
Conclusions: This study has demonstrated the association between low PMCT rates and cardiomyocyte early cell cycle exit, caused by Meis1. The extremely low number of PMCT cases is a result of the heart being unable to regenerate. However, this inability to regenerate causes high incidence rates of cardiovascular disease, due to the formation of scar tissue.
Keywords: Malignant; cardiomyocyte; meis1; regeneration; proliferation; mutation; tumour.
Citation: Karthik R, Ravikumar PT, Fenn SM. Capillary hemangioma on the tip of the tongue. J Clin Images Med Case Rep. 2021; 2(4): 1216.
Introduction
Background
Malignant tumours that originate in the heart are extremely rare and are therefore one of the least investigated subjects in oncology. Published autopsy studies reveal a 0.02% prevalence of cardiac tumours, 75% of which are benign and 25% malignant. The most common type of PMCT is the angiosarcoma, which arises from the inner lining of blood vessels. These account for 30% of cardiac malignancies [1].
Types of heart malignancies
All tumours are classified as either benign or malignant. In the heart, 75% of tumours are benign, meaning the tumour does not invade the surrounding cells, therefore not spreading around the body. Malignant tumours make up the remaining 25% of cardiac tumours and are cancerous tumours that grow quickly and metastasise, often accompanied by a worse prognosis and clinical outcome. The WHO Histological Classification of Tumours of the Heart and Pericardium states that the majority of PMCTs can be classified as different types of sarcomas [2]. Sarcomas are a heterogeneous group of tumours that develop from soft tissue mesenchymal cells and are most common in the trunk, retroperitoneum, head and neck [3]. Even though sarcomas form 75% of all malignant cardiac tumours [1], they account for less than 1% of overall cancer cases. In America in 2004, soft tissue sarcomas made up 0.63% of all cancer cases, showing how rare they are [3].
Sarcomas form 75% of PMCTs, and can be found in any chamber of the heart, however are more frequently found in the right atrium [1]. Angiosarcomas are the most common, arising from the inner lining of blood vessels and represent 30-45 % cardiac malignancies. Other types of sarcomas seen within the heart include; rhabdomyosarcomas, arising from skeletal muscle cells that have failed to fully differentiate, fibrosarcomas, which are derived from fibrous connective tissue, leiomyosarcomas, formed from soft tissue and osteosarcomas, which form in the bones [4]. Table 1 compares these different types of PMCTs.
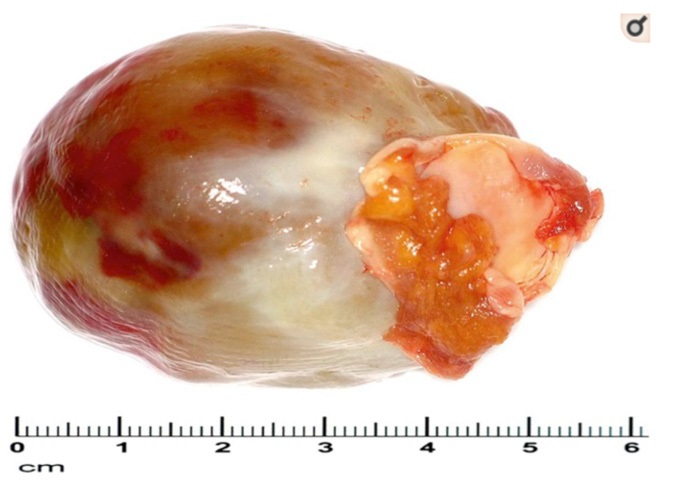
Cardiac myxomas are a myxoid group of tumours of primitive connective tissue and are the most prevalent type of primary cardiac tumour, accounting for 50-70% of cardiac benign tumours [1]. Myxomas can occur in any age group; affecting women more than men, however they are rare amongst children and extremely rare in neonatal periods of life. It is most common for cardiac myxomas to be diagnosed within the left atrium (60-80%). Around 15-28% of myxomas are found in the right atrium, however they are very rarely found in the ventricles [5]. On average myxomas have a diameter of 5-6 cm (Figure 1), however they can grow as large as 15 cm [1].
Cardiomyocyte cell cycle
In humans, the cells of some tissues such as the bone marrow continue to divide throughout life. Other cells such as the light-sensitive cells of the eye show virtually no replacement [6]. The neonatal heart has regenerative capacity due to cardiomyocyte proliferation, however at postnatal day seven, this capacity is lost [7]. This is due to cardiomyocytes exiting the cell cycle during early postnatal days, where they undergo a final round of DNA synthesis and then terminally differentiate [8].
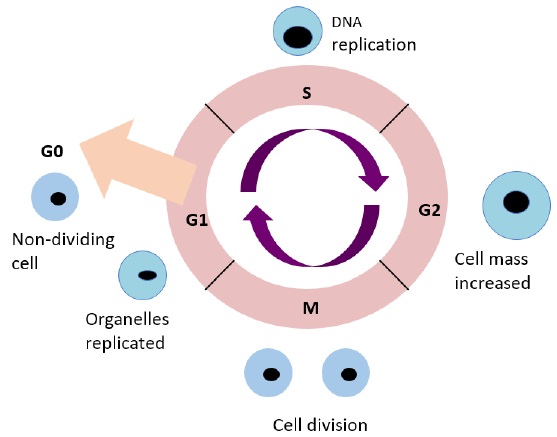
The mammalian cell cycle is a series of events that leads to cell growth and development. In eukaryotes these events are split into two main stages: The interphase and the Mitotic (M) phase. During interphase, the cell grows and replicates its DNA. The interphase is split into three sub phases, gap 1 (G1), S and gap 2 (G2) (Figure 2). G1 is the phase in which the cell grows larger by replicating organelles. During the S phase the amount of DNA in the nucleus is doubled. G2 includes rapid cell growth and protein synthesis to prepare the cell for M phase [9]. Finally, the M phase consists of the cell dividing into two new cells.
Genetic link
There are limited studies discussing whether a genetic link can be drawn in terms of PMCTs being hereditary. This is due to the small number of cases recorded, resulting in inadequate research. Genetic links involving benign myxomas have been made, concluding that 7% of these cases occur in familial clusters, as part of the rare Carney complex [1]. The Carney complex is an autosomal dominant disorder associated with myxomas [10]. A genetically heterogeneous mutation of the tumour suppressor gene PRKAR1A (protein kinase A regulatory subunit-1- alpha gene) on chromosome 17q22-24 has been found to cause this syndrome [11].
Research question
Recent studies, such as Mahmoud et al. [8] and Porrello et al, [12] have explored the hearts limited capacity for regeneration and cell proliferation. One key hallmark of cancer is the ability to maintain cell proliferation, however initial findings suggest the heart lacks this ability, which may affect primary malignancy growth in this area.
Therefore, this paper aims to:
• Determine why the heart is such an inhospitable host to
primary malignancies, and how it appears to have a near
immunity to cancer.
• Explore why the heart has limited capacity to regenerate
and consider whether this results in low numbers of primary malignancies in the heart.
• Assess whether age, race, gender or other factors have an
effect on primary cardiac malignancy prevalence.
Table 1: Comparison of PMCTs. Each primary malignancy can be distinguished between due to different characteristics. This table compares how prevalent each tumour is, as well as the chamber it is most commonly found in. It also compares the ratio between men and women, in addition to where the tumour arises (original table using Burazor et al. 2014 and Hoffmeier et al. 2014).
Type of tumour |
Percentage prevalence |
Most common chamber |
Male: Female ratio |
Arise from |
Angiosarcoma |
30-45 % |
80% of cases found in right atrium |
2:01 |
Vascular endothelium and epicardial surface |
Rhabdomyosarcoma |
20% |
Found in any heart chamber |
Equal frequency |
Striated cardiac musculature |
Fibrosarcoma |
10% |
Most frequent in right atrium |
Equal frequency |
Fibroblasts of cardiac connective tissue |
Leiomyosarcoma |
8-9 % |
Left atrium (posterior wall) |
Equal frequency |
Smooth cardiac muscles |
Osteosarcoma |
3-9 % |
Left atrium wall |
2:01 |
Mesenchymal stem cells of the endocardium |
Methods and results
This study is a review of current literature that has been systematically analysed in order to determine why primary malignancies of the heart and pericardium are so rare.
Selection outline
Chosen scientific databases were used to conduct literature searches and assess the available literature. Publications were filtered using a criteria based selection process, which involved selecting the most relevant literature and enabled a shortlist of six final publications to be created (Figure 3). These shortlisted publications are to be critically analysed in the literature analysis section.
Database selection
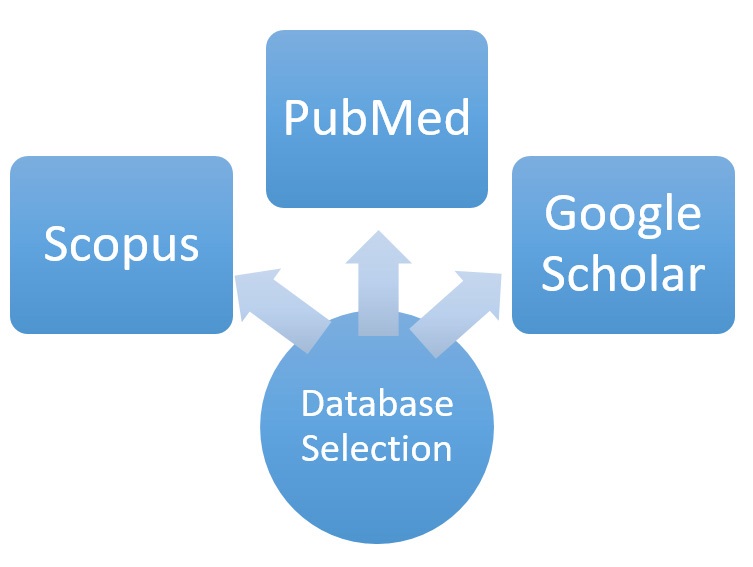
Scientific publications are widely available and can be searched for using a number of databases (Figure 4). Scopus, Google Scholar, PubMed and Web of Science are some of the most renowned databases, so an initial basic search on the topic was performed. Scopus claims to be the largest citation database of peer-reviewed literature and has advanced filtering features, which is useful when narrowing down results. PubMed also has a strong filtering system as well as a ‘See Related Citations’ feature, which enables a wider study of the topic. Although Google Scholar has a limited ability to filter the search, the large number of publications allows a broad reading of the topic, and covers a long time period. Web of Science produced similar results to the other databases, however did not cover such a large time frame, so this database was not used.
Literature search
The chosen scientific databases were used to conduct a literature search addressing PMCTs and reasons why they occur so infrequently. On initial search it became apparent that the majority of publications discussing primary heart malignancies were mainly single-centre studies, prone to small sample size and referral bias. This resulted in an unexpectedly large number of hits during the literature search (Table 2), however only a small number of papers addressed potential reasons why primary tumours of the heart are so rare. Due to this, a large criteria based selection process was put in place, to enable a short list to be created.
Table 2: The number of hits each database provided when the search term ‘Cardiac Primary Malignancies’ was applied. A large number of hits are seen, especially with Google Scholar, however most of these studies are individual case reports (original table).
Database |
Number of Hits: Cardiac Primary Malignancies |
Scopus |
20,290 |
PubMed |
8,833 |
Google Scholar |
433,000 |
Three main searches were conducted in order to link potential reasons why PMCTs are so rare. First, relevant chapters of Moore’s ‘Essential Clinical Anatomy’ and Palastanga’s ‘Anatomy and human movement’ were read in order to gain a clear understanding of the anatomy of the heart and pericardium. Secondly the different types of primary cardiac tumours were explored in order to draw potential links between types of tumour and prevalence. Finally, a search on the cardiomyocyte cell cycle was conducted, as cardiomyocyte exit from the cell cycle can be linked to the small number of cardiac tumours. The search conducted on types of cardiac malignancy produced the most publications due to being the most widely studied, compared to cardiomyocyte exit from the cell cycle, which is less studied.
Results
Through using the selection process above, a shortlist of six publications was created as shown in Table 3.
Table 3: Shortlist of publications used (original table).
Paper Title |
Aim of Paper |
Methods |
Key Findings |
Use in Current Study |
Characteristics, survival and incidence rates and trends of primary cardiac malignancies in the United States (Saad et al. 2018) |
|
|
|
|
Risk factors for early death in primary malignant cardiac tumours: An analysis of over 40 years and 500 patients (He et al. 2018) |
|
|
|
|
Meis1 regulates postnatal cardiomyocyte cell cycle arrest (Mahmoud et al. 2013) |
|
|
|
|
Cardiomyocyte cell cycle- Meis-ing something? (Mahmoud et al. 2014) |
|
|
|
|
Transient Regenerative Potential of the Neonatal Mouse Heart (Porrello et al. 2011) |
|
|
|
|
Evidence for cardiomyocyte renewal in humans (Bergmann et al. (2009) |
|
|
|
|
Critical based selection
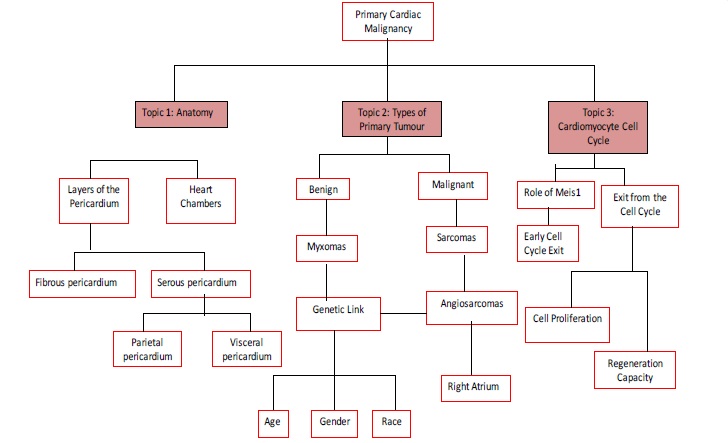
During the literature search, filtering took place to ensure any review publications were avoided, due to their subjective nature. This removed any chance of positive or negative biased and left only primary publications for the shortlist to be selected from. The majority of the literature surrounding PMCTs involves single centre case studies, which is not useful when coming to research why cardiac tumours occur so infrequently. After removing these individual case reports, 286 publications remained. 119 duplicates were removed to leave 167 unique publications. Literature published before 1989 was discarded due to being superseded by more recent studies, leaving 139 publications. A relevance check was performed on the remaining 139 publication’s titles using a relevance tree (Figure 5), which allowed oversight of the whole topic. This helped to eliminate literature that did not directly relate to the various areas of research, leaving 32 publications. The abstracts of the remaining 32 publications were scanned and a shortlist of the most up to date and relevant six publications was created. These final six publications are to be discussed and critically analysed in the literature analysis section.
Limitations of selection process
The main limitation associated with this selection process is the large number of publications obtained in the initial search, however only a small number of these publications relate to this literature review (Table 2). Ideally all publications would undergo the relevance check, however the sheer number of publications made this impossible. Single-case studies were automatically discarded, as analysing one individual case will not provide information that is of use to this study. It would be preferable to read and analyse all 32 pieces of literature remaining after the relevance check, however the quantity of publications meant this would not be practical. All papers in the shortlist were written within the last ten years, making them up to date and relevant. There is of course the chance that relevant literature may have been missed during this process, however given the large amount of literature and time constraints, this method appeared to be the most practical. This process ensured the most relevant and up-to-date literature was shortlisted and allowed quick and easy removal of all non-relevant publications.
Discussion
Cardiac regeneration potential
The failure of the adult heart to regenerate myocardium has until present been seen as a negative with regard to human health. Mammals develop extensive scarring instead of regeneration, which is a leading cause of heart failure and therefore death, worldwide [13]. However, the heart being unable to regenerate may result in a very low number of PMCTs, which in turn, is positive in respect to human health. This makes heart regeneration clinically relevant and fascinating, with increased research into therapeutically stimulating regeneration in injured human hearts [14]. Mahmoud et al. [8] found that a deletion in the Meis1 gene resulted in the extension of the postnatal regeneration period and reactivation of myocyte proliferation in the heart. This is advantageous in the development of treatments for heart diseases, however, could be detrimental with respect to heart cancer formation, as heart regeneration and proliferation could increase the potential for cardiac malignancies to form.
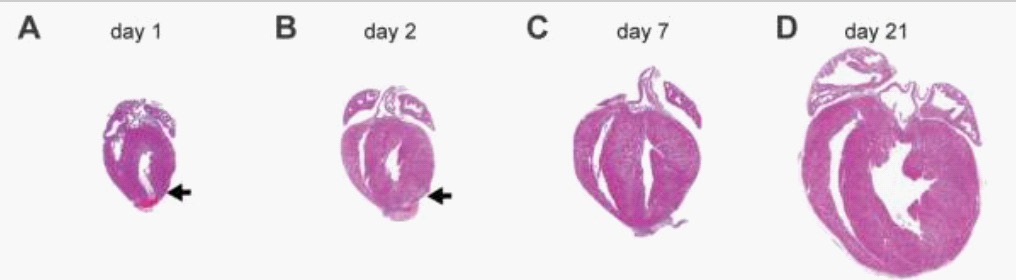
Many decades ago, mammalian hearts were thought to be post-mitotic organs without the capacity to regenerate [15]. In 2011, Porrello and colleagues discovered that mammalian hearts maintain the capacity to regenerate for a short period after birth. This is significant as it is the first study reporting regeneration potential in mammalian hearts. This study had a positive impact, shown by the fact it is cited 1266 times, with many subsequent papers building on its findings. The study showed that the hearts of 1-day-old neonatal mice were able to regenerate after partial surgical resection (Figure 6), however this capacity was lost at 7-days-old. This regenerative response of the 1-day-old mice was characterised by cardiomyocyte proliferation [12]. An Echocardiogram was performed after two months and revealed that the regenerated ventricular apex had a normal function, showing successful regeneration capacity.
Interestingly, not all animal hearts lose the ability to regenerate shortly after birth. Certain fish and amphibians retain regeneration capacity throughout life. For example, the hearts of adult zebrafish can completely regenerate after resection, which is due to cardiomyocyte proliferation. However, the adult hearts of mammals and fish are anatomically very different, which may influence the difference in regeneration potential. The mammalian heart is a four chambered, double circulation system that works at high pressure. This differs from fish hearts, which comprise of only two chambers and single circulation. Cardiomyocytes also differ between the two, with mammalian myocytes withdrawing from the cell cycle seven days after birth, resulting in the loss of ability to proliferate. On the other hand, zebrafish cardiomyocytes are mononucleated and maintain the ability to proliferate throughout adult life [12]. Despite the differences between adult hearts, the neonatal mammal heart, before undergoing septation, has a regeneration capacity reminiscent of lower vertebrates [8]. Both involve a single circulation system and cardiomyocytes that are able to proliferate, indicating that cardiomyocyte proliferation is a key factor of successful cardiac regeneration.
Bergmann et al. [16] discovered that cardiomyocyte proliferation could occur in adult hearts, but at an extremely low level. This contradicts studies such as Porrello et al. [12], who suggest no cardiomyocyte proliferation occurs in adult hearts, however it is important to explore both findings. Due to a nuclear bomb test during the Cold War, carbon-14 (14C) was integrated into DNA. Bergmann and colleagues [16] measured the 14C in genomic DNA of human myocardial cells, which allowed the identification of the cell’s birth date. It was found that 1% of human cardiomyocytes are renewed annually at the age of 25, which is reduced to 0.3% at the age of 75. This suggests that an extremely small number of cardiomyocytes retain the ability to proliferate, however an insufficient number to have an impactful effect on overall cardiac regeneration.
The role of Meis1
Meis1 belongs to the TALE (3-amino-acid loop extension) family of homeodomain transcription factors. Originally the role of Meis1 in cardiomyocytes was unknown, however in 2013 Mahmoud and colleagues identified Meis1 as a key regulator of postnatal cardiomyocyte arrest. They also found Meis1 to be a regulator of cardiomyocyte proliferation and a possible beneficial target for heart regeneration. The effect Meis1 deletion and overexpression had on cardiomyocyte proliferation and heart regeneration was investigated. Myocyte-specific Meis1 deletion resulted in an extension of the postnatal regeneration period and caused re-activation of cardiomyocyte mitosis in the adult heart. However, no change in cardiac function was observed, making Meis1 deletion a promising area of research for potential heart regeneration treatments. On the other hand, overexpression of Meis1 inhibited cardiac regeneration and decreased neonatal myocyte proliferation. Mahmoud and colleague’s (2013 and 2014) studies are significant as they provide evidence on the effects of Meis1 deletion and overexpression that have not been studied before.
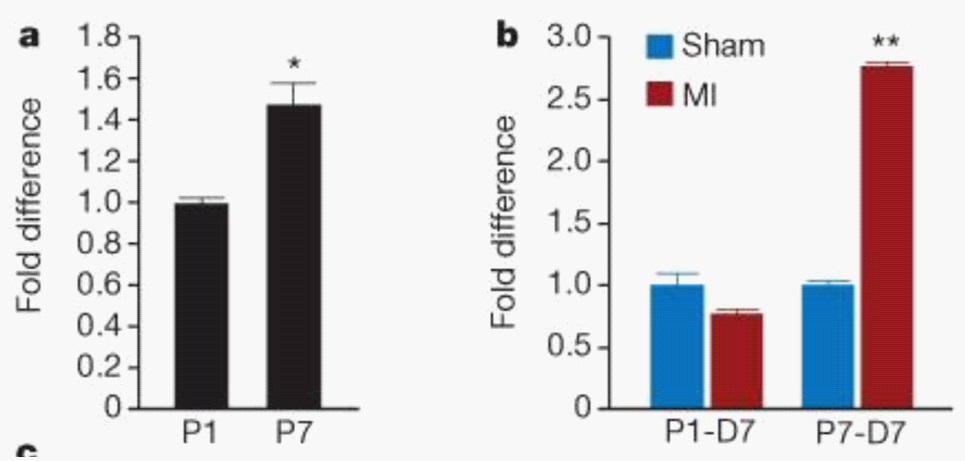
Mahmoud et al. [7] investigated the role of Meis1 in the regulation of the cardiomyocyte cell cycle. They started with examining the Meis1 expression pattern. Quantitative PCR showed an increase in Meis1 expression at postnatal day seven (P7), compared to postnatal day one (P1) (Figure 7a). Immunostaining revealed expression of Meis1 from as early as four days after birth and throughout adult life. Myocardial Infarction (MI) at P1 was linked to a slight decrease in expression of Meis1. Meis1 mRNA expression levels were significantly increased following MI at P7 (Figure 7b). This coincides with an absence of mitotic induction of cardiomyocytes [7].
Mahmoud et al. [7] undertook multiple experiments during their study. For example, they investigated the effect Meis1 deletion had on cardiomyocyte proliferation and whether this deletion resulted in cell cycle re-entry. In vitro siRNA knockdown was performed using rat neonatal cardiomyocytes. Meis1 knockdown caused a threefold increase in cardiomyocyte proliferation. A cardiomyocyte-specific Meis1 knockout mouse was created and compared to a control. Heart size and cardiac function at P14 were unaffected in the knockout mouse, however the cardiomyocytes were smaller than the controls. An inducible knockout mouse was also created and proved that conditional deletion of Meis1 in the adult heart induces cardiomyocyte cell cycle re-entry.
As well as investigating the effects of Meis1 deletion, they also investigated the effect of Meis1 overexpression. A cardiomyocyte-specific Meis1 overexpressing mouse was generated, which showed a decrease in the number of mitotic neonatal cardiomyocytes and inhibition of neonatal heart regeneration following the induction of MI at P1. This indicates that Meis1 overexpression in neonatal hearts results in early cardiomyocyte cell cycle arrest, confirming that Meis1 is the key regulator of cardiomyocyte cell cycle exit.
The results that Mahmoud et al. [7] present are proven at an immunochemical, molecular and genetic level, with good use of statistical analysis. The conclusions made are justified, with no further experiments needed to validate the results presented. Due to its significant findings, this paper has been widely referenced (cited by 270) and built upon, with research into why cardiomyocytes exit the cell cycle early being conducted in response. Mahmoud et al [8] states that there is now a clear understanding that targeting the cardiomyocyte cell cycle is a feasible therapeutic option. This paper also suggests that the reason why cardiomyocytes exit the cell cycle permanently seven days after birth may be a protective mechanism against malignant transformation. This is due to cardiomyocytes not being able to proliferate, meaning any mutations picked up before cell cycle arrest cannot accumulate.
Comparison of regeneration in other cell types
Cardiomyocytes, along with neurons in the brain, are one of the only cell types that are unable to regenerate. As mentioned previously, the lost ability of cardiomyocytes to proliferate is due to an early exit from the cell cycle that occurs seven days after birth in mammals, and is regulated by Meis1 [12]. Several mutations may occur during the cell cycle [17], and it is the accumulation of these mutations that leads to cancer, a disease of the genome [18]. Cells exiting the cell cycle after only seven days have less chance of being exposed to mutations, and can no longer divide, meaning any mutations picked up during fetal development are not multiplied and passed onto daughter cells.
Neurons also lack the ability to regenerate and are viewed as post-mitotic cells that undergo apoptosis in response to cell cycle reactivation. They permanently block their capacity to proliferate once they are differentiated and remain in the quiescent state during adult life [19]. The incidence of Primary Malignant Brain Tumours (PMBT) is 3.7 per 100,000 for men, and 2.6 per 100,000 for women. Approximately 21,810 individuals were diagnosed with PMBTs in the United States in 2008 [20]. In spite of these statistics, only a very small number of PMBTs originate from the neurons. At present a total of only 62 cases of neuronal tumours have been recorded [21]. This is a result of neurons being unable to proliferate and undergo mitosis, leaving less chance for mutations to be picked up during the cell cycle that may accumulate and result in cancer [19]. Gliomas are the most common type of PMBTs, arising from glial cells. These are the non-neural cells of the Central Nervous System (CNS) that provide nutrients and support to neurons. They retain the ability to undergo cell division throughout adult life. Consequently, mutations are more likely to develop in these cells, resulting in cancer that is much more common than in neurons. Gliomas account for approximately 10,000 cases of PMBTs diagnosed every year in the United States [22].
Epithelial cells are the polar opposite of cardiomyocytes and neurones in terms of proliferation. Epithelial cells function as a mechanical and chemical barrier between the body and environment and are continuously growing and regenerating throughout life. These cells do not exit the cell cycle like cardiomyocytes do; they undergo mitosis throughout their life. They are constantly being replaced by new cell divisions, which allows mutations to accumulate, which can eventually lead to cancer. Carcinomas are cancers that arise from epithelial tissue. They are extremely common and account for 90% of cancer cases, with the other 10% arising as leukemias and sarcomas [23]. Cancer is a disease that develops slowly. For example, in solid human tumours there is often, but notably not always, an interval of 20 years from carcinogen exposure to clinical detection [18]. This highlights the benefits of cardiomyocyte early cell cycle exit as a protective mechanism against malignant formations, as cells such as epithelial cells that remain in the cell cycle are not protected from the accumulation of mutations, and therefore the formation of cancer.
Primary cardiac malignancy trends
Primary cardiac malignancies are rare events, and the literature on their incidence, cause and treatment is largely limited. The incidence of cardiac tumour diagnosis has risen in the past few decades, however it is uncertain if this is due to over diagnosis or due to an actual rise in malignancy formation [2]. Very recently, two studies have been published with data collected over a wide time frame, allowing trends and risk factors of primary malignant cardiac tumours to be analysed. Saad et al. [24] conducted a study on the characteristics, survival and incidence rates and trends of primary cardiac malignancies in the United States. He et al. [2] studied the risk factors associated with early death in patients diagnosed with primary malignant cardiac tumours. These studies are useful as many previous publications related to cardiac tumours are single centre studies, which do not allow for trends or risk factors to be investigated.
Saad et al. [24] selected patients with PMCTs diagnosed between 2000 and 2014, excluding cases where the diagnosis relied only on autopsy or death certificates. Data was selected from the Surveillance, Epidemiology and End Results (SEER)-18 registry. 497 cases were reviewed, 136 of these being angiosarcomas and 134 were non-Hodgkin’s lymphomas (NHL). The rest were divided into ‘other sarcomas’ which included all sarcomas apart from angiosarcomas, and ‘other’ which included epithelial tumours, germ cell tumours and any other types that did not fit the previous groups. Within these groups, Saad et al. [24] looked into multiple variables, including: sex, race and age at diagnosis. Most patients with angiosarcomas were male, white and aged 20-50. Most NHL patients were also male and white, however they were older than 50 years of age (Table 4) [24].
Table 4: Characteristics of PMCT patients diagnosed between the year 2000 and 2014 in the United States. (Adapted from Saad et al. 2018).
|
Histological type no. (%) |
|||
Angiosarcoma |
NHL |
Other sarcomas |
Other |
|
Overall |
136 (100) |
134 (100) |
173 (100) |
54 (100) |
Sex: |
|
|
|
|
Male |
75 (55.1) |
74 (55.2) |
81 (46.8) |
30 (55.6) |
Female |
61 (44.9) |
60 (44.8) |
92 (53.2) |
24 (44.4) |
Age at diagnosis: |
|
|
|
|
<20 |
3 (2.2) |
3 (2.2) |
15 (8.7) |
7 (13.0) |
20-49 |
80 (58.8) |
16 (11.9) |
71 (41.0) |
17 (31.5) |
>50 |
53 (39.0) |
115 (85.8) |
87 (50.3) |
30 (55.6) |
Race: |
|
|
|
|
White |
91 (66.9) |
103 (76.9) |
146 (84.4) |
42 (77.8) |
Black |
27 (19.9) |
10 (7.5) |
11 (6.4) |
7 (13.0) |
Other |
18 (13.2) |
20 (14.9) |
16 (9.2) |
5 (9.3) |
Vital status: |
|
|
|
|
Alive |
17 (12.5) |
59 (44) |
35 (20.2) |
8 (14.8) |
Dead |
119 (87.5) |
75 (56) |
138 (79.8) |
46 (85.2) |
Chemotherapy: |
|
|
|
|
No/Unknown |
59 (43.4) |
37 (27.6) |
100 (57.8) |
35 (64.8) |
Yes |
77 (56.6) |
97 (72.4) |
73 (42.2) |
19 (35.2) |
Surgery: |
|
|
|
|
No |
67 (49.3) |
109 (81.3) |
40 (23.1) |
30 (55.6) |
Yes |
68 (50) |
23 (17.2) |
132 (76.3) |
23 (42.6) |
Saad et al. [24] calculated the incidence rate of angiosarcoma and NHL between the year 2000 and 2014. The incidence rate of angiosarcoma did not change significantly (p>0.05), with a TTest producing a p-value of 0.483. However, the incidence rate of NHL was seen to increase significantly over the time period (p<0.05), with a T-Test producing a p-value of 0.003. The median survival time of both angiosarcomas and NHL was also calculated. Results concluded a mean survival time of nine months for angiosarcomas, and 34 months for NHLs. The survival time for NHLs was significantly better than angiosarcomas (P<0.001). Saad et al. [24] include good use of statistics throughout their study, which allowed deeper analysis of results.
He et al. [2] conducted a similar study in the United States, involving 564 patients over a 40-year time frame. With this time frame being longer than the previous paper, trends can be drawn back to an even earlier date. This paper is the first of its kind to investigate the risk factors that contribute to early deaths in patients diagnosed with PMCTs. This is impactful as understanding the risks contributing to early death could eventually lead to better survival rates in PMCTs. Data selected from the SEER registry showed 744 patients with PMCTs from 1973 to 2014. Patients (180) were excluded due to only having an autopsy report or because they had had multiple primary tumours. This left a total of 564 patients, 214 of which were registered as early deaths (survived ≤3 months after diagnosis). The two groups, early death and not early death, were compared on the basis of: sex, age, race and ethnicity. The types of PMCTs were also compared between the two groups. The early death group had a lower proportion of sarcomas and lymphomas compared to the non-early death group.
In patients with sarcomas, results showed that men had a higher risk of early death compared to women. Individuals younger than 20 had a lower risk of early death compared to individuals over 80, who showed a significant increase in the risk of early death. The risk of early death was not affected by race, ethnicity, education or income. In patients with lymphomas, the only group observed to be statistically significant was the age group. Individuals in age groups over 41 showed significantly high early death rates compared to individuals aged 20-40. The overall early death rate decreased slightly over the five decades that this study covered. He et al. [2] included a broad range of statistical analysis such as the X2 and Fisher’s exact test, which is useful when comparing variables.
Both Saad et al. [24] and He et al. [2] have produced impactful studies that were conducted over long time periods . However, there are some limitations, for example the fact that due to the rarity of PMCTs the number of patients included in both studies is relatively small [25-29]. The SEER database cannot provide information on chemotherapy and radiotherapy treatments, making it unclear which patients received treatment [2]. The terminology for different classifications of PMCTs has changed many times since the 1970’s. This makes the comparisons that He et al. [2] made between different types of PMCT less reliable. Despite these limitations, both studies provide evidence for trends and risk factors that have not been investigated before, proving very useful for future studies [29-32].
Conclusion
Impact of cardiomyocyte cell cycle arrest
This study explored many aspects of PMCTs, including the cardiomyocyte cell cycle, the role of Meis1 and the trends and incidence rates of PMCTs. The main conclusion that can be drawn from this literature review is that PMCTs rarity is largely due to cardiomyocyte cell cycle arrest. Cardiomyocytes exit the cell cycle at postnatal day seven, meaning they are only exposed to mutations picked up during the cell cycle for a short period of time. As cardiomyocytes can no longer undergo mitosis once they exit the cell cycle, any mutations picked up in the first seven days cannot be excessively multiplied and therefore will rarely accumulate to form cancer. As a result, there are an extremely low number of heart cancer cases. However, the mechanism that enables protection against malignancy contributes to the very high number of cardiovascular disease cases. Cardiomyocytes being unable to divide results in the hearts inability to regenerate and instead scar tissue is formed, resulting in cardiovascular disease. This highlights that the early exit of cardiomyocytes from the cell cycle has both a positive and negative effect on human health. It results in extremely low rates of cardiac malignant transformations, conversely, the inability of the heart to regenerate causes very high incidence rates of cardiovascular disease.
Future aims
Although there have been some important conclusions drawn from this literature review, there are still some questions that remain unanswered. Going forward in this investigation into the rarity of heart cancer, new aims should be set, in order for more advances to be made. For example, we have established that PMCTs are rare due to cardiomyocyte early cell cycle exit. However, we do not know the reason why cardiomyocytes undergo cell cycle arrest. A future aim would be to investigate the reason why Meis1 causes cardiomyocytes to exit the cell cycle. Is it a protective mechanism for malignant transformation, or is there another reason why cardiomyocytes are made to exit the cell cycle so early? The same question can be asked in regard to the trends in PMCTs. It has been established within this review that PMCTs are more common in males than females and are more common in the white ethnicity compared to the black ethnicity. These are important findings, however a future aim would be to understand why this is the case. Is there a reason why PMCTs are more common in males and white people compared to females and black people? Creating new aims will allow the development of future investigations, which will be discussed in the next chapter of this review.
Limitations of this review
As with most studies, this literature review has some limitations. Due to the rarity of PMCTs, research on this subject is largely limited. There is a very restricted amount of literature relating to PMCTs, with most of these publications being single centre studies. These are not of use to this literature review as single centre studies are largely subjective and do not allow for comparisons to be made. There is no research comparing PMCTs in neonates and adult humans and very limited research on reasons why these malignancies do not form very frequently. This has made it difficult to find research that relates to the question being asked in this review. The rarity of PMCT cases also results in a very small sample size when analysing trends and incidence rates. For example, in this review, trends were analysed, however the sample size used was only 500 cases, which is relatively small. Consequently, the results are less reliable than if trends were analysed using a much larger sample size. Furthermore, the terminology for different classifications of PMCTs has changed over the years. For example, new tumour types have been discovered and existing terminology has been changed. This makes comparisons between tumour types over a long period of time more difficult. In spite of these limitations, this review records significant findings. It provides a broad analysis of recent literature, enabling positive advances to be made in the cardiology field.
References
- Hoffmeier A, Sindermann JR, Scheld HH, Martens S. Cardiac Tumours-Diagnosis and Surgical Treatment. Dtsch Arztebl Int. 2014.
- He S, Cao Y, Qin W, Chen W, Yin L, Chai H, et al. Prevalence of primary cardiac tumour malignancies in retrospective studies over six decades: A systematic review and meta-analysis. Oncotarget. 2017.
- Cormier J, Pollock R. Soft Tissue Sarcomas. CA: A Cancer Journal for Clinicians. 2008.
- Burazor I, Aviel-Ronen S, Imazio M, Markel G, Grossmand Y, Yosepovich A, et al. Primary Malignancies of the Heart and Pericardium. Clinical Cardiology. 2014.
- Gošev I, Paic F, Duric Z, Gošev M, Ivcevic S, Bulic J, et al. Cardiac myxoma the great imitators: Comprehensive histopathological and molecular approach. International Journal of Cardiology. 2013.
- Bolsover S. Cell biology. 3rd edition. 2011.
- Mahmoud A, Kocabas F, Muralidhar SA, Kimura W, Koura AS, Thet S, et al. Meis1 regulates postnatal cardiomyocyte cell cycle arrest. Nature. 2013.
- Mahmoud A, Canseco D, Xiao F, Sadek HA. Cardiomyocyte cell cycle: Meis-ing something?. Cell Cycle. 2014.
- Pollard T, Earnshaw W, Lippincott-Schwartz J, Johnson G. Cell biology. 3rd edition. 2016.
- Gür AK, Kaya Y, Karakurt A, Guvenc TS. The Carney syndrome associated fibroelastoma and myxoma. Dicle Medical Journal. 2012.
- Serio A, Favalli V, Giuliani LP, Narula N, Grasso M, Borroni R, et al. Cardio-Oncology: The Carney Complex Type I. Journal of the American College of Cardiology. 2016.
- Porrello ER, Mahmoud AI, Simpson E, Hill JA, Richardson JA, Olson EN, et al. Transient Regenerative Potential of the Neonatal Mouse Heart. Science. 2011.
- Uygur A, Lee RT. Mechanisms of Cardiac Regeneration. Developmental Cell. 2016.
- Kituchi K, Poss KD. Cardiac Regenerative Capacity and Mechanisms. Annual Reviews Cell Developmental Biology. 2012.
- Carvalho A, Campos de Carvalho A. Heart regeneration: Past, present and future. World Journal of Cardiology. 2010.
- Bergmann O, Bhardwaj RD, Bernard S, Zdunek S, Barnabé-Heider F, Walsh S, et al. Evidence for cardiomyocyte renewal in humans. Science. 2009.
- Cooper GM. The Cell: A molecular approach. 2nd edition. 2000.
- Loeb LA, Loeb KR, Anderson JP. Multiple mutations and cancer. PNAS. 2003.
- Frade JM, Ovejero-Benito MC. Neuronal cell cycle: the neurone itself and its circumstances. Cell Cycle. 2015.
- Bondy ML, Scheurer ME, Malmer B, Barnholtz-Sloan JS, Davis FG, Il’yasova D, et al. Brain tumor epidemiology: Consensus from the Brain Tumor Epidemiology Consortium. Cancer. 2008.
- Choi E, Kim,S, Won J-K, Chung CH, Kim SK, Choi S-H, et al. Clinicopathological and molecular analysis of multiodular and vacuolating neuronal tumors of the cerebrum. Human Pathology. 2018.
- Chang HJ, Burke AE, Glass RM. Gliomas. JAMA. 2010.
- Frank SA. Dynamics of Cancer: Incidence, Inheritance and Evolution. 2017.
- Saad AM, Abushouk AI, Al-Husseini MJ, Salahia S, Alrefai A, Afifi AM, et al. Characteristics, survival and incidence rates and trends of primary cardiac malignancies in the United States. Cardiovascular Pathology. 2018.
- Cvejic A, Serbanovic-Canic J, Stemple DL, Ouwehand WH. The role of Meis1 in primitive and definitive hematopoiesis during zebrafish development. Haematologica. 2011.
- Etchin J, Kanki JP, Look AT. Zebrafish as a model for the study of human cancer. Methods in Cell Biology. 2011.
- Hartwell LH, Kastan MB. Cell cycle control and cancer. Science. 1994.
- Mahadevan V. Anatomy of the heart: Basic science. 2011.
- Moore KL, Agur AMR, Dalley AF. Essential clinical anatomy. 5th edition. 2014.
- Palastanga N, Soames R. Anatomy and human movement. 6th edition. 2012; 510-514.
- Smits AM, Vilet PV, Hassink RJ, Goumans M-J, Doevendans PA. The role of stem cells in cardiac regeneration. Journal of Cellular and Molecular Medicine. 2005.
- Tortora G, Derrickson B. Principles of anatomy and physiology. 2009.