Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Deep vein thrombosis and our treatment applications: 12-year results
Borulu F; Erkut B*
Atatürk University, Medical Faculty, Department of Cardiovascular Surgery, Erzurum-Turkey.
*Corresponding Author : Bilgehan Erkut
Atatürk University Medical Faculty, Department of Cardiovascular Surgery, Erzurum, Turkey
Email: bilgehanerkut@yahoo.com
Received : May 04,, 2021
Accepted : Aug 02, 2021
Published : Aug 05, 2021
Archived : www.jcimcr.org
Copyright : © Erkut B (2021).
Abstract
Introduction: Deep vein thrombosis is an important health problem that is frequently encountered in the general population and especially in surgical clinics and has a negative impact on quality of life. In this study, treatment options and results of patients with deep vein thrombosis who have been hospitalized for 12 years in Atatürk University and Erzurum Regional Hospital were examined and discussed.
Methods: In our clinic, 412 cases of deep vein thrombosis (211 female, 201 male) were hospitalized between 2009 and 2021. The mean age of the patients was 49 ± 19 years. While medical treatment with heparin was given to all 412 patients; thrombolytic therapy, surgical embolectomy (vascular and pulmoner), and pharmaco-mechanical thrombectomy were applied to some of these patients.
Results: Deep vein thrombosis was more common in the lower extremity (n=322, 78.2%). All patients had at least one of the complaints of pain, swelling and redness. All cases were diagnosed by color Doppler ultrasonography. Medically, standard and low molecular weight heparin therapy was given to all patients after hospitalization. Thrombolytic therapy was applied to 66 (16,1%) of the cases, pulmonary embolectomy to 8 (1,9%), surgical thrombectomy to 10 (2,4%) and pharmacomechanical thrombectomy to 44 (10,7%) patients. In addition to these 120 patients (29.1%), a vena cava filter was placed for prophylactic purposes.
Conclusions: In patients diagnosed with deep vein thrombosis and hospitalized, the diagnosis should be supported by Doppler ultrasound in addition to clinical diagnosis after an etiology investigation. Early diagnosis, rapid and effective treatment methods are important for the subsequent quality of life of patients. In addition to low molecular weight heparins being the first choice agents, standard heparin administration still needs to be applied in some clinical pictures. In addition, we believe that pharmacomechanical thrombectomy can be applied in appropriate acute cases.
Keywords: Deep vein thrombosis; anticoagulant treatment; heparin therapy; low-molecular-weight heparin; mechanical thrombectomy.
Citation: Borulu F, Erkut B. Deep vein thrombosis and our treatment applications: 12-year results. J Clin Images Med Case Rep. 2021; 2(4): 1253.
Introduction
Deep Vein Thrombosis (DVT) is a serious disease that is common in inpatients and the general population. It derives its severity from Pulmonary Embolism (PE) and post-phlebitic syndrome, which are complications leading to significant morbidity and mortality. The common point of the treatment in this disease picture, where the existing risks can be reduced with appropriate treatment and prophylaxis. It should be to relieve the symptoms, prevent relapses and restore the patient’s normal activity by reducing treatment time and economic costs as much as possible.
Although the etiology of venous thrombosis is not fully understood, the triad introduced by Virchow in 1856; stasis, hypercoagulability and endothelial damage are still valid [1]. Experimental and clinical studies, in the pathogenesis of venous thromboembolism; demonstrated that events such as vascular wall, stasis, coagulation factor inhibitors and a decrease in the potential of the fibrinolytic system all play a role [2,3]. While the technical and scientific developments in the field of cardiovascular surgery are progressing at a dizzying speed, “venous diseases”, which is the area of interest of this discipline, have not been emphasized with the same excitement and sensitivity until today. However, in recent years, attention has turned to this aspect of cardiovascular surgery, thanks to the better understanding of the complications and socio-economic losses caused by vein diseases, especially DVT. In this study, we discussed the treatment practices and results of patients hospitalized with deep vein thrombosis.
Methods
A total of 412 cases between the ages of 21-88 diagnosed with DVT Between Aug 2009 Dec 2020 were hospitalized in our clinic for follow-up and treatment. These patients consisted of patients who were hospitalized from the cardiovascular surgery outpatient clinic and who underwent surgical intervention in other clinics with another diagnosis. The mean age of the patients was 49 ± 19 years. 201 (48,8%) of the cases were female and 211 (51,2%) were male. 322 (78,2%) of our patients were proximal DVT cases involving the lower extremity and the inferior distal of the vena cava. Patients with uncontrolled hypertension, liver or kidney biopsy, over the age of 90, who received thrombolytic therapy in the last 6 months, and those who had streptococcal infection, those who were diagnosed with DVT in another center and applied to our outpatient clinic for control, in addition to those who recently had gastrointestinal system bleeding and cerebrovascular stroke and bleeding, patients with profound anemia, thrombocytopenia, liver dysfunction, uremia, and coagulation disorders were excluded from the study.
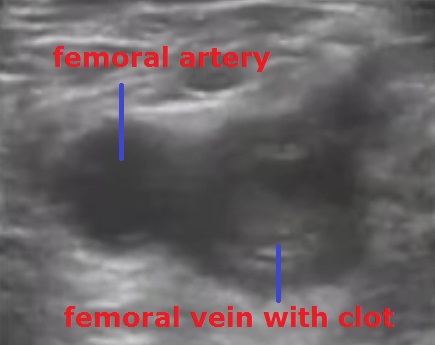
Patients with symptoms of pain, swelling, and redness in the upper and lower extremities were hospitalized, diagnosed with deep venous thrombosis and treated. In the physical examination, Homans test (increase in pain with dorsiflexion of the foot) was also evaluated in addition to the circumference of the extremities, edema and redness. In order to investigate the response to the treatment, the differences in thigh and calf circumference between the extremities, which were measured and recorded daily from the day of hospitalization to discharge, were recorded (15 cm below and 20 cm above the patella). In all patients, on the day of hospitalization, chest radiography, electrocardiography, hemogram, biochemical analysis, complete urinalysis, activated Prothrombin Time (aPTT), international normalized ratio (INR) values were measured. The patients were examined etiologically. In addition to effective factors such as long-term inactivity, pregnancy and orthopedic intervention, genetic parameters such as Antithrombin III deficiency, Factor V Leiden mutation, Protein-C and S deficiency and plasminogen disorders were also examined. In addition, D-dimer values were measured to support the diagnosis of PE in patients with DVT, chest pain, and shortness of breath. Color Doppler USG was used as a definitive diagnostic method to aid history and physical examination (Figure 1). Additional imaging methods such as venography or magnetic resonance were not used in any of our patients. Doppler USG examination was repeated within 7-10 days and the response to treatment was evaluated. Diagnostic tests were performed and interpreted by experienced radiologists in the radiology clinic of our hospital.
Standard Heparin (SH) (Liquemine-Roche) treatment was started with an intravenous bolus of 5,000 IU and was treated as intermittent intravenous administration (6x5000 IU or 8x5000 IU 30,000-40,000 IU/day) or continuous IV infusion (1000-1500 IU/hour) it was continued. An additional dose of 2500 IU was administered to the cases deemed necessary according to the coagulation time (PT: 2-2.5 times the normal value). For Low Molecular Weight Heparin (LMWH) treatment, enoxaparin (Oksapar-Koçak Farma or Clexane-Aventis Pharma) was administered by subcutaneous injection of 4 mg/kg twice a day. Laboratory monitoring was not performed in patients receiving LMWH. In our patients, heparin (SH and LMWH) treatment was continued for at least 7 days in the first years, and warfarin sodium (Coumadin-Eczacıbaşı), an oral anticoagulant agent, was added to the treatment on the 5th day of the treatment. However, since the last 5 years, this practice was abandoned and heparin treatment and oral anticoagulant therapy were started in the same mirror and the treatment continued. Heparin treatment was continued until the period when INR would be above 2.0. Continuation of maintenance therapy for at least 3 months was recommended, however, in the presence of recurrent DVT or persistent and irreversible risk factors (such as thrombophilic status), patients were discharged by recommending continued anticoagulation for 6 months or lifelong. In recent years, LMWHs have been used more in treatment and prophylaxis. In patients given warfarin as an oral anticoagulant, INR levels were kept within therapeutic limits with intermittent polyclinic controls. Maintenance treatment with LMWH was used in cases where warfarin sodium treatment was found to be harmful. Thrombolytic therapy was administered to patients who presented with acute diffuse iliofemoral vein thrombosis, whose diagnosis was supported by Doppler USG, did not have contraindication to fibrinolytic therapy, and whose symptoms did not exceed 7 days. Streptokinase (Kabikinase-Pharmacia) was initiated at a dose of 250,000 IU IV bolus in 30 minutes and continued systemically for 3 days with an IV infusion at a dose of 100,000 IU per hour. Urokinase (Urokinase-Koçsel) was continued at a dose of 100,000 IU / hour, followed by 100,000 IU IV bolus, and completed to 1,000,000 IU in total. rt-PA (Actilyse-Boehringer Ingelheim) was applied by the loco-regional lysis method. For this purpose, a branule was inserted into the saphenous vein in front of the dorsum or medial malleolus. Two pressure cuffs were attached to the calf and thigh with pressures of 40 and 80 mmHg, respectively, during the treatment, and rt-PA was administered at a daily dose of 20 mg for 2 days with 8 hour infusions. In cases deemed necessary, the dose was increased to 40 mg and rt-PA was administered for 2 more days. During thrombolytic therapy, at a dose of 1000-1500 IU / hour, PT was adjusted to be 2-2.5 times the normal value, heparin infusion was continued for at least 10 days, and oral anticoagulant therapy was continued for 3-6 months.
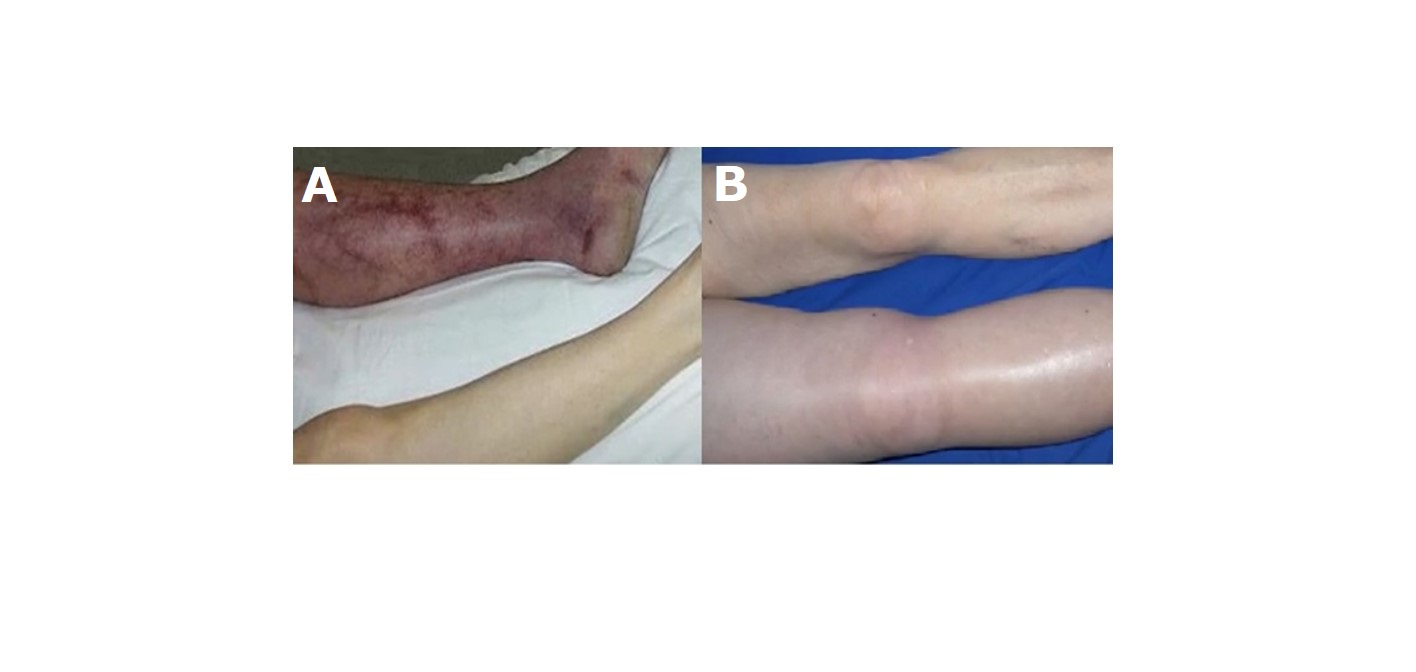
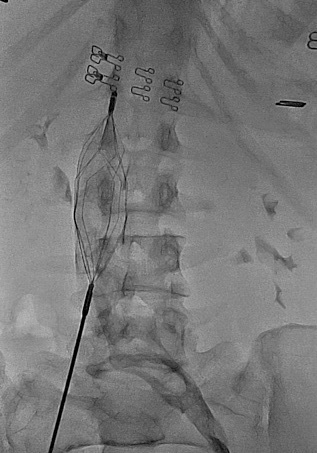
In the early days of the DVT clinic, very few patients admitted to the hospital for venous thrombosis with large ilio-femoral and vena vaca inferior thrombosis, including phlegmasia cerulea dolens, phlegmasia alba dolens (Figure 2) were not suitable for anticoagulation and thrombolytic therapy, and femoral vein embolectomy was performed with a 4F and 5F fogarty valsalva maneuver. In addition, recently, Pharmaco-Mechanical Thrombectomy (PMT) (Clean Rotational Thrombectomy System, Argon Medical Devices, Plano, TX) has been applied in acute and proximal DVT cases [4]. Patients who underwent mechanical thrombectomy were also given r-tPA treatment after the application. A temporary inferior vena cava filter was inserted in all these cases and removed one day after the procedure. In addition, in cases with recurrent multiple PE history, persistent vena cava inferior and/or ilio-femoral vein thrombosis despite appropriate anticoagulation, a permanent filter (Titanium-Greenfield filters) was placed in the inferior vena cava via the jugular vein (Figure 3). The diagnosis of PE was made by history, clinical findings, electrocardiography, echocardiography, lung ventilation-perfusion scintigraphy, spiral computed tomography angiography and pulmonary angiography. In patients with pulmonary embolism, if hemodynamic instability was present, emergency pulmonary embolectomy was decided and pulmonary embolectomy was performed with median sternotomy under extracorporeal circulation. Oral anticoagulant treatment was given to all patients in whom medical, thrombolytic, surgical embolectomy and PMT treatment was initiated. Recently, oral anticoagulants dabigatran, rivaroxaban, apixaban, edoxaban have been used instead of warfarin, which requires monitoring of INR. All patients were discharged by recommending continuous use of compression stockings (with a pressure of 30 mm Hg below the knee) for at least 6 months.
Results
In this study, which we discussed in order to convey our clinical experiences on diagnosis, treatment and prophylaxis in DVT, first of all, it was tried to identify known risk factors in the formation of venous thrombosis. While the most common reason in our patients is DVT, which occurs after orthopedic interventions; the least reason was venous thrombosis due to excessive external effort (Paget-Schroetter Syndrome). Apart from these factors, immobilization, cardiopulmonary disease, direct trauma, varicose veins, malignancy, pregnancy, use of oral contraceptives, protein-C, protein-S and antithrombin-III deficiency and unidentified causes (possible hereditary reasons) were found to be effective. The cause could not be determined in 18 patients. Table 1 shows the risk factors for our patients.
398 of our patients (96,6%) consisted of symptomatic cases. Pain, swelling and redness were the most prominent complaints in these patients. There was no obvious complaint in 14 patients. Homans test was positive in 296 cases (71,8%). There was a significant difference in diameter between above and below the knee in all patients. Daily measurements were made in terms of the effectiveness of the treatment. The most common extremity was the left lower extremity (191; 46,4%); it was seen in the right lower 144, left upper 44 and right upper 33 patient extremities. Exercise thrombosis (Paget-Schroetter Syndrome) was caused by overuse in 6 of the upper extremity DVT cases. In all cases, the diagnosis of DVT was made with color DUS in the light of clinical findings. After 7-10 days of treatment, Doppler USG was performed to determine the nature of the thrombus (acute, sub-acute and chronic), and it was decided to continue anticoagulation therapy parenterally or orally according to the condition of the thrombus. Venography or magnetic resonance imaging methods were not performed in any of our patients for diagnostic purposes. Patients who achieved significant improvement in the symptoms and clinical findings of the cases, who did not develop complications and did not undergo additional interventions, were discharged with warfarin sodium by keeping INR levels between 2-3 for maintenance treatment.
Table 1: Risk factors for our patients.
Etiologies |
n=412 |
% |
Bone fractures |
36 |
8,7 |
Hip/knee prosthesis |
30 |
7,3 |
Arthroscopic knee surgery |
38 |
9,2 |
Major trauma and injuries |
26 |
6,3 |
Spinal cord injuries |
21 |
5,1 |
Major general surgery operations |
19 |
4,6 |
Paget-Schroetter Syndrome |
6 |
1,5 |
Hormone therapy |
21 |
5,1 |
Contraceptive pills |
23 |
5,6 |
Stroke/paralysis |
22 |
5,3 |
Malignancy |
32 |
7,8 |
Genetic predisposition to coagulation |
24 |
5,8 |
Pregnancy/Postpartum period |
15 |
3,6 |
Drug therapy in cancer |
14 |
3,4 |
Heart and respiratory failure |
7 |
1,7 |
Central venous catheters |
17 |
4,1 |
Advanced age |
9 |
2,2 |
Varicous vein |
8 |
1,9 |
Obesity |
12 |
2,9 |
Laparoscopic surgery |
14 |
3,4 |
Unidentified cause |
18 |
4,4 |
Patients taking standard and LMW heparin
All our patients started to receive heparin therapy (standard or LMWH) from the first day. While SH (n=88) was given to our patients (generally between 2009-2012) in the first years; LMWH (n=324) was given frequently after 2012. SH has also been preferred in patients with acute and high diameter difference (between the knee and below the knee), and in patients with a prominent clinical phlegmasia cerulea dolens-phlegmasia alba dolens as intermittent or continuous therapy. In the color DUS examination repeated on the 10th day of the initial treatment, it was observed that flow started with partial recanalization in 166 (40,3%) of the patients in both groups receiving heparin. On the other hand, 172 (41,7%) had venous flow showing a flow pattern, but significant obstruction continued. While normal venous flow was provided with complete recanalization in 16 of the cases (3,9%), there was no significant change in the color DUS image in 58 (14,1%). Of these 16 patients who showed complete recanalization, 9 were receiving LMWH and 7 were receiving SH. Of the partially recanalized patients, 82 (49,4%) were receiving LMWH and the others were receiving SH. Recurrent venous thrombosis developed in 12 of these patients who received SH and in 8 of those who received LMWH. Pulmonary embolism occurred in 5 patients who received SH and 3 patients who received LMWH. One of each was lost in these patients. After the patients were discharged from the hospital, they were called for control with monthly intervals. Patients with partial recanalization, partial obstruction and complete recanalization were followed up in terms of edema-diameter difference, pain, and redness in the leg. During the control, the development process of venous thrombosis was followed by doppler USG, if deemed necessary or at least in 3-month periods. Patients were called for control with 6-month and 1-year follow-up. Most of the patients who showed recanalization were the patients who were started using heparin and oral anticoagulants, which we have been using in recent years. In addition, it was determined that these patients had a higher INR value, which indicates the effectiveness of oral anticoagulants. There were no significant differences in terms of bleeding in patients who were given heparin therapy. Conditions such as hematoma, hematemesis, hematuria, epistaxis, ecchymosis of the skin, hematurias were observed in 11 patients who were given SH and 13 patients who were given LMWH heparin. There was no significant difference in terms of hospital stay in both heparin groups.
Patients treated with thrombolytic therapy
Traditional treatment of DVT is in the form of oral anticoagulants following anticoagulation with heparin. Anticoagulant therapy effectively prevents thrombus spread and embolization, but has no thrombolytic effect. Lack of thrombolytic effect in patients who are anticoagulated with only heparin causes deterioration of venous valve functions and prevention of venous return in 2/3 of the patients and causes post-phlebitic syndrome in half of the patients [5]. Thrombolytic therapy has the potential to preserve valve function and prevent the development of postphlebitic syndrome because it dissolves the clot formed [5,6]. For this reason, thrombolytic therapy has been used in DVT treatment for a long time in many centers. In many comparative studies conducted since the late 1960s, it has been shown that thrombolytic therapy has a significant advantage in restoring venous flow compared to anticoagulation with heparin [6]. Although the efficacy of thrombolytic therapy in DVT treatment is widely accepted, the potential for catastrophic bleeding, uncertainty in dosage and administration, and the belief that extensive laboratory facilities are required for its implementation limit its use. Thrombolytic therapy was administered in 66 (16,1%) patients not exceeding 7 days from the onset of symptoms. In the treatment, recombinant tissue plasminogen activator (rt-PA) was administered to 55 patients; streptokinase was administered to 8 patients, and urokinase to 3 patients. These patients were often patients with plagmasia clinic and severe edema in the leg. The most commonly used agent was rt-PA (Actilyse-Boehringer Ingelheim). According to clinical and Doppler USG findings, success was achieved in 54 (81,8%) of these patients who received fibrinolytic therapy. The cases with no success were those who were applied late. During fibrinolytic therapy, complications such as bleeding, mild fever, pulmonary embolism and distal embolism may be encountered. Local bleeding often occurred at the procedure site in our patients. Boysen et al reported hemorrhagic complications in 10 (53%) of 19 patients who received rt-PA [7]. There was a marked decrease in the complaints in the patients we gave thrombolytic therapy. When examined in terms of complications; no clinical pulmonary embolism was detected in any of the patients. Clinically, bleeding developed in one patient who was given streptokinase. Bleeding was in the form of hematoma development in the lumbar disc operation area 1 month ago. It was taken under control in a short time with fresh frozen plasma and did not cause any additional problems. Urokinase infusion was started in 2 cases that were given streptokinase because of allergic reaction. Surgical thrombectomy was performed in 1 case with thrombosis up to the inferior vena cava. Patients who received thrombolytic therapy were called for controls once every 15 days in the first months, and then monthly. These patients were examined in terms of recurrence and pulmonary embolism, as well as physical examination, radiological follow-up with DUS. Venous insufficiency occurred in the months after the procedure in all patients with complete recanalization. Medical anticoagulant treatment was continued in cases with partial success and without complete sewage.
Patients undergoing surgical embolectomy
During their treatment, a total of 10 (2,4%) patients, 5 of whom were young and 5 with massive iliofemoral vein thrombosis and 5 elderly patients who did not respond to heparin therapy and were condensed to thrombolytic therapy, had no clinical improvement despite thrombolytic therapy. Venous thrombectomy was performed on days. For surgical embolectomy performed using a fogarty catheter with the valsalva maneuver from the femoral vein, a vena cava filter was placed in the patients before the procedure. While thrombectomy was successful in 3 cases, it was not successful in the elderly patient who was tried thrombectomy on the tenth day because the thrombus was organized and adhered to the surrounding. Two elderly patients died during the postoperative period. No complications such as recurrence or pulmonary embolism were observed in the follow-up of patients whose thrombectomy was successful and who took oral anticoagulants.
Patients undergoing Pharmaco-Mechanical Thrombectomy (PMT)
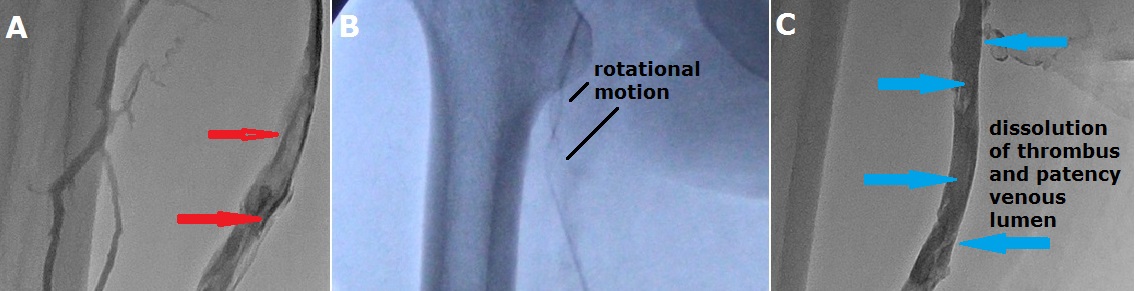
Pharmaco-mechanical thrombectomy, which has been used in recent years, has also been used in our patients with acute DVT (Figure 4,5). In these patients, the procedure was performed after placing a filter in the vena cava inferior. It was applied to a total of 44 (10,7%) patients and success was achieved in 40 patients. Thrombolytic therapy was also given to the patients after this procedure. No death or symptomatic pulmonary embolism occurred during the procedure. After the procedure, there was a thrombus stuck in the vena cava filter in 4 patients. There were no systemic bleeding complications. Minor bleeding occurred at the popliteal entry site in 9 patients (20,4%), blood transfusion occurred in 2 patients. Balloon application was required in 14 patients (31,8%) who underwent mechanical thrombectomy to the superficial femoral vein. The patients were discharged with oral anticoagulant treatment the day after the procedure and were followed up.
Patients undergoing pulmonary embolectomy
Surgical pulmonary embolectomy was decided in 8 (1,9%) patients who developed pulmonary embolism due to clinical and hemodynamic instability. A vena cava filter was placed in these patients before the surgery. Six patients undergoing emergency surgery died due to bleeding and low cardiac output during surgery.
Despite adequate anticoagulant therapy, 120 patients (29,1%) with a history of multiple previous PE and persistent vena cava inferior and/or iliofemoral vein thrombosis had a vena cava inferior filter under fluoroscopy (Excluding PMT and surgical thrombectomy patients). The patients were followed up for 15 days at first and then monthly. In addition to anamnesis and physical examination, the state of thrombi in the deep veins was examined with DUS. These patients who were given oral anticoagulant treatment were followed up monthly. There were no signs of pulmonary embolism during the follow up.
Discussion
Today, DVT continues to be a serious problem by causing pulmonary embolism, venous gangrene and chronic venous insufficiency, although both medical and surgical treatment possibilities are advanced. In other words, DVT is an important group of diseases that can have high morbidity and mortality not only with its own morbidity, but also with the complications it causes and the complications that can be brought about by its treatment [2]. DVT can cause asymptomatic or silent findings to catastrophic clinical findings such as pulmonary embolism that can be life-threatening [8].
Due to the presence of asymptomatic cases, the true incidence of the disease is not fully known. It is a disease that affects 1 in every 1000 inpatients in Europe and the United States. In one study, the prevalence of DVT in hospitalized patients was found to be 1,0%, and in autopsies it was 14,6%. While this rate is 1/100,000 in the early stages of life, it reaches a frequency of 1/100 in advanced ages [9,10]. In our study, the mean age of our patients was 49 ± 19 years, and 60% of the cases were determined to be 40 years or older, making the opinion that advanced age is a risk factor for DVT, in our opinion, makes it an accepted approach.
The triad of hypercoagulability, venous stasis and vascular intima damage suggested by Virchow for the development of venous thrombosis is still valid today. Over-tendency to systemic coagulation may be inherited or acquired. Generally, these thrombophilic conditions alone do not lead to a clinical thrombotic event unless there is a second risk factor or a predisposing condition. Etiological factors may also be effective such as Factor V Leiden mutation (activated protein C resistance), deficiencies of circulating anticoagulants (Protein C, protein S, Antithrombin-III), prothrombin gene mutation, abnormalities in fibrin destruction, presence of lupus-related anticoagulants, increased procoagulant activity (malignant diseases and severe trauma) [11-13]. If there is a family history in young DVT cases, the history of sudden venous thrombosis and miscarriages and stillbirths in pregnancies should be investigated [14]. In this retrospective review of DVT etiology, we could not identify a specific risk factor in our 18 cases. In the hematological examination performed for thrombophilic conditions, we have seen that genetic disorders are effective in etiology in some of our patients. However, we thought that a genetic thrombophilic condition may be effective as a risk factor in some young patients whose cause has not been determined.
Surgical intervention is the most important acquired risk factor for DVT. Thrombogenic factors in surgery; venous stasis due to immobilization, the effect of anesthetic agents, tissue trauma and tissue factors released as a result of endothelial damage increase the tendency to coagulate. Surgical patients at risk of developing DVT are divided into three groups: (1) Low-risk patients; these are cases under 40 years of age and without systemic disease. The surgery is completed in less than 60 minutes and without complications. The risk of developing DVT is less than 2% and the probability of proximal progression is less than 1%. (2) Moderate risk patients; these are cases over the age of 40 who are treated under general anesthesia for longer than 60 minutes. They also carry a number of risk factors such as cancer, obesity, enlarged veins, bed rest or heart failure. If prophylaxis is not given, the risk of developing DVT is 10-40 % and proximal spread is 2-8 % in these cases. The risk of fatal pulmonary embolism is 1%. (3) High risk cases; those with a history of DVT and pulmonary embolism. These are the ones who are exposed to abdominal or pelvic intervention due to some major pathology and some orthopedic indications. The risk of developing leg DVT without prophylaxis is between 40-80 %. The proximal spread is between 10-20 % and the risk of pulmonary embolism can reach up to 5% [15-18]. In some studies, the presence of immobilization in the postoperative period after surgery has been shown as an important risk factor for venous thrombosis [19,20]. It was determined as the most common etiological risk factor of orthopedic surgical procedures among our patients. These patients were mostly in the moderate risk patient group and mostly venous thrombosis occurred after applications to the pelvic, thigh and knee joints. After orthopedic surgery and immobilization, it was determined in our patients that the most common reasons were hormonal therapy (oral contraceptives), pregnancy and genetic reasons (protein C and S deficiency). It has been observed that such reasons increase the risk of venous thrombosis 3-7 times [21-24].
DVT often occurs in the calf vein (posterior tibial vein), the popliteal vein, and the common femoral vein of the thigh. While the calf vein is the most common DVT, only 15-20 % of the cases have proximal spread. The left lower extremity suffers from disease more often than the right [25,26]. Demir et al. in the cases they followed up, the left lower extremity was determined as the most common location of DVT (59,6%) [20]. Venous thrombosis was observed most frequently in the left lower extremity in our patients. Central venous catheter, malignancy, hypercoagulability conditions, radiation, thoracic outlet syndrome, intravenous drug use and peripheral venous access are the main risk factors in the location of DVT in the upper extremity [27,28]. In most of our upper extremity venous thrombosis cases, the cause was catheter-related thrombosis and malignancy. Effort thrombosis was the least observed cause.
Doppler USG came into prominence as the main diagnostic method in our study. Doppler USG has been the main method of investigation in evaluating the presence or absence of DVT and response to treatment. Failure to collapse in Doppler USG or demonstration of intravenous echoes is among the findings of DVT. Generally, there is 89-98 % sensitivity for proximal DVT [29,30]. In our study, the diagnosis was supported by DUS in all patients diagnosed with DVT based on anamnesis and physical examination findings. Although we think that anamnesis and physical examination have a high value in diagnosis, we can say that it should be preferred in the first place in non-invasive and highly reliable DUS. While Doppler USG was used both as a diagnostic tool in all our patients, ultrasonography was also used to evaluate the response of the patients to treatment and to follow up.
There are many diseases that have similar symptoms and signs to DVT. Differential diagnosis is especially important in thrombosis in the calf veins. Reported that the Baker cyst mimicked the symptoms of DVT [31,32]. Langsfeld et al detected Baker cyst with venous DUS in 3,1% of the cases they were followed up with the pre-diagnosis of DVT [33]. Among our patients, some patients who were thought to have venous thrombosis during outpatient examination were diagnosed with Baker's cyst and were referred to the orthopedic clinic. There are also patients who are evaluated with a pre-diagnosis of deep venous thrombosis and diagnosed with superficial thrombophlebitis, venous reflux, lymphedema, hematoma, and congestive heart failure.
Regarding the natural course of DVT after its diagnosis, treatment should be started immediately because of its complications such as PE, which is a fatal complication, and post-thrombophlebitic syndrome, which has extremely high morbidity in the long term [34]. Systemic anticoagulation reduces the mortality and morbidity of DVT [35]. In cases of proximal DVT, the risk of post-thrombophlebitic syndrome and PE is greater, which outweighs the risks associated with anticoagulation. The probability of severe PE in calf DVT is relatively low and the need for systemic anticoagulation is less. Whether isolated calf DVT should be treated with systemic anticoagulation or through analgesia and local measures is still a matter of debate today. Kurtoglu states that symptomatic DVT detected in calf veins should be treated with anticoagulation [34]. Hull and Lohr reported that in patients with symptomatic calf DVT, if left untreated, the risk of thrombosis spreading proximally is 20-30 % [36,37]. In our study, the majority of the cases were proximal DVT cases. All patients who underwent pharmaco-mechanical thrombectomy, thrombolytic therapy, surgical thrombectomy, and pulmonary embolectomy were proximal DVT cases. Patients who developed recurrence and died due to PE were also patients with iliac or vena cava thrombosis.
SH, LMWH and warfarin sodium, which are among the anticoagulant drugs group, are the main components of DVT treatment. After heparin came into use after the 1940s, LMWHs have been used more frequently in the last 10 years. DVT cases can be effectively treated with heparin given by continuous IV infusion or SC injection [38]. Many studies have been conducted on the use of SH and LMWH in the initial treatment of DVT and are still being done [39,40]. LMWHs have taken the place of SH in the treatment and protection of DVT in many countries. Harenberg et al emphasized that classical heparin should be preferred first in the initial treatment of proximal DVT [41]. On the other hand, in another study conducted with enoxaparin, it was stated that LMWHs can be used safely without the need for daily laboratory follow-up, and they are an easy-to-apply and effective method in the treatment of DVT [42]. In patients diagnosed with venous thrombosis, long-term anticoagulants should be used to ensure that the treatment is effective and the disease does not recur. Lifelong treatment should be considered especially in thrombophilic patients with atypical localization [43,44]. In a study comparing LMWH (tinzaparin sodium) and continuous IV heparin in the treatment of proximal DVT, patients treated with LMWH had better results in terms of survival and major bleeding compared to SH patients, and LMWHs were at least as effective and safe as classical heparins, It has been stated that it is easy to use and will provide outpatient treatment for patients with uncomplicated proximal DVT [45]. Gonzalez-Fajardo et al compared warfarin sodium with enoxaparin in their study and found a lower incidence of symptomatic DVT recurrence and bleeding in patients who received enoxaparin [46]. It was observed that SH were used in the first years and LMWHs were used in the last years for the patients we presented in our study. There was no difference in efficacy and complications in both patient groups. No difference was found between LMWH and SH in terms of recurrence thrombosis, bleeding, length of stay, recurrent and massive PE. Although it is outside of our field of study, the length of stay has decreased significantly due to outpatient follow-up and treatment without hospitalization with LMWH. In line with these findings, we would like to emphasize that although LMWHs are more expensive drugs, they do not require medical personnel, shorten the length of hospital stay, are easy to use, do not require laboratory follow-up, they have reliable and sufficient efficacy. We think that it should be the first choice in its treatment and prophylaxis.
It has been reported that with fibrinolytic therapy, venous functions are better preserved in the short and long term, especially as a result of rapid and complete clearance of iliofemoral vein thrombosis. Therefore, in our study, we preferred fibrinolytic therapy in cases where there were no contraindications and in cases where fibrinolytic drug was available, with patient approval. Fibrinolytic therapy was generally preferred in patients with severe thrombosis such as VCI or iliofemoral vein thrombosis or phlegmacia cerulea dolens. Successful patients were generally seen to be patients receiving treatment for 5-9 days. Success in treatment was 60-75 % if it was applied for 5-7 days, while it was observed that this rate decreased to 0-33 % within 7-21 days [47,48]. We found that thrombolytic therapy in the form of intravenous infusion is easily applicable and safe and significantly increases the rate of recanalization compared to infusion with heparin. We determined that patients who applied to thrombolytic treatment at an early period especially benefited.
Venous thrombectomy was performed in 10 cases with massive iliofemoral vein thrombosis who did not show clinical improvement despite thrombolytic therapy and who had thrombolytic therapy. While thrombectomy was successful in 3 cases, it was not successful in an elderly patient who was tried thrombectomy on the tenth day because the thrombus was organized and adhered to the surrounding. No complications such as recurrence or pulmonary embolism were observed in the follow-up of the patients whose thrombectomy was successful and who received oral anticoagulants. Decousus et al. reported that the use of permanent vena cava filters reduced the development of asymptomatic or symptomatic PE without major complications in their study consisting of 400 cases, which they conducted in the direction of the effectiveness and reliability of vena cava filters in preventing PE in patients with proximal DVT [49]. In our study, despite adequate anticoagulant therapy, the vena cava inferior filter was placed under scopy in high-risk patients with multiple previous PE history, contraindicated or unsuccessful anticoagulant therapy, and in patients with vena cava inferior and/or iliofemoral vein thrombosis. PE was not found in the 6 months-1 year follow-up of these cases.
An important complication of DVT of the lower extremities is PE. In up to 90% of patients, thrombus arises from the proximal veins of the lower extremities, and the remainder mainly from the pelvic veins. Heparin anticoagulation is the preferred method in most cases in the treatment of PE. In acute PE cases where hemodynamic impairment is not seen, LMWHs have been shown to be as effective as conventional heparin [50]. Thrombolytic therapy takes an important place especially in PE cases where right ventricular functions are impaired. In addition, in cases where the pulmonary artery is anatomically occluded for 50% or more, namely major pulmonary embolism or cardiac arrest or severe arterial hypotension, massive PE, the patient can be lost in a very short time (50% is 30 minutes, 70% is 60 minutes, and 85% within 6 hours) may be indicated for pulmonary embolectomy in treatment [51]. In many articles, mortality rates in pulmonary embolectomies performed using cardiopulmonary bypass are reported between 11-60 % [51,52]. Meyer et al. recommends to try thrombolytic therapy first in massive PE if there are no contraindications, and if there is a contraindication, surgical intervention is not available for patients who do not have time for medical treatment or who do not improve despite intensive medical treatment [53]. Surgical indication is accepted by most surgeons for the treatment of irreversible dyspnea in critically ill patients with massive PE with uncorrectable shock and pulmonary hypertension due to acute or chronic pulmonary embolism [54]. Pulmonary embolectomy was applied to 8 patients who developed massive PE in our clinic, 6 of them died due to cardiopulmonary insufficiency in the early postoperative period.
In the last decade, PMT has been used as an effective alternative to surgical thrombectomy and catheter thrombolysis in patients with acute DVT. Although several studies have shown that PMT can be used successfully, the long-term benefits of this treatment are still under study [55,56]. In general, PMT catheters have been recommended to be used in combination with auxiliary thrombolytic agents for faster thrombus destruction [57]. These combination therapies result in lower average dosage, lytic infusion time, lower overall cost, and reduction in hemorrhagic complications [57,58]. In addition, it is important to reduce the socioeconomic cost of post-thrombotic complications after DVT. Venous ulcers develop in up to 80-90 % of patients with acute DVT over the years, and therefore patients have required readmissions and various medical and surgical procedures to alleviate the complaints. In addition, patients with chronic venous disease cause labor loss. We preferred to use Cleaner thrombectomy device in our patients with acute DVT. This device is a battery operated percutaneous thrombectomy catheter that functions by rotating a flexible S-shaped guidewire inside the vessel to be treated. This device softened and aspirated the clot through an introducer sheath. PMT, to which lytic agents were added, restored the venous patency in the operating room or intervention room, eliminating the need for an intensive care unit stay or repeat venography. Although we did not analyze the cost, it is seen that PMT cost is less than other DVT treatment methods in the early and long term. Most of our patients who underwent PMT had success and no obvious complications were observed. There were no deaths or complications of pulmonary embolism.
Conclusion
In patients diagnosed with deep vein thrombosis, the etiology should be questioned according to the literature and our personal experiences, and the diagnosis should be supported by Doppler ultrasound. Early diagnosis, rapid and effective treatment is important in terms of the course of the clinical picture and prevention of complications. In addition, the use of low molecular weight heparin is equally effective as an intravenous standard heparin infusion in the initial treatment of deep vein thrombosis. However, if indicated, fibrinolytic agents are more beneficial for pulmonary embolism than heparin. Medical treatment should be applied quickly and adequately to reduce the rate of surgery in patients with pulmonary embolism. Success in venous thrombectomy and fibrinolytic therapy is closely related to the timing of the treatment. Vena cava filters can be used safely in patients at risk of preventing pulmonary embolism. Recently, PMT should be applied in patients indicated for acute cases and proximal DVT cases. It is of great importance to start oral anticoagulant therapy with heparin and to reach the effective INR dose in order to prevent complications related to deep vein thrombosis in the long term.
Acknowledgements
Consent: Written informed consent was obtained from the patient’s legal guardians/parents for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Declaration of patient consent: The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
References
- Virchow R. Neuer fail von todlicher emboli der lungenraterie. Arch Pathol Anat. 1856; 10: 225-228.
- Rahaghi FN, Minhas JK, Heresi GA. Diagnosis of Deep Venous Thrombosis and Pulmonary Embolism: New Imaging Tools and Modalities. Clin Chest Med. 2018; 39: 493-504.
- Velioglu Y, Erkut B, Unlu Y, Becit N, Ceviz M, Kocak H. Deep Venous Thrombosis and Treatment Procedures (Experience with 505 Patients). Turkish J Vasc Surg. 2006; 15: 1-13.
- CLEANER™ Rotational Thrombectomy System. 2021.
- Turpie GG, Levine MN, Hirsh J, Ginsberg JS, Cruickshank M, Jay R, et al. Tissue plasminogen activator (rt-PA) vs heparin in deep venous thrombosis. Results of a randomized trial. Chest. 1990; 97: 172S-175S.
- Rogers LQ, Lutcher CL. Streptokinase therapy for deep vein thrombosis: a comprehensive review of the english literature. Am J Med. 1990; 88: 389-395.
- Boysen G. Overview on thrombolysis in acute ischemic stroke. Fibrinolysis. 1995; 9: 29-32.
- Stoeva N, Staneva M, Kirova G, Bakalova R. Deep venous thrombosis in the clinical course of pulmonary embolism. Phlebology. 2019; 34: 453-458.
- Okuhara A, Navarro TP, Procópio RJ, Bernardes RC, Oliveira LC, Nishiyama MP. Incidence of deep vein thrombosis and quality of venous thromboembolism prophylaxis. Rev Col Bras Cir. 2014; 41: 2-6.
- Cook D, Crowther M, Meade M, Rabbat C, Griffith L, Schiff D, et al. Deep venous thrombosis in medical-surgical critically ill patients: prevalence, incidence, and risk factors. Crit Care Med. 2005; 33: 1565-1571.
- Franco RF, Reitsma PH. Genetic risk factors of venous thrombosis. Hum Genet. 2001; 109: 369-384.
- Mitat T, Kimura R, Kokubo Y, Sakata T. Genetic risk factors for deep vein thrombosis among Japanese: importance of protein S K196E mutation. Int J Hematol. 2006; 83: 217-223.
- Milio G, Siragusa S, Minà C, Amato C, Corrado E, Grimaudo S, et al. Superficial venous thrombosis: prevalence of common genetic risk factors and their role on spreading to deep veins. Thromb Res. 2008; 123: 194-199.
- Nagato LC, de Souza PMA, de Godoy JM, Souza DR. Association of proximal deep venous and Apo E genetic polymorphisms. J Thromb Thrombolysis. 2012; 33: 116-119.
- Herold CJ, Bankier AA, Burghuber OC. Pulmonary embolism: Pulmonary vascular disorders, vasculitides and hemorrhage. In: Albert RK, Spiro SG, Jett JR (eds). Comprehensive Respiratory Medicine. Philadelphia: Mosby. 1999; 1-12.
- Wang P, Kandemir U, Zhang B, Wang B, Li J, Zhuang Y, et al. Incidence and Risk Factors of Deep Vein Thrombosis in Patients With Pelvic and Acetabular Fractures. Clin Appl Thromb Hemost. 1999; 25: 1076029619845066.
- Porfidia A, Porceddu E, Feliciani D, Giordano M, Agostini F, Ciocci G, et al. Differences in Clinical Presentation, Rate of Pulmonary Embolism, and Risk Factors Among Patients With Deep Vein Thrombosis in Unusual Sites. Clin Appl Thromb Hemost. 2019; 25: 1076029619872550.
- Tick LW, Kramer MH, Rosendaal FR, Faber WR, Doggen CJ. Risk factors for post-thrombotic syndrome in patients with a first deep venous thrombosis. J Thromb Haemost. 2008; 6: 2075-2081.
- Kılıç M. Etiopathogenesis in Deep Vein Trombosis. Akdeniz University Faculty of Medicine, Department of Thoracic and Cardiovascular Surgery. Thesis. Antalya. 1990.
- Demir I, Ekim H, Demircan A. Effectiveness of Surgical Treatment in Deep Vein Trombosis. Haydarpaşa Cardiology and Cardiovascular Surgery Bulletin. 1993; 1: 70-75.
- Citruk-Ware R. Hormonal contraception and thrombosis. Fertil Steril. 2016; 106: 1289-1294.
- Tanis BC, Rosendaal FR. Venous and arterial thrombosis during oral contraceptive use: risks and risk factors. Semin Vasc Med. 2003; 3: 69-84.
- Hotoleanu C. Genetic Risk Factors in Venous Thromboembolism. Adv Exp Med Biol. 2017; 906: 253-272.
- Lefrançois C, Derlon A, Le Querrec A, Lochu T, Sillard B, Deshayes JP., et al. Hereditary deficiency of antithrombin III, protein C and protein S. A study of 31 patients from 8 unrelated families. Ann Fr Anesth Reanim. 1990; 9: 485-494.
- Liu XC, Chen XW, Li ZL, Wang SC, Chen C. Anatomical distribution of lower-extremity deep venous thrombosis in patients with acute stroke. J Stroke Cerebrovasc Dis. 2020; 29: 104866.
- Luo Z, Chen W, Li Y, Wang X, Zhang W, Zhu Y, et al. Preoperative incidence and locations of deep venous thrombosis (DVT) of lower extremity following ankle fractures. Sci Rep. 2020; 10: 10266.
- Sindhu KK, Cohen B, Blood T, Gil JA, Owens B. Upper Extremity Deep Venous Thrombosis Prophylaxis after Elective Upper Extremity Surgery. Orthopedics. 2018; 41: 21-27.
- Mai C, Hunt D. Upper-extremity deep venous thrombosis: a review. Am J Med. 2019; 124: 402-407.
- Pinto SM, Yassin M, Galang G. Cost-Effectiveness Analysis of Routine Venous Doppler Ultrasound for Diagnosis of Deep Venous Thrombosis at Admission to Inpatient Rehabilitation. Am J Phys Med Rehabil. 2018; 97: 747-753.
- Valentin ML, Clemens R, Thalhammer C. Duplex ultrasound of deep vein thrombosis of the leg. Dtsch Med Wochenschr. 2016; 141: 946-949.
- Tejero S, Fenero-Delgado BT, López-Lobato R, Carranza-Bencano A. Ruptured Baker’s cyst: complications due to misdiagnosis. Emergencias. 2018; 30: 412-414.
- Hoşgör I, Sarı H. Baker cyst mimicking the symptoms of deep vein thrombosis. Turkish Thoracic and Cardiovascular Surgery Journal. 2000; 8: 550-553.
- Langsfeld M, Matteson B, Johnson W, Wascher D, Goodnough J, Weinstein E. Baker’s cysts mimicking the symptoms of deep vein thrombosis: diagnosis with venous duplex scanning. J Vasc Surg. 1997; 25: 658-662.
- Kurtoǧlu M. Venöz tromboembolizm: tanı-tedavi. Flebology Journal. 2002; 1: 21-29.
- Nicolaides AN, Fareed J, Kakkar AK, Comerota AJ, Goldhaber SZ, Hull R, et al. Prevention and treatment of venous thromboembolism--International Consensus Statement. Int Angiol. 2013; 32: 111-260.
- Hull RD, Hirsh J, Carter C, Jay RM, Ockelford PA, Buller HR. et al. Diagnostic efficacy of impedance plethysmography for clinically suspected deep-vein thrombosis: a randomized trial. Ann Intern Med. 1985; 102: 21-28.
- Lohr JM, Kerr YM, Lutter KS, Cranley RD, Spirtoff K, Cranley JJ, et al. Lower extremity calf thrombosis: to treat or not to treat. J Vasc Surg. 2016; 14: 618-623.
- Russell DH, Graham FP. Medical treatment of acute deep venous thrombosis. In: Gloviczki P, Yao JSY (eds). Handbook of Venous Disorders. London: Arnold. 2001; 184-193.
- Li A, Garcia DA, Lyman GH, Carrier M. Direct oral anticoagulant (DOAC) versus low-molecular-weight heparin (LMWH) for treatment of cancer associated thrombosis (CAT): A systematic review and meta-analysis. Thromb Res. 2019; 173: 158-163.
- Robinson R, Wirt TC, Barbosa C, Amidi A, Chen S, Joseph RM, et al. Routine Use of Low-Molecular-Weight Heparin for Deep Venous Thrombosis Prophylaxis after Foot and Ankle Surgery: A Cost-Effectiveness Analysis. J Foot Ankle Surg. 2018; 57: 543-551.
- Harenberg J, Schmidt JA, Kopenhagen K, Tolle A, Huisman MV, Büller HR, et al. Fixed dose body weight independent subcutaneous low molecular weight heparin versus adjusted dose unfractionated intravenous heparin in the initial treatment of proximal venous thrombosis. Thromb Haemost. 2000; 83: 652-656.
- Kurtoǧlu M, Dolay K, Güloǧlu R. Düşük Molekül Aǧırlıklı Heparin (Enoksaparin) Ile Proksimal Derin Ven Trombozu Tedavisi (erken Sonuçlarımız). Ulusal Cerrahi Dergisi. 1998; 14: 346-352.
- Wypasek E, Undas A. Protein C and protein S deficiency - practical diagnostic issues. Adv Clin Exp Med. 2013; 22: 459-467.
- van den Belt AG, Sanson BJ, Simioni P, Prandoni P, Büller HR, Girolami A, et al. Prins MH. Recurrence of venous thromboembolism in patients with familial thrombophilia. Arch Intern Med. 1997; 157: 2227-2232.
- Hull RD. Raskob GE, Pineo GF, Green D, Trowbridge AA, Elliott CG, et al. Subcutaneus low- molecular-weight heparin compared with continuous intravenous heparin in the treatment of proximal vein thrombosis. N Engl J Med. 1992; 326: 975-982.
- Gonzalez JA, Arreba E, Castrodeza J, Perez JL, Fernandez L, Agundez I, et al. Venographic comparison of subcutaneous LMWH with oral anticoagulant therapy in the long-term treatment of deep venous thrombosis. J Vasc Surg. 1999; 30: 283-292.
- Mannuccio MP, Altieri D, Mari D. Thrombolytic therapy in acute deep venous thrombosis of the lower limbs: selection, indications and limits. Minerva Chir. 1987; 42: 2031-2035.
- Albuquerque LC, Silveira F, Zago AJ, Bettio J, Petracco JB. Thrombolytic therapy in deep venous thrombosis. Initial clinical experience. Arq Bras Cardiol. 1997; 68: 125-128.
- Decousus H, Leizorovicz A, Parent F, Page Y, Tardy B, Girard P, et al. A clinical trial of vena caval filters in the prevention of pulmonary embolism in patients with proximal deep-vein thrombosis. N Engl J Med. 1998; 338: 409-415.
- Simonneau G, Sors H, Charbonnier B, Page Y, Laaban JP, Azarian R, et al. A comparison of low-molecular-weight heparin with unfractionated heparin for acute pulmonary embolism. The THESEE Study Group. Tinzaparine ou Heparine Standard: Evaluations dans l’Embolie Pulmonaire. N Engl J Med. 2018; 337: 663-669
- Mattox KL, Feldtman RW, Beal AC, DeBakey ME. Pulmonary embolectomy acut massive pulmonary embolism. Ann Surg. 1982; 195: 726-731.
- Goldhaber SZ, Morpurgo M. Pulmonary embolism. Supplement, treatment and prevention. JAMA. 1993; 6: 424-432.
- Meyer G, Yamisier D, Sors H, Stern M, Vouhé P, Makowski S, et al. Pulmonary embolectomy: a 20 -year experience at one center. Ann Thorac Surg. 1991; 51: 232-236.
- Stuiz P, Schiapfer R, Feer R, Habicht J, Grädel E. Decision making in the surgical treatment of massive pulmonary embolism. Eur J Cardiothorac Surg. 1994; 8: 188-193.
- O’Sullivan GJ, Lohan DG, Gough N, Cronin CG, Kee ST. Pharmacomechanical thrombectomy of acute deep vein thrombosis with the Trellis-8 isolated thrombolysis catheter. J Vasc Interv Radiol. 2007; 18: 715-724.
- Köksoy C, Yilmaz MF, Başbuğ HS, Calik E, Erkut B, Kaygin MA, et al. Pharmacomechanical thrombolysis of symptomatic acute and subacute deep vein thrombosis with a rotational thrombectomy device. J Vasc Interv Radiol. 2014; 25: 1895-900.
- Calik E, Dag O, Kaygin MA, Onk OA, Erkut B. Pharmacomechanical Thrombectomy for Acute Symptomatic Lower Extremity Deep Venous Thrombosis. The Heart Surgery Forum. 2015; 18: 178-183.
- Bush RL, Lin PH, Bates JT, Mureebe L, Zhou W, Lumsden AB. Pharmacomechanical thrombectomy for treatment of symptomatic lower extremity deep venous thrombosis: safety and feasibility study. J Vasc Surg. 2004; 40: 965-970.