Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Mesotherapy vs PRP: Hair loss
Elahe Najafizade1; Abdol Karim Jamali2; Seyed Ali Hossein Zahraei2*
1 Assistant Professor in BGMU UFA, Russia.
2 Shiraz University of Medical Sciences Shiraz, Iran.
*Corresponding Author: Seyed Ali Hossein Zahraei
Shiraz University of Medical Sciences Shiraz, Iran.
Email: Eli.najafizade@yahoo.com
Received : Oct 22, 2021
Accepted : Dec 20, 2021
Published : Dec 27, 2021
Archived : www.jcimcr.org
Copyright : © Zahraei SAH (2021).
Abstract
Hair loss is a very serious problem that men and women struggle with almost every day. Given that many people suffer from this disease, scientists try to find optimal solutions every year. There are many products and methods that claim to restore hair growth or reduce hair loss. Some of these methods seem to be almost completely useless, while others make significant changes. Fortunately, there are many inventions within the field of hair restoration in recent years. Nowadays, many hair loss treatments are available, and therefore the hottest ones are Mesotherapy and PRP. Both these treatments have completely modified the planet of hair loss treatments.
Both methods are approved by the World Health Organization. PRP and mesotherapy are among the tried and tested methods of hair restoration which will make an enormous difference. It is not uncommon to mistake these two hair restoration methods, many people make this mistake. However, knowing the difference between PRP and mesotherapy will assist you opt which option is best for you. Because, although both try to repair hair, their performance is slightly different. Therefore, one of these methods may help you to get better results according to your individual characteristics.
Although these two treatments have proven effective, people are often confused about choosing one. Each therapy has its pros and cons, so we’ve administered a comparison of mesotherapy vs PRP for hair.
Citation: Najafizade E, Jamali AK, Zahraei SAH. Mesotherapy vs PRP: Hair Loss. J Clin Images Med Case Rep. 2021; 2(6): 1518.
Introduction
In the present world, hair loss is a problem faced by individuals of all age groups and ethnicity. Environmental, psychological, genetic, and pathological factors are associated with hair loss [1]. Hair follicles undergo periodic growth phases consisting of anagen (active phase), catagen (transitional phase) and telogen (rest phase), where hair growth takes place followed by shedding.
Traditional treatment strategies have largely been limited to pharmaceutical and surgical modalities. Currently approved medications, such as Finasteride and Minoxidil, require a great degree of compliance for long periods of time with varying degrees of effectiveness [2,3]. Side effects are also common and often dissuade use, including sexual dysfunction, mood disorders, increased risks of prostate/breast cancer, and birth defects [4,5].
Recent discoveries in the molecular pathways of the hair cycle, however, have provided the foundation for novel investigations in a “biologically oriented” approach to cell based therapies in hair restoration [6-9].
Platelet-rich plasma (PRP) is an autologous blood product containing high concentrations of platelets in a small volume of plasma. Platelets contain hundreds of bioactive molecules in their alpha granules, including growth factors, cytokines, and cell adhesion molecules [1]. The biological rationale for the use of PRP in regenerative medicine involves platelet degranulation, thus allowing for the local delivery of growth factors, modification of the inflammatory response, and stimulation of cell proliferation and differentiation within the target tissue [10-12].
“Mesotherapy” is a non-surgical, minimally invasive method of drug delivery that consists of multiple intradermal or subcutaneous injections of a mixture of compounds “melange” in minute doses [13]. Plant extracts, homeopathic agents, pharmaceuticals, vitamins and other bioactive substances can be used, but alcohol- or oil-based substances should not be used for mesotherapy because of the risk of cutaneous necrosis [14].
History of mesotherapy
Insertion of needle(s) for alleviation of diseases features an extended history, starting from Hippocrates (400 B.C.) who used a neighborhood application of cactus for shoulder pain, the Chinese using acupuncture (since 2000 years) then, injection of medicine following the invention of the hollow needle since nineteenth century
In 1952, Dr. Michel Pistor, a generalist practicing in rural France, administered 10 ml of procaine intravenously in an attempt to abort an acute asthmatic attack during a patient. While the treatment didn’t ameliorate the patient’s respiratory status, upon follow-up, the patient reported an enormous improvement in his impaired hearing. Soon thereafter, Dr. Pistor began experimenting with superficial injections of procaine around the ear of hearing-impaired patients and experienced some success. Soon his practice was crammed with hearing-impaired patients seeking treatment. His results were mixed, however. Many of these patients saw improvement in seemingly unrelated concomitant conditions, like eczema of the acoustic meatus and TMJ pain. Additionally, patients reported improvement with tinnitus, which can be related to hearing impairment [15].
Dr. Pistor continued experimenting with superficial injections of procaine for the treatment of a spectrum of disorders, and on June 4th of 1958, he published a bit of writing describing his clinical success with this novel procedure during which he stated, “the action on the tissues originating from the mesoderm is so extensive that these treatments should be called mesotherapy” (author’s translation). This was the first time the term “mesotherapy” appeared in print. Dr. Pistor described mesotherapy as, “smallest dose, infrequently, within the right location [16].
The primary international conference on mesotherapy happened in 1976, which was also the year when mesotherapy was first utilized in in-patient settings in France. In 1981, Dr. Jacques LeCoz introduced mesotherapy into the orthopedic clinic at the Institute Nationale du Sports (National Institute of Sports) in Paris. In 1987, the French Academy of drugs officially recognized mesotherapy as a legitimate treatment modality within conventional medicine [17].
What is mesotherapy?
Mesotherapy is a wide range of minimally invasive injections that have many uses [18]. In mesotherapy, a mixture of various tonics is injected into the skin. These include plant extracts, various medications, vitamins, enzymes, hormones, growth factors and other factors which will help treat alopecia (hair loss or baldness) [19]. The original meaning of the word “Meso” goes back to the embryonic stem cells. When the human body is not yet formed and the embryo is several thousand cells, three layers of stem cells are formed. Ectoderm, mesoderm and endoderm are the names of these three layers, each of which later forms different parts of the body. In the case of the skin, for example, ectodermal stem cells form the outer surface of the skin (the first 5 layers of the skin) or the epidermis, and mesodermal stem cells form the dermis or middle layer of the skin. So in mesotherapy, our goal is to inject the drug into the middle layer of the skin, or dermis, which is made up of mesoderm stem cells [14,20].
Our skin is formed from three general layers [21]: Epidermis, Dermis (middle layer), Hypodermis (under the skin).
The part of the skin we see is the epidermis, which is less than a millimeter thick and whose surface is covered with dead cells. Epidermal cells are constantly dividing and replacing dead cells that gradually shed from our skin. The dermis, or middle layer, is made up of connective tissue that contains collagen fibers, elastin fibers, and hyaluronic acid. This section makes the skin flexible and contains a variety of important skin structures [22,23]. One of these structures is the hair follicle. The follicle contains 20 different types of cells that contain one or more hair follicles. Hair follicles are also the place where our hair strands are made, grow and come out of the skin. The part of the hair that we see is named the hair shaft. In hair mesotherapy, special drugs and compounds are injected into the dermis layer using a syringe. Because the hair follicles are in this layer, and if we want to get the restorative compounds to it effectively, we have to send them directly to this layer. Topical medications such as minoxidil usually do not cross the epidermis well; Therefore, they have less effect than hair mesotherapy
Effects and benefits of hair mesotherapy
Mesotherapy affects various skin layers like the epidermis, middle layer and animal tissue, also because the cardiovascular system, system and neurosensory system of the injection site. For many types of alopecia, hair mesotherapy has been shown to be effective and proven. The effects of mesotherapy can be summarized in the following list [24,25]:
Improve blood flow in the formula of many mesotherapy solutions; there are vasodilators that increase tissue circulation.
Nourishing skin tissue and follicles a variety of vitamins, amino acids and minerals are used in combination with
mesotherapy injections.
Prevents the method of degeneration of follicles increases strength and improves hair condition.
Regulation of sebum secretion. Drugs utilized in mesotherapy control the secretion of sebaceous glands.
Improve the effects of other treatments Mesotherapy
can be used as a complementary treatment for hair loss, steroid
injections, general hair care and more.
Useful for improving hair transplant results Mesotherapy can be used in three ways:
Before implantation: To improve blood flow to the scalp.
Immediately after implantation: To treat edema or swelling.
Sometime after implantation: For better healing of
wounds as well as strengthening of grafts.
History of PRP
Platelet-rich plasma, also referred to as PRP, contains a high concentration of growth factors and platelets. PRP that’s utilized in regenerative medicine is autologous, meaning that it’s generated from a person’s own body [26]. About six percent of normal blood is formed from platelets. On the opposite hand, PRP contains about 94 percent platelets. These platelets are important within the body because they’re ready to improve and speed up the healing processes within the body. This is possible due to the various proteins, system cells, and other bioactive factors that PRP consists of.
Platelet-Rich Plasma (PRP) is additionally referred to as plateletrich Growth Factors (GFs), Platelet-Rich Fibrin (PRF) matrix, PRF, and platelet concentrate. The concept and outline of PRP started within the field of hematology [27]. Hematologists created the term PRP within the 1970s so on explain the plasma with a platelet count above that of peripheral blood, which was initially used as a transfusion product to treat patients with thrombocytopenia [28]. Ten years later, PRP began to be utilized in maxillofacial surgery as PRF. Fibrin had the potential for adherence and homeostatic properties, and PRP with its anti-inflammatory characteristics stimulated cell proliferation [29]. Subsequently, PRP has been used predominantly within the musculoskeletal field in sports injuries. With its use in professional sportspersons, it’s attracted widespread attention within the media and has been extensively utilized in this field [30]. Other medical fields that also use PRP are cardiac surgery, pediatric surgery, gynecology, urology, face lift, and ophthalmology [31].
More recently, the interest within the appliance of PRP in dermatology; i.e., in tissue regeneration, wound healing, scar revision, skin rejuvenating effects, and alopecia, has increased. [26,31-34].
Wounds have a proinflammatory biochemical environment that impairs healing in chronic ulcers. In addition, it’s characterized by a high protease activity, which decreases the effective GF concentration. PRP is employed as a stimulating alternative treatment for recalcitrant wounds because it’s a source of GFs and consequently has mitogenic, angiogenic, and chemotactic properties [26].
In cosmetic dermatology, a study performed in vitro demonstrated that PRP can stimulate human dermal fibroblast proliferation and increase type I collagen synthesis [33]. Additionally, supported histological evidence, PRP injected in human deep dermis and immediate subdermis induces soft-tissue augmentation, activation of fibroblasts, and new collagen deposition, also as new blood vessels and adipose tissue formation [26,34].
Another application of PRP is that the development of burn scars, postsurgical scars, and acne scars [35]. According to the few articles available, PRP alone or together with other techniques seems to enhance the standard of the skin and results in a rise in collagen and elastic fibers.
In 2006, PRP has begun to be considered a possible therapeutic tool for promoting hair growth and has been postulated as a replacement therapy for alopecia, in both androgenetic alopecia and alopecia areata. Several studies are published that ask the positive effect PRP has on androgenetic alopecia, although a recent meta-analysis suggested the shortage of randomized controlled trials [36]. As stated by the authors, controlled clinical trials are considered the simplest thanks to provide scientific evidence for a treatment and avoid potential bias when assessing efficacy [37].
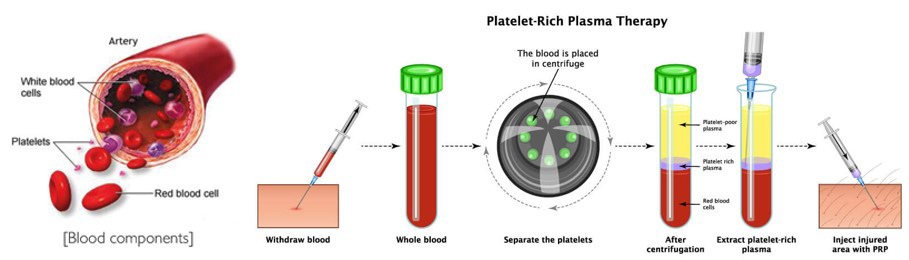
The procedure is performed by drawing blood from a patient and placing it during a centrifuge to separate the platelets from the remainder of the blood components. The concentrated platelets are then directly injected into the injured area of tissue. The “rich” platelets contain multiple growth factors which will stimulate new tissue growth. Some of these factors include transforming protein, animal tissue protein, epidermal protein, and vascular protein. These growth factors recruit new cells to the world to stimulate healing [38,39].
Platelet-rich plasma mechanism
The main mechanism of action of PRP isthru platelets, which besides their well-known function in homeostasis, release also factors that promotes angiogenesis and tissue repair [41]. Secretory granules of the platelets contain growth factors, coagulation factors, cytokines, adhesion molecules and integrins. Releasing of the active growth factors is realized through degranulation of alfa granules [42]. The main protein s included in alfa granules are: Platelet Derived Growth Factor (PDGF), Platelet Derived Endothelial Protein (PDEGF), Vascular Endothelial Protein (VEGF), Epidermal Protein (EGF), Insulin Like Protein (IGF), Platelet Derived Angiogenesis Factor (PDAF), Transforming Growth Factor (TGF), Keratinocyte Protein (KGF), Fibroblast Growth Factor (FGF), Connective Tissue Protein (CTGF), Tumor Necrosis Factor Alpha (TNF-α), Interleukin 1 β (IL 1 β) and Interleukin 8 (IL8) [41]. All these growth factors stimulate healing by attracting cells within the new formed matrix and triggering division of the cells.
Leukocytes provide high concentrations of matrix metalloproteinases, which are involved in angiogenesis, extracellular membrane remodeling, and hair cycling [43]; however, they may also have detrimental effects that can damage follicles and lead to scarring. Erythrocytes are a possible source of reactive oxygen species and should contribute to a burning sensation [43]. While there are no studies investigating the ideal therapeutic concentration of platelets, some suggest that maximum results occur when the platelet concentration is 2-5-fold higher than that of normal whole blood.
Growth factors in PRP promote hair regrowth by binding to their respective receptors expressed by stem cells of the follicle bulge region and associated tissues. Upon ligand binding, stem cells induce the proliferative phase of the follicle, producing the anagen follicular unit and facilitating hair regrowth [38,44]. Further, they activate downstream cascades leading to angiogenesis and stimulation and generation of adnexal structures. Anagen-associated angiogenesis has been linked to the secretion of VEGF by keratinocytes within the outer root sheath and fibroblasts of the dermal papilla. This increased production of VEGF promotes the expansion of normal and pathological dermal structures [45]. Activated autologous PRP has also been noted to activate the proliferation of dermal papilla cells by upregulating fibroblast growth factor-7 and β-catenin, additionally to extracellular signal-related kinase and Akt signaling [45].
Side effects of PRP
As the PRP solution consists of a person’s own blood
components, there are few risks of a reaction to the answer itself. This reduces the risks for an allergy which can occur from
injecting other medications, like cortisone or mucopolysaccharide. However, people undergoing PRP treatments for hair loss
may experience subsequent side effects [46,47]: mild pain at
the injection site
• Scalp tenderness
• Swelling
• A headache
• Itching
• Temporary bleeding at the injection site
Side effects of mesotherapy
Although this method is known as a low-risk method, but a small percentage of side effects are likely to be seen in this method [48,49]:
• Feeling of pain at the injection site.
• Itching at the injection site.
• See bruising.
• See redness.
• Allergic reactions to the anesthetic
For whom is the PRP method not recommended [50]?
• People with a history of drug use
• People with a history of alcohol use
• People taking blood thinners
• People who smoke on a daily and regular basis
For whom is hair mesotherapy not recommended [51]?
This method is not recommended for people with diseases such as diabetes, AIDS, hemophilia and hepatitis. It should be noted that this method cannot be useful for people with complete baldness.
Studies with positive results for PRP
A recent pilot study by Anitua et al. [52] evaluated the utilization of plasma rich in growth factors in 19 subjects with AGA. Subjects got 5 injections of PRP enhanced with platelet-rich protein (PRGF) activator to impress release of growth factors and morphogens from the specimen. Compared to baseline, all outcome measures showed positive results after 1 year of followup. Mean hair density, hair diameter, and terminal/vellus hair ratio were among the measures showing statistically significant improvement (p<0.05). Histomorphometric evaluation also favored the utilization of PRP, showing improvement in epidermal thickness, perifollicular neoangiogenesis, and terminal/miniaturized hair ratio, also as decreased perivascular inflammatory infiltrates. Overall, patients were satisfied with their clinical improvement.
In 2019, a team of researchers administered a scientific review of the research on PRP as a treatment for hair loss. Their findings appear within the journal Aesthetic cosmetic surgery [53].
The analysis ultimately focused on 11 research papers that included a whole of 262 participants with androgenetic alopecia. According to the authors, most of the studies found that injections of PRP reduced hair loss and increased the diameter of hairs and thus the density of hair growth.
They acknowledged, however, that the treatment is controversial, noting that tiny sample sizes and inferiority of research were among the limiting factors of their investigation.
Another 2019 systematic review, which featured in Dermatologic Surgery Trusted Source [54], examined the findings of 19 studies investigating PRP as a treatment for hair loss. These studies recruited 460 people in total. According to the authors of the review, most studies reported that PRP treatments led to hair regrowth in those with androgenetic alopecia and alopecia areata.
The authors of an extra review of clinical studies, which the International Journal of Women’s Dermatology Trusted Source [55] published, considered PRP to be a “promising” treatment for hair loss, based on their findings [55].
However, the team noted that because various researchers and clinics use different preparations, session intervals, and injection techniques to administer PRP, its effects can vary. At now, without a uniform protocol for injections, the authors explain, it's difficult to conclude that the treatment is effective.
Another randomized, blinded, half-head study performed by Gentile et al. [56] evaluated treatment outcomes of PRP in 20 male subjects. PRP was injected on half the affected scalp of each patient, while the other side received physiological solution as control. The study found a statistically significant increase altogether outcome measures, including mean hair count, hair density and terminal hair density, after 3 months of PRP treatment compared to placebo.
Gkini et al. [44] performed a prospective cohort study with 22 subjects, of which 20 completed the study. After 3 treatments, they reported increased hair density compared to baseline at 3, 6, and 12 months after PRP (p<0.001), as well as improvements in density and thickness. In this study, milder sorts of alopecia (Norwood Hamilton grade II–III) responded better to PRP treatment than more advanced cases. In addition, subjects with vellus hair had better results. Investigators also suggested that the PRP treatment seemed to cause increases in hair diameter quite hair count.
Khatu et al. [57] also led alittle prospective cohort study to research PRP efficacy in 11 subjects. After 4 sessions of PRP, 9 subjects reverted to having a negative hair pull test. Hair volume, coverage and follicular hair unit count were improved. Hair counts were noted to be increased from 71 to 93.09 on the standard. Significant reduction in hair loss was evident per patient questionnaires. Both Gkini et al. [27] and Khatu et al. [57] assessed patient satisfaction, and located the reported means of seven .1 and 7.0 out of 10, respectively
Cervelli et al. [45] performed a very similar study thereto of Gentile’s group with 10 men, and located similar positive results after 3 months of PRP. All outcome measures showed statistically significant improvement. In both studies, microscopic analysis revealed that epidermal thickness and density of follicles were both increased compared to base-line (p<0.05) 2 weeks after completion of PRP therapy. Immunohistochemistry was performed, and thus the share of Ki67+ cells was also increased in both basal keratinocytes of the epidermis and follicle bulge cells at 2 weeks after PRP treatment (p < 0.05 compared with baseline), suggesting an increase in keratinocyte proliferation. Investigators also observed an increase in small blood vessel count around the hair follicles (p<0.05) at 2 weeks after PRP compared with baseline), supporting the notion that PRP promotes angiogenesis via the discharge of vascular growth factors.
Swapna S Khatu et al. [57] performed a prospective study, safety, efficacy and feasibility of PRP injections in treating androgenic alopecia. Eleven patients affected by hair loss thanks to androgenic alopecia and not responding to six months treatment with minoxidil and finasteride were included during this study. The hair pull test was performed before every treatment session. A total volume of 2-3 cc PRP was injected within the scalp by using an insulin syringe. The treatment was repeated every fortnight, for a complete of 4 times. The outcome was assessed after 3 months by clinical examination, macroscopic photos, hair pull test and patient's overall satisfaction.
A significant reduction in hair loss was observed between first and fourth injection. Hair count increased from average number of 71 hair follicular units to 93 hair follicular units. Therefore, average mean gain is 22.09 follicular units per cm2 . After the fourth session, the pull test was negative in 9 patients.
They concluded that PRP injection could also be an easy, cost effective and feasible treatment option for androgenic alopecia, with high overall patient satisfaction.
Pietro Gentile et al. [56] reported the results of a randomized, evaluator-blinded, placebo-controlled, half-head group study to match, with the assistance of computerized trichograms, hair regrowth with PRP versus placebo. The safety and clinical efficacy of autologous PRP injections for pattern hair loss were investigated. PRP, prepared from alittle volume of blood, was injected on half the chosen patients' scalps with pattern hair loss. The other half was treated with placebo. Three treatments were administered to every patient at 30-day intervals. The endpoints were hair regrowth, hair dystrophy as measured by dermoscopy, burning or itching sensation, and cell proliferation as measured by Ki67 evaluation. Patients were followed for 2 years. Of the 23 patients enrolled, 3 were excluded. At the highest of the three treatment cycles, the patients presented clinical improvement within the mean number of hairs, with a mean increase of 33.6 hairs within the target area, and a mean increase in total hair density of 45.9 hairs per cm2 compared with baseline values. No side effects were noted during treatment. Microscopic evaluation showed the increase of epidermis thickness and of the number of hair follicles 2 weeks after the last PRP treatment compared with baseline value (p< .05). They also observed an increase of Ki67+ keratinocytes within the epidermis and of hair follicular bulge cells, and alittle increase of small blood vessels around hair follicles in the treated skin compared with baseline (p< .05). Relapse of androgenic alopecia wasn't evaluated altogether patients until 12 months after the last treatment. After 12 months, 4 patients reported progressive hair loss; this was more evident 16 months after the last treatment. Those four patients were re-treated. Their data clearly highlight the positive effects of PRP injections on male pattern hair loss and absence of major side effects. PRP may function a secure and effective treatment option against hair loss.
JS Kang et al [58] investigated the clinical efficacy of interfollicular injection of CD34+ cell-containing PRP preparation for pattern hair loss. CD34+ cell-containing PRP preparation was injected on the scalps of 13 patients with pattern hair loss, and 13 patients were treated with interfollicular placental extract injection as an impression. The numbers of platelets in PRP were microscopically counted and CD34+ cells were evaluated with flow cytometry. Three months after the primary treatment, the patients presented clinical improvement within the mean number of hairs, 20.5 ± 17.0% (P<0.0001), mean hair thickness, 31.3 ± 30.1% (P<0.0001), and mean two-point score, 84.4 ± 51.7% (P<0.0001) compared with baseline values. At 6 months, the patients presented clinical improvement in mean hair count, 29.2 ± 17.8% (P <0.0001), mean hair thickness, 46.4 ± 37.5% (P<0.0001), and mean two-point score, 121.3 ± 66.8% (P<0.0001) compared with baseline. The MIXED procedure revealed that CD34+ cell-containing PRP treatment presented a higher degree of improvement than placental extract treatment in hair thickness (P=0.027) and overall clinical improvement (P=0.023).
Their data suggest that the interfollicular injection of autologous CD34+ cell-containing PRP preparation features a positive therapeutic effect on male and feminine pattern hair loss without remarkable major side-effects.
Abeer Attia et al. [59] evaluated the efficacy and safety of autologous platelet-rich plasma within the treatment of female pattern hair loss.
Thirty female patients with female pattern hair loss were randomly assigned to receive autologous PRP injection into a specific area, and another area was injected with normal saline as a placebo. Sessions were performed weekly for a maximum total of 4 sessions. Patients were followed up 6 months after the top of last session. The outcome was assessed both subjectively and objectively.
There was a statistical significant difference between PRP and placebo areas regarding both hair density and hair thickness as measured by a folliscope. The hair pull test became negative in PRP-injected areas in 25 patients (83%) with average number of three hairs. Global pictures showed a big improvement in hair volume and quality along side a high overall patient satisfaction in PRP-injected sites, and these results were maintained during the 6-month follow- up. Platelet-rich plasma injections are often considered an alternate for the treatment of female pattern hair loss with minimal morbidity and a coffee cost-to-benefit ratio.
This was a case control study performed by Elham Behrangi et al. [60] on 120 patients with grade II and III androgenetic alopecia, supported the Hamilton-Norwood arrangement and grade 1 to five female pattern alopecia. Participants were randomly divided into two groups of 30 patients. The primary group was treated with Finastride and thus the second underwent PRP treatment; with a gaggle of 60 participants who received placebo. Follow-up was performed using macroscopic photography and Pull test bottom line, monthly for 3 months, and eventually three months after PRP-treatment for patients who received PRP.
A complete number of 120 participants enrolled during this study; 28 patients within the PRP group, 26 patients within the Finastride group and 60 patients within the control group. The results of 1 month follow up showed that hair growth and hair loss reduction weren't significantly improved within the PRP group compared to the Finastride and control groups (P ≥ 0.05). Significant differences were observed in results of progression of hair growth and reduction of hair loss between groups after three and 6 months (P value: 00.).
PRP-treatment could also be a completely unique therapeutic approach. The effective results of PRP-treatment might be because of this eagerness. Furthermore, the time duration of this procedure is brief, which may be a crucial factor for the patients since most of the participants are young men who don't tend to undergo long-time treatments, like Finastride consumption and Minoxidil Application Alopecia (AA) could also be a standard autoimmune condition, causing inflammationinduced hair loss.
A. Trink et al. [61] evaluated the efficacy and safety of PRP for the treatment of AA during a randomized, double-blind, placebo- and active-controlled, half-head, parallel-group study.
Forty-five patients with AA were randomized to receive in tralesional injections of PRP, Triamcinolone Acetonide (TrA) or placebo on one half their scalp. The opposite half wasn't treated. Three treatments got for each patient, with intervals of 1 month. The endpoints were hair regrowth, hair dystrophy as measured by dermoscopy, burning or itching sensation, and cell proliferation as measured by Ki-67 evaluation. Patients were followed for 1 year.
PRP was found to increase hair regrowth significantly and to decrease hair dystrophy and burning or itching sensation compared with TrA or placebo. Ki-67 levels, which served as markers for cell proliferation, were significantly higher with PRP. No sideeffects were noted during treatment.
This pilot study, which is that the primary to research the results of PRP on AA, suggests that PRP may function a secure and effective treatment option in AA, and involves more extensive controlled studies with this method.
Dilip Kachhawa et al. [62] compared the efficacy of placebo versus PRP injections within the treatment of male AGA.
Fifty male patients with AGA (Grade III to VI) were enrolled within the study. PRP was prepared using the double-spin method and injected within the androgen-related areas of scalp on the left side. Normal saline was injected on the right side during an identical fashion. Treatment sessions were performed with an interval of 21 days, and 6 sittings were completed for every patient.
Hair loss reduced with evidence of latest hair growth. Digital image analysis showed an overall improvement in hair density and quality as lanugo-like hair became thicker, normal hair. An improvement in hair density, quality and thickness on trichoscopy was noted.
Their data suggest that PRP injections have therapeutic effect on male pattern hair loss with no major side effects and high patient satisfaction overall.
Qian Qu et al. [63] studied the ascertain and compare the efficacy and safety of PRP treatment for various grades of AGA in males and females over 6 months.
During this study, 51 MPHL patients with Norwood-Hamilton stage II-V and 42 FPHL patients with Ludwig stage I to III were enrolled for 6 monthly sessions of PRP injections. A longitudinal analysis was wont to compare the hair density, thickness, and hair pull test over 6 months for MPHL and FPHL through Generalized Estimating Equation (GEE) models. Phototrichograms of scalp inflammation and oil secretion, global photographs and overall patient satisfaction were also assessed.
Consequently, improvement of hair density, hair thickness, hair pull test, the extent of scalp inflammation and oil secretion were observed with statistical significance altogether stages for both MPHL and FPHL at 6 months. Noteworthy, lower level of alopecia (Grade II, III in MPHL and Grade I in FPHL) had better response to PRP, and also had a way better tendency of increment of hair growth than that of high-grade patients with prolonged treatment.
They concluded that PRP injections, as an efficacious and reliable therapy, are often recommended for Grade II and Grade III in MPHL and Grade I in FPHL.
Waleed et al. [64] evaluated safety and efficacy of (PRP) vsIntralesional Corticosteroid (ILCs) in treatment of alopecia (AA).
This study was conducted on 80 patients of both sexes who had AA. Patients were classified into Group I treated by ILCs and group II treated by (PRP). Results were assessed by dermoscopic evaluation and by hair Re-Growth Score (RGS) at (baseline), 2, 4, 6, 8, and 12 weeks. Patients were followed up for six months.
There was greater hair re-growth after treatment in both groups. In group I, (26) patients (65%) showed improvement & gt; 70% compared to 29 patients (72.5%) in group II. There was significant re-growth of pigmented hair and decrease in dystrophic hair (P<0.001) by dermoscopic evaluation in both groups. The difference between both groups was insignificant (P=0.57). At follow-up, two (5%) patients in group II had relapse compared to 10 (25%) patients in group I. Platelets rich plasma is safe and promising therapeutic option in AA.
Hausauer et al. [65] compared the efficacy, satisfaction, tolerability, and safety of two initial PRP injection protocols over 6 months.
Prospective, randomized, single-blinded trial among 40 patients with moderate AGA. Participants received subdermal PRP injections consistent with 1 of two treatment protocols: 3 monthly sessions with booster 3 months later (Group 1) or 2 sessions every 3 months (Group 2). Folliscope hair count and shaft caliber, global photography, and patient satisfaction questionnaires were obtained at baseline, 3 months, and 6 months.
At 6 months, both groups demonstrated statistically significant increases in hair count (p<.001). These improvements occurred more rapidly and more profoundly for Group 1 (mean percent change: Group 1, 29.6 ± 13.6 vs Group 2, 7.2 ± 10.4; p<.001). Shaft caliber also increased significantly with no difference between groups. Treatments produced high satisfaction (82% “satisfied” or “highly satisfied”) and were safe and well tolerated (mean pain score 2.1).
Subdermal PRP injections are an efficacious and tolerable therapy among men and ladies with AGA [66]. The benefits could also be greater if first administered monthly. Clinicians should consider these findings when designing treatment plans.
Hieronymus et al. [66] evaluated injections of Stromal Vascular Fraction (SVF), which is rich in Adipose-Derived Stromal Cells (ASCs) in combination with Platelet-Rich Plasma (PRP) in the upper scalp as a new autologous treatment option for AGA.
Ten male patients (age range, 25-72 years), affected by AGA at stage II to III consistent with the Norwood-Hamilton scale, are treated with one injection of autologous PRS (ACPSVF: combination of PRP and SVF) within the upper scalp. Preinjection and 6 and 12 weeks postinjection changes in hair density were assessed using ultra high-resolution photography (Fotofinder).
Hair density was significantly increased after 6 weeks and 12 weeks postinjection (P=0.013 and P<0.001). In hair-to-hair matching analyses, new hair grew from active follicles. Furhtermore nonfunctioning hair follicles crammed with hyperkeartotic plugs, up to today assumed incapable of forming new hair, proved to grow new hair. No side effects were noted after treatment.
A single treatment of platelet-rich stroma injected within the scalp of patients with AGA significantly increased hair density within 6 to 12 weeks. Further research is required to work out the optimal treatment regimen.
Another May 2020 study showed that PRP injections had a positive effect on hair density in men with androgenic alopecia [67].
Studies with negative results for PRP
A recent pilot study by Puig et al. [68] evaluated the effect of PRP scalp injections in women with female androgenetic alopecia.
This double-blind, multicenter, placebo-controlled study compared the effect of PRP thereupon of saline placebo as scalp injection. The endpoints were hair count and Hair Mass Index (HMI), in conjunction with patient-opinion survey responses. Platelet-rich plasma was manufactured from patients' blood using the Angel PRP system.
Hair mass index or hair count didn't statistically significantly differ between the study and placebo groups. However, 13.3% of the treatment subjects (vs 0% of the placebo subjects) experienced substantial improvement in hair loss, rate of hair loss, hair thickness, and easy managing/styling hair, and 26.7% (vs 18.2% of the placebo group) reported that their hair felt coarser or heavier after the treatment.
Platelet-rich plasma didn't demonstrate any statistically significant improvement in HMI or hair count in women with congenital female pattern hair loss. The patient survey results suggest a therapeutic advantage of PRP as perceived by patients but not consistent with hair count or HMI.
Faezeh Khademi et al. [69] investigated the effect of PRP on hair regrowth in patients with alopecia (AA) totalis. Ten subjects (28.9 ± 6.28 years; five males and five females) with clinically diagnosed AA totalis for a minimum of three years who had not received any treatment within 3 months before the study were recruited. Blood sample was collected in thrombocyte harvesting tubes. The PRP was separated via centrifugation. The patients' scalp was divided sagittally into two approximately equal parts. In each patient, 4 mL of PRP was injected intradermally into the left or right side of the scalp; in each point, 0.1 mL of PRP was injected. Each patient was followed up monthly for 4 months. No hair regrowth was seen in eight patients and in two patients only .05). There was no side effect during treatment. Single dermal PRP injection didn't convince have any effect on hair regrowth in these patients.
6Studies with positive results for mesotherapy N. Moftah et al. [70] evaluated the efficacy and safety of mesotherapy using dutasteride-containing preparation in treatment of FPHL. This study included 126 female patients with FPHL. They were classified into two groups; group I (86 patients) injected with dutasteride-containing preparation and group II (40 control patients) injected with saline. Patients received 12 sessions and were evaluated at the 18th week by: photographic assessment, hair pull test, hair diameter and patient self-assessment. Ultrastructural evaluation was finished three patients.Results
After mesotherapy with dutasteride-containing preparation, photographic improvement occurred in 62.8% of patients compared with 17.5% on top of things group, mean number of epilated hairs was significantly decreased, mean hair diameter was significantly increased. Patient self-assessment showed statistically significant improvement compared with the controls. There was a indirect correlation between degree of improvement and duration of FPHL. Side effects were minimal with no statistically significant difference between the two groups. Ultrastructural examination of pretreated hairs revealed absent cuticle in one patient and focal destruction of the cuticle within the second patient, which reappeared in both after therapy [70].
They concluded that mesotherapy with dutasteride-containing preparation was effective, tolerable and minimally invasive treatment modality in FPHL with better response for shorter duration of the disease [70].
NahlaHunter et al. [71] compared the initial efficacy and safety of mesotherapy containing nutritional supplements to topical minoxidil 5% solution in FPHL. 30 patients with FPHL were randomly classified into two equal groups: A applied minoxidil 5% lotion twice daily; B was injected with mesotherapy once weekly. For both groups Ultrasound Biomicroscopy (UBM) was performed before and at the top of the 12th week of treatment. After treatment, no significant difference was found between both groups with reference to either improvement of hair density and hair loss (P=0.27 and 0.056, respectively), nor the degree of improvement of Ludwig’s classification as assessed by the investigator(P=0.210). A significant difference was observed between both groups (P=0.001) with the absolute best degree of satisfaction within the mesotherapy group. In group A, no significant difference was found within the amount of follicle s or the diameter of the foremost important follicle (P=0.244 and 0.925, respectively). In group B, an enormous difference was found within the amount of hair follicles (P=0.001), with no significant difference within the diameter of the foremost important follicle (P=0.105). The mesotherapy group showed more improvement with regard to the increase within the amount of the hair follicles after treatment (P=0.007). Limitation of the study is little sample size, and comparatively short duration of treatment [71].
Mesotherapy, containing nutritional supplements only, is an efficient , more acceptable to patients, and more tolerable modality compared with topical minoxidil within the treatment of FPHL [71].
Marwa et al. [72] evaluated the efficacy and safety of injection of 0.05% dutasteride containing solution as compared to 0.9% saline injected via nappage technique. Twenty-eight male patients with MPHL types III, IV and V completed the study. They were randomly assigned to 2 groups: group I the treated group (n=14) and group II controls (n=14). Patients received seven injections at weeks 0, 1, 2, 3, 5, 7 and 11 and were evaluated at week 12.
The dutasteride-containing preparation was significantly simpler than placebo. This was evident by three assessment methods; difference in hair count, professional independent observers’ assessment and patients’ self assessment. The less the duration of MPHL, the higher was the response to mesotherapy. Minimal side effects within the sort of mild pain and headache were detected.
Mesotherapy using dutasteridecontaining solution is an efficient method in treating moderate degrees of MPHL [72].
Sima Marzban et al. [73] examined the safety and efficacy of mesotherapy within the treatment of androgenetic alopecia.
Five studies including 344 patients were considered eligible for the review. Of five studies included during this review, three were Randomized Controlled Trials (RCTs) and two were nonRCTs. In previous studies, mesotherapy was performed using dutasteride, minoxidil, and finasteride. As the analysis revealed, quality of retrieved studies was poor. The results showed that mesotherapy results in the development of efficacy outcomes. However, in one study, mesotherapy wasn't shown to be effective regarding some outcomes. No significant adverse effects were reported for mesotherapy
Although the findings of previous studies suggest that mesotherapy could even be a secure and effective treatment for androgenetic alopecia, further research is required to verify this finding.
Studies with negative results for Mesotherapy
Bruna Duque et al. [74] reported two cases of patchy alopecia that developed after mesotherapy for the treatment of androgenetic alopecia. In the first patient, alopecia developed after injections of the heparinoid vasodilator mesoglycan; the 3-month follow-up examination revealed alittle residual area of cicatricial alopecia. The second patient developed reversible alopecia after multiple scalp injections of homeopathic agents. These cases underline the possible risks of mesotherapy as a therapeutic technique for hair loss.
Three cases of alopecia secondary to mesotherapy were reported [74,75]. The first patient developed alopecia after being injected with a cocktail that contained mesoglycan (Prisma), a heparinoid vasodilator. The 3-month follow up examination revealed alittle residual area of cicatricial alopecia [74]. The second patient developed reversible alopecia after undergoing multiple sessions of mesotherapy containing homeopathic agents of unknown constituents [74]. Another article reports complications of scalp mesotherapy during a lady who developed multifocal scalp abscesses with subcutaneous fat necrosis and scarring alopecia [25,75].
Prachi Chetankumar et al. [76] compared efficacy and safety between mesotherapy (intralesional injection) and 5% topical minoxidil solution in male Androgenic Alopecia (AGA) by dermoscopic evaluation.
In a randomized active controlled trial, we enrolled 49 clinically diagnosed males of AGA and randomly allotted them into two groups – mesotherapy (A) (25) and minoxidil (B) (24).
Grade II was the foremost common presentation in our study. All dermoscopic parameters like variation of hair shaft diameter, follicular units with single and multiple hairs, yellow dots, and perifollicular halo didn't show any significant difference between the groups in the least time points except at 1st month where the difference in variation of hair shaft diameter between the two group was 0.04. They observed a big increase within the variation of hair shaft diameter between pre- and post-treatment in mesotherapy group compared to minoxidil group. The rest of parameters didn't show any significant difference within the group in mesotherapy and minoxidil.
In their study, they observed a big increase within the variation of hair shaft diameter between pre- and post-treatment in A compared to B. Other dermoscopic, trichoscan, and subjective measurement tool did not show significant difference between two groups. Our observation suggests that there is no significant improvement of mesotherapy in male AGA over minoxidil [76].
Discussion
We must first consider the definition of these two words:
Mesotherapy is that the use of low doses of drugs with repeated injections within the desired area.
PRP is blood plasma with concentrated platelets and is derived from the patient's own blood.
In both methods, the target substance (drug or plateletcontaining plasma) is delivered by repeated injections. Because PRP injection is similar to mesotherapy, but the drug is taken from the body itself, it is also called biological mesotherapy.
Both methods are suitable for people with thinning hair and hair loss, not people with baldness or severe hair loss.
Usually in these two methods, there is no possibility of infection or skin disorders.
Both methods help the body's metabolism and blood circulation, especially mesotherapy.
In hair mesotherapy, the injection is often done in the middle skin area, but in the prp injection, it can be done in all layers of the skin.
Mesotherapy is cheaper than the PRP method and therefore more customers use it.
Each of these treatments is done in several sessions, but in the PRP method, each session lasts about an hour, while each session of mesotherapy is a maximum of half.
Both of these procedures should be performed by a specialist doctor and a professional and experienced team in hair transplant clinics.
There is not much pain and bleeding in these two methods.
PRP appears to possess a potent anti-inflammatory action, because it promotes the discharge of mediators like IL-1 Receptor Antagonist (IL-1ra), soluble TNF Receptors (sTNF-R) I, IL-4, IL-10, IL-13, and interferon γ [10,44,77].
Conclusion
Aim of this systematic review was to guage efficacy, safety, and therapeutic durability of Platelet Rich Plasma (PRP) and Mesotherapy in treating various kinds of hair loss. In most studies patients' conditions had improved by receiving PRP therapy; these cases experienced a rise in growth and thickness of hair. Simultaneous use of PRP and Minoxidil demonstrated absolutely the best rate of improvement and satisfaction. The main side effect was pain thanks to PRP injection, which disappeared after ending the treatment and only one article reported more serious side effects. Optimization of this method depends on dosage, number of sessions and their intervals, and injection techniques. According to the results, the use of PRP because of its relatively high efficiency, low and tolerable side effects, and low recurrence rate are often an honest method for the treatment of alopecia and hair loss.
Also, the utilization of mesotherapy thanks to its relatively high and tolerable efficiency and low recurrence rate are often an honest thanks to treat hair loss.
Side effects of PRP are less than mesotherapy.
PRP is also more natural, but mesotherapy responds better.
There are more articles that have examined the effect of PRP than mesotherapy.
In summary, most respondents were satisfied with PRP for the treatment of their FPHL, with a majority of satisfied patients reporting marked or exceptional results.
A number of recent publications support the role of PRP within the treatment of genetic hair loss. In addition, some authors showed that pretreatment of follicular units with PRP before transplantation resulted in improved hair growth and density.
Most of the studies reviewed showed effectiveness of PRP in increasing terminal hair density/diameter.
During a PRP treatment session, doctors inject cells that belong to your body, which means that the procedure is completely safe. There is no possibility for your body to refuse the injection or show allergic reactions. This is just one of the many things that makes PRP treatment safer than other hair restoration methods.
More research is needed to help standardize technology, methodologies, and techniques to offer the assurances of treatment consistency, maximum efficacy, and safety of PRP and mesotherapy for hair loss.
PRP has proven to be highly effective in helping hair follicles remain within the expansion phase resulting in thicker and longer hair. Moreover, it also promotes the expansion of latest hair and strengthening the prevailing hair follicles.
On the opposite hand, mesotherapy is employed to treat hair loss caused because of genetic reasons, stress, diet, or chronic illness. It aims at improving the absorbance of necessary ingredients and compounds through the scalp for correct hair health and growth. The results from mesotherapy are often maximized by a series of sessions over a course of a few of of months.
In general, both therapies are effective within the treatment of hair loss and fewer expensive than hair transplant surgery. They can be merged also for delivering even better results. While PRP for hair is used right after the hair transplant surgery to initiate the tactic of faster healing, mesotherapy for hair loss are often used to maintain the results of hair transplantation for an extended time.
As observed, PRP, also as Mesotherapy, are often equally effective, and be even more so when utilized in combination with each other. They both function safer, less costly, and fewer invasive alternatives to hair transplant surgery.
So in this article, we tried to answer the question of whether hair mesotherapy is better or PRP by examining the difference between mesotherapy and PRP. But as you can see, each of these methods has advantages and disadvantages that can be the best choice for a person depending on the situation and health status. Therefore, we recommend that you consult your doctor to choose the best method.
References
- Hunt, N. and S. McHale, The psychological impact of alopecia. Bmj. 2005; 331: p. 951-953.
- Shin, H.S., et al., Efficacy of 5% minoxidil versus combined 5% minoxidil and 0.01% tretinoin for male pattern hair loss. American journal of clinical dermatology. 2007; 8: p. 285-290.
- Mella, J.M., et al., Efficacy and safety of finasteride therapy for androgenetic alopecia: A systematic review. Archives of dermatology. 2010; 146: p. 1141-1150.
- Shenoy, N.K. and S.M. Prabhakar, Finasteride and male breast cancer: Does the MHRA report show a link? Journal of cutaneous and aesthetic surgery. 2010; 3: p. 102.
- Traish, A.M., A. Mulgaonkar, and N. Giordano, The dark side of 5α-reductase inhibitors’ therapy: Sexual dysfunction, high Gleason grade prostate cancer and depression. Korean journal of urology. 2014; 55: p. 367-379.
- Talavera-Adame, D., D. Newman, and N. Newman, Conventional and novel stem cell based therapies for androgenic alopecia. Stem cells and cloning: Advances and applications. 2017; 10: p. 11.
- Marshall, B.T., et al., Future horizons in hair restoration. Facial Plastic Surgery Clinics. 2013; 21: p. 521-528.
- Falto-Aizpurua, L., S. Choudhary, and A. Tosti, Emerging treatments in alopecia. Expert opinion on emerging drugs. 2014; 19: p. 545-556.
- Miteva, M. and A. Tosti, Treatment options for alopecia: An update, looking to the future. Expert opinion on pharmacotherapy. 2012; 13: p. 1271-1281.
- Gupta, A.K. and J. Carviel, A mechanistic model of platelet-rich plasma treatment for androgenetic alopecia. Dermatologic Surgery. 2016; 42: p. 1335-1339.
- Mautner, K., et al., A call for a standard classification system for future biologic research: The rationale for new PRP nomenclature. PM&R. 2015; 7: p. S53-S59
- Hamilton, J.B., Patterned loss of hair in man: types and incidence. Annals of the New York Academy of Sciences. 1951; 53: p. 708-728.
- Raghvendra, T.S., et al., Mesotherapy-A non surgical cosmetic medicine treatment: A review. Int J Pharm Sci Rev Res. 2010; 4: p. 45-7.
- Herreros, F.O.C., A.M.d. Moraes, and P.E.N.F. Velho, Mesotherapy: A bibliographical review. Anais brasileiros de dermatologia. 2011; 86: p. 96-101.
- Matarasso, A., T.M. Pfeifer, and P.S.E.F.D. Committee, Mesotherapy for body contouring. Plastic and reconstructive surgery. 2005; 115: p. 1420-1424.
- Mammucari, M., et al., What is mesotherapy? Recommendations from an international consensus. 2014.
- Mammucari, M., et al., Mesotherapy, definition, rationale and clinical role: A consensus report from the Italian Society of Mesotherapy. Eur Rev Med Pharmacol Sci. 2011; 15: p. 682-694.
- Rohrich, R.J., Mesotherapy: what is it? Does it work? 2005, LWW.
- Vedamurthy M., Mesotherapy. Indian Journal of Dermatology, Venereology, and Leprology. 2007; 73.
- Konda D, D.M. Thappa, Mesotherapy: What is new. Indian J Dermatol Venereol Leprol. 2013; 79: p. 127-34.
- Kober J, et al. Generation of a fibrin based three-layered skin substitute. BioMed research international, 2015.
- Lee, W., et al., Multi-layered culture of human skin fibroblasts and keratinocytes through three-dimensional freeform fabrication. Biomaterials. 2009; 30: p. 1587-1595.
- Matsusaki, M., et al., Development of full thickness human skin equivalents with blood and lymph like capillary networks by cell coating technology. Journal of Biomedical Materials Research Part A. 2015; 103: p. 3386-3396.
- Saceda, D. and C. Bernárdez, Mesotherapy: Dutasteride, Minoxidil, Vitamins, in Techniques in the Evaluation and Management of Hair Diseases. 2021; CRC Press. p. 116-128.
- Al Faresi, F. and H.I. Galadari, Mesotherapy: Myth and reality. Expert Review of Dermatology. 2011; 6: p. 157-162.
- Alves, R. and R. Grimalt, A review of platelet-rich plasma: History, biology, mechanism of action, and classification. Skin appendage disorders. 2018; 4(1): p. 18-24.
- Andia, I., Platelet-rich plasma biology. Clinical Indications and Treatment Protocols with Platelet-Rich Plasma in Dermatology. Barcelona, Ediciones. 2016; p. 3-15.
- Montero, E.C., M.F. Santos, and R.S. Fernández, Platelet-rich plasma: Applications in dermatology. Actas Dermo-Sifiliográficas (English Edition). 2015; 106: p. 104-111.
- Lynch, M. and S. Bashir, Applications of platelet-rich plasma in dermatology: A critical appraisal of the literature. Journal of Dermatological Treatment. 2016; 27: p. 285-289.
- Andia, I., et al., Current concepts and translational uses of platelet-rich plasma biotechnology. Biotechnology, 2015.
- Andia, I. and M. Abate, Platelet-rich plasma: Underlying biology and clinical correlates. Regenerative medicine. 2013; 8: p. 645- 658.
- Li, Z.J., et al., Autologous platelet-rich plasma: A potential therapeutic tool for promoting hair growth. Dermatologic Surgery. 2012; 38: p. 1040-1046.
- Kim, D.H., et al., Can platelet-rich plasma be used for skin rejuvenation? Evaluation of effects of platelet- rich plasma on human dermal fibroblast. Annals of dermatology. 2011; 23: p. 424-431.
- Sclafani, A.P. and S.A. McCormick, Induction of dermal collagenesis, angiogenesis, and adipogenesis in human skin by injection of platelet-rich fibrin matrix. Archives of facial plastic surgery. 2012; 14: p. 132-136.
- Pathak, L., et al., Platelet -rich plasma: A recent review. Journal on Recent Advances in Pain. 2020. 6: p. 21.
- Giordano, S., M. Romeo, and P. Lankinen, Platelet-rich plasma for androgenetic alopecia: Does it work? Evidence from meta analysis. Journal of cosmetic dermatology. 2017; 16: p. 374-381.
- Ayatollahi, A., et al., Platelet-rich plasma for treatment of nonscarring hair loss: Systematic review of literature. Journal of Dermatological Treatment. 2017; 28: p. 574-581.
- Akiyama, M., L.T. Smith, and K.A. Holbrook, Growth factor and growth factor receptor localization in the hair follicle bulge and associated tissue in human fetus. Journal of Investigative Dermatology. 1996; 106: p. 391-396.
- Myung, P.S., et al., Epithelial Wnt ligand secretion is required for adult hair follicle growth and regeneration. Journal of Investigative Dermatology. 2013; 133: p. 31-41.
- Ehrenfest, D.M.D., L. Rasmusson, and T. Albrektsson, Classification of platelet concentrates: From Pure Platelet-Rich Plasma (P-PRP) to Leucocyte-and Platelet-Rich Fibrin (L-PRF). Trends in biotechnology. 2009; 27: p. 158-167.
- Rozman, P. and Z. Bolta, Use of platelet growth factors in treating wounds and soft-tissue injuries. Acta Dermatovenerologica Alpina Panonica et Adriatica. 2007; 16: p. 156.
- Lacci, K.M. and A. Dardik, Platelet-rich plasma: Support for its use in wound healing. The Yale journal of biology and medicine. 2010; 83: p. 1.
- Hausauer, A.K. and S. Humphrey, The physician’s guide to platelet-rich plasma in dermatologic surgery part II: Clinical evidence. Dermatologic Surgery. 2020; 46: p. 447-456.
- Gkini, M.A., et al., Study of platelet-rich plasma injections in the treatment of androgenetic alopecia through an one-year period. Journal of cutaneous and aesthetic surgery. 2014; 7: p. 213.
- Cervelli, V., et al., The effect of Autologous Activated Platelet-Rich Plasma (AA-PRP) injection on pattern hair loss: clinical and histomorphometric evaluation. BioMed research international. 2014.
- Zargar Baboldashti, N., et al., Platelet-rich plasma protects tenocytes from adverse side effects of dexamethasone and ciprofloxacin. The American journal of sports medicine. 2011; 39: p. 1929-1935.
- Kon, E. and G. Filardo, PRP or not PRP? That is the question. 2011, Springer.
- Rosina, P., et al., Psoriasis and side effects of mesotherapy. International journal of dermatology. 2001; 40: p. 581-5783.
- Brandao, C., et al., Abdominal haematoma a mesotherapy complication. ACTA Dermatovenereologica-Stockholm. 2005; 85: p. 446.
- Gobbi, A., D. Lad, and G. Karnatzikos, The effects of repeated intra-articular PRP injections on clinical outcomes of early osteoarthritis of the knee. Knee Surgery, Sports Traumatology, Arthroscopy. 2015; 23: p. 2170-2177.
- Luthra, A., Complications of Mesotherapy. Complications in Cosmetic Dermatology: Crafting Cures. 2016; 143.
- Anitua, E., et al., The effect of plasma-rich in growth factors on pattern hair loss: A pilot study. Dermatologic Surgery. 2017; 43: p. 658-670.
- Mao, G., G. Zhang, and W. Fan, Platelet-rich plasma for treating androgenic alopecia: A systematic review. Aesthetic plastic surgery. 2019; 43: p. 1326-1336.
- Hesseler, M.J. and N. Shyam, Platelet-rich plasma and its utilities in alopecia: A systematic review. Dermatologic Surgery. 2020; 46: p. 93-102.
- Stevens, J. and S. Khetarpal, Platelet-rich plasma for androgenetic alopecia: a review of the literature and proposed treatment protocol. International journal of women’s dermatology. 2019; 5: p. 46-51.
- Gentile, P., et al., The effect of platelet-rich plasma in hair regrowth: A randomized placebo controlled trial. Stem cells translational medicine. 2015; 4: p. 1317-1323.
- Khatu, S.S., et al., Platelet-rich plasma in androgenic alopecia: Myth or an effective tool. Journal of cutaneous and aesthetic surgery. 2014; 7: p. 107.
- Kang, J.S., et al., The effect of CD34+ cell containing autologous platelet-rich plasma injection on pattern hair loss: A preliminary study. Journal of the European Academy of Dermatology and Venereology. 2014; 28: p. 72-79.
- Tawfik, A.A. and M.A.R. Osman, The effect of autologous activated platelet-rich plasma injection on female pattern hair loss: A randomized placebo controlled study. Journal of cosmetic dermatology. 2018; 17: p. 47-53.
- Behrangi, E., et al., Platelet-Rich Plasma (PRP) Effect on Androgenetic Alopecia and Female Pattern Hair Loss. Journal of Skin and Stem Cell. 2019; 6.
- Trink, A., et al., A randomized, double blind, placebo and active controlled, half head study to evaluate the effects of plateletrich plasma on alopecia areata. British Journal of Dermatology. 2013; 169: p. 690-694.
- Kachhawa, D., et al., A spilt head study of efficacy of placebo versus platelet-rich plasma injections in the treatment of androgenic alopecia. Journal of cutaneous and aesthetic surgery. 2017; 10: p. 86.
- Qu, Q., et al., Efficacy of platelet-rich plasma for treating androgenic alopecia of varying grades. Clinical drug investigation. 2019; 39: p. 865-872.
- Albalat, W. and H.M. Ebrahim, Evaluation of platelet-rich plasma vs intralesional steroid in treatment of alopecia areata. Journal of cosmetic dermatology. 2019; 18: p. 1456-1462.
- Hausauer, A.K. and D.H. Jones, Evaluating the efficacy of different platelet-rich plasma regimens for management of androgenetic alopecia: A single-center, blinded, randomized clinical trial. Dermatologic Surgery. 2018; 44: p. 1191-1200.
- Stevens, H.P., S. Donners, and J. de Bruijn, Introducing plateletrich stroma: Platelet-Rich Plasma (PRP) and Stromal Vascular Fraction (SVF) combined for the treatment of androgenetic alopecia. Aesthetic surgery journal. 2018; 38: p. 811-822.
- Roohaninasab, M., et al., Systematic review of platelet-rich plasma in treating alopecia: Focusing on efficacy, safety, and therapeutic durability. Dermatologic Therapy. 2021; 34: p. e14768.
- Puig, C.J., R. Reese, and M. Peters, Double-blind, placebo-controlled pilot study on the use of platelet-rich plasma in women with female androgenetic alopecia. Dermatologic Surgery. 2016; 42: p. 1243-1247.
- Khademi, F., et al., The effect of platelet rich plasma on hair regrowth in patients with alopecia areata totalis: A clinical pilot study. Dermatologic therapy. 2019; 32: p. e12989.
- Moftah, N., et al., Mesotherapy using dutasteride containing preparation in treatment of female pattern hair loss: Photographic, morphometric and ultrustructural evaluation. Journal of the European Academy of Dermatology and Venereology. 2013; 27: p. 686-693.
- Hunter, N., et al., Comparing the efficacy of mesotherapy to topical minoxidil in the treatment of female pattern hair loss using ultrasound biomicroscopy: A randomized controlled trial. Acta Dermatovenerologica Croatica. 2019; 27: p. 1-1.
- Abdallah, M., K. El-Zawahry, and H. Besar, Mesotherapy using dutasteride containing solution in male pattern hair loss: A controlled pilot study. J Pan Arab Leag Dermatol. 2009; 20: p. 137-45.
- Marzban, S., B. Amani, and A. Asgharzadeh, Safety and Efficacy of Mesotherapy in the Treatment of Androgenetic Alopecia: A Systematic Review. Health Technology Assessment in Action, 2017.
- Duque Estrada, B., et al., Alopecia secondary to mesotherapy. Journal of the American Academy of Dermatology. 2009; 61: p. 707-709.
- Kadry, R., et al., Multifocal scalp abscess with subcutaneous fat necrosis and scarring alopecia as a complication of scalp mesotherapy. Journal of drugs in dermatology: JDD. 2008; 7: p. 72-73.76. Gajjar, P.C., et al., Comparative study between mesotherapy and topical 5% minoxidil by dermoscopic evaluation for androgenic alopecia in male: A randomized controlled trial. International journal of trichology. 2019; 11: p. 58.
- Mercuri, S., L. Vollono, and G. Paolino, The usefulness of Platelet-Rich Plasma (PRP) for the treatment of vitiligo: State of the art and review. Drug Design, Development and Therapy. 2020; 14: p. 1749.