Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Imaging thumbprint of molecular subtypes of breast cancer
Swati Sharma, MD1; Savas Ozdemir, MD1; Civan Altunkaynak, MD2; Smita Sharma, MD1
1 Department of Radiology, University of Florida College of Medicine, Jacksonville, Florida, USA
2 Department of Pathology and Laboratory Medicine, University of Florida College of Medicine, Jacksonville, Florida, USA.
*Corresponding Author: Swati Sharma
Department of Radiology, University of Florida College
of Medicine, Jacksonville, Florida, USA
Email: swati.sharma@jax.ufl.edu
Received : Dec 01, 2021
Accepted : Jan 11, 2022
Published : Jan 18, 2022
Archived : www.jcimcr.org
Copyright : © Sharma S (2022).
Abstract
Breast cancers have traditionally been classified based on histology. However, with the development in imaging modalities and available treatment options, it is imperative to also focus on the molecular classes of breast cancer. Molecular profile and gene expression are important predictors of treatment response and overall prognosis. Radiologists should have a good understanding of molecular categories of breast cancer along with their imaging hallmarks.
Highlights: • To familiarize the readers with the principal biomarkers and molecular classes of breast cancer. • To highlight the characteristic imaging features of each molecular class of breast cancer. • To focus on the pivotal role of molecular classification in the dynamic model of breast cancer treatment.
Citation: Sharma S, Ozdemir S, Altunkaynak C, Sharma S. Imaging thumbprint of molecular subtypes of breast cancer. J Clin Images Med Case Rep. 2022; 3(1): 1580.
Introduction
Breast cancer is the most common cancer in women, with a lifetime risk of 12% (one in eight) for average-risk women and is the second most common cause of death from cancer in women [1]. This review article aims to familiarize the readers with the principal biomarkers and molecular classes of breast cancer. Through a series of case review, we aim to highlight how presence of some findings as well as the absence of some other findings on mammogram, ultrasound, magnetic resonance imaging (MRI) and positron emission tomography/computed tomography (PET/CT), can indicate these molecular subtypes. Finally, the article focuses on how molecular classification plays a pivotal role in the dynamic model of breast cancer treatment.
Discussion of molecular classification
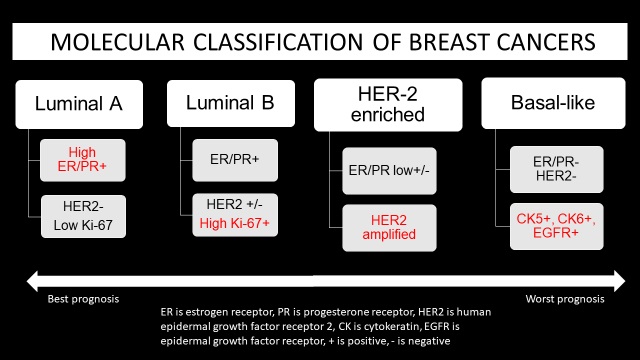
Breast cancers had traditionally been classified based on histology, but histologic classification offers limited prognostic value [2,3]. Molecular classification, which is based on biomarkers and gene expression, plays an important role in the approach to breast cancer treatment and its prognosis [4,5]. The three molecular classes of breast cancer are luminal cancers, HER2 enriched cancers and basal-like cancers. In 2000, Perou and colleagues were the first to propose a molecular classification of breast cancer. When tumor cells displayed features similar to the epithelial cells lining the lactiferous ducts, expressing, for example, cytokeratin 8/18 and genes associated with the estrogen receptor, the cancers were labeled luminal cancers. Alternatively, when cancer cells displayed characteristics similar to the myoepithelial cells (also known as basal cells) lining the inner surface of the basement membranes, expressing, for example, cytokeratin 5/6 and laminin, the cancers were labeled basal-like [6].
Testing for four molecular markers is standard for invasive breast cancer biopsy. For ductal carcinoma in situ (DCIS), biomarker status is provided after surgical excision. These markers include estrogen activated nuclear transcription factor (ER), progesterone nuclear transcription factor (PR), human epidermal growth factor receptor 2 (HER2) and Ki-67 which is the nuclear protein involved in cell proliferation. ER/PR are considered together for hormone receptor status.
Luminal cancers express ER and PR, as well as luminal cytokeratins (CK 7, CK 8, CK 18 and CK 19 among others) expressed in normal luminal epithelial cells [3,7]. Luminal A subtypes are HER2 negative and have low Ki-67 proliferative index. Luminal B subtypes can be HER2 positive in up to 30% and have higher Ki-67 proliferative index. Luminal B cancers have a higher histologic grade than Luminal A cancers [3]. A threshold of 20%, higher than the previous threshold of 14%, is used to determine high versus low proliferation of Ki-67 in breast cancers, based on the panel decision at the St Gallen International Breast Cancer Conference in 2013 [8-10].
HER2 enriched cancers are characterized by overexpression of HER2 gene and low expression of ER/PR [6]. Half of HER2- positive breast cancers are ER-positive, but they generally have lower ER levels, and many have p53 alterations [11,12]. HER2 cancers have a high histologic grade [3].
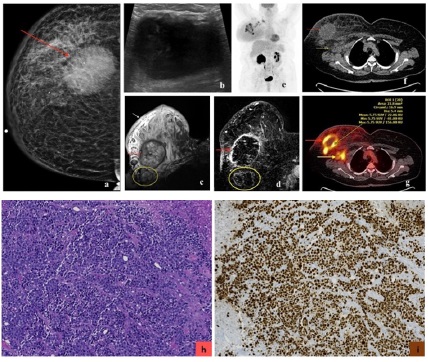
Basal-like cancers express basal cytokeratins (CK 5, CK 14 and CK 17 among others) and genes expressed in breast basal and/ or myoepithelial cells. Basal-like cancers have high proliferation index and higher association with genetic mutations [3,7] (Figures 1,2).
Imaging features of molecular classes
There are certain imaging characteristics that can be indicative of the individual molecular classes of breast cancer. These include, but are not limited to, shape, density, margins of a mass, presence or absence of calcifications on mammogram, shape, margins, orientation, posterior features of a mass on ultrasound and shape, margins, T2 signal, enhancement pattern, wash-out kinetics of a mass on MRI and morphology of a mass as well as pattern of 18F-fluorodeoxyglucose (FDG) uptake on PET/CT.
Molecular classes and role of nuclear medicine
Sentinel lymph node biopsy is considered the gold standard for axillary lymph node evaluation and preoperative lymphoscintigraphy yields a high identification rate for these sentinel lymph nodes [15]. 18F-FDG PET/CT can play an important role in breast cancer treatment due to its high rate of accuracy in detecting non-axillary lymphatic involvement and distant metastasis. The standardized uptake value (SUV), which is defined as the uptake level of 18F-FDG in cancer cells by invivo glucose hyper metabolism, might have predictive value for the diagnosis and prognosis of the disease. Additionally, 18F-FDG uptake in breast cancer has a close relation with histopathological and molecular classification. Higher SUVmax is associated with aggressive histopathological subtype, distant metastasis, and worse prognosis [16-19]. Advancement in molecular imaging has applications in breast cancer treatment. The uptake of more specific radiotracers targeting receptors helps in identifying molecular subtypes and in evaluating response to therapy such as estrogen receptor (ER) tracer 16 α-18F fluoroestradiol-17β (FES).
Imaging features of luminal cancers
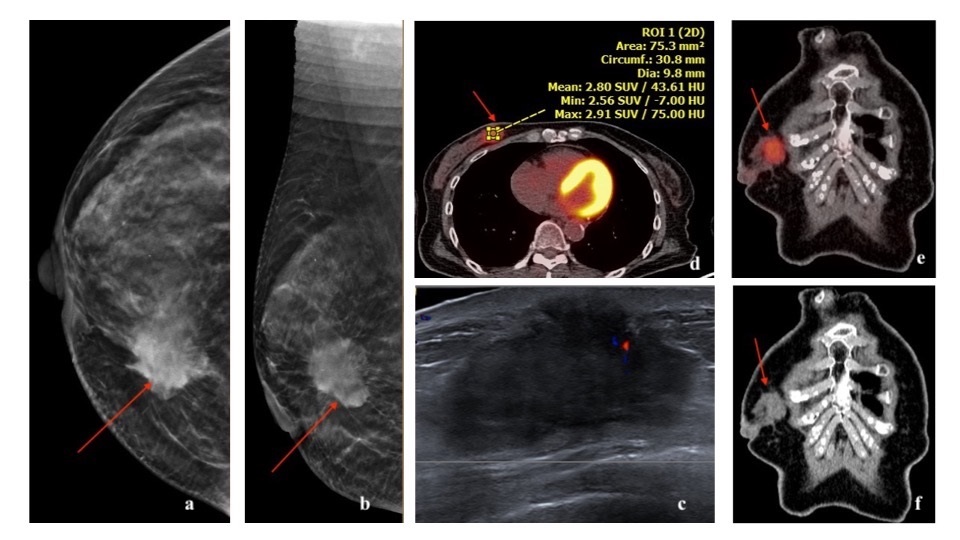
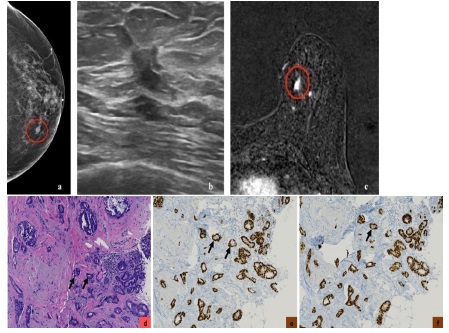
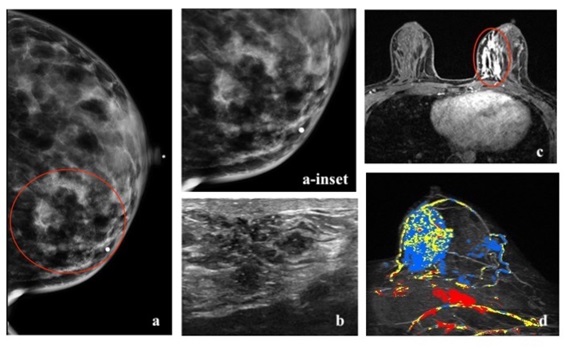
Luminal A subtype is the most common molecular type, constituting 50% of all invasive breast cancers [3]. It has the best overall prognosis, with a 5-year survival rate of 90% [20]. Luminal B subtype constitute 20% of all invasive breast cancers [3]. It has a 5-year survival rate of approximately 40% [20]. They can recur after a long disease-free interval. They are more likely to metastasize to bones [21]. Luminal cancer on mammogram presents most commonly as an irregular mass with spiculated margins, however it may present as a mass with calcifications or as microcalcifications (such as amorphous or coarse heterogeneous morphology) without a discrete mass [22]. On ultrasound, an irregular mass with non-circumscribed margins and posterior acoustic shadowing can be seen [22-24]. On MRI, luminal cancer can present commonly as irregular mass with spiculated margins. Mass is typically isointense to hypointense on T2-weighted imaging and enhances heterogeneously with plateau or wash-out kinetics on dynamic post contrast imaging [25]. With regards to 18F-FDG PET/CT, studies have shown association of higher SUVmax in tumors with high Ki-67 expression [16,17,26-28] (Figures 3,4,5).
18F-FES PET/CT can be used to quantify the entire volume of ER-positive disease for all the lesions in a patient. There can be heterogeneous 18F-FES uptake within the same tumor and between metastatic lesions. A higher level of 18F-FES activity in advanced tumors indicates a greater chance of response to endocrine therapy. 18F-FES PET/CT is, therefore, a great tool to guide treatment strategies and possibly to predict prognosis [29,30].
Imaging features of HER2 cancers
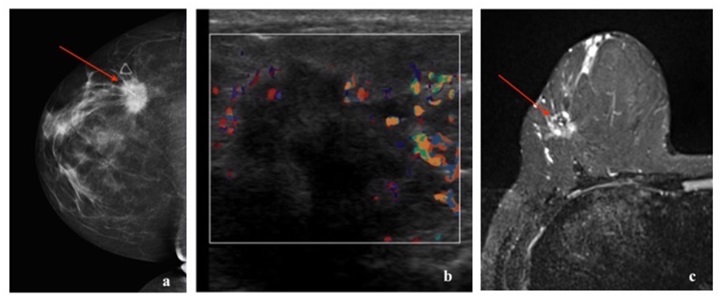
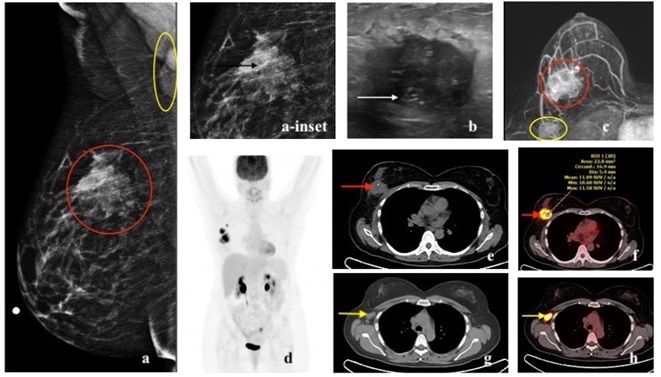
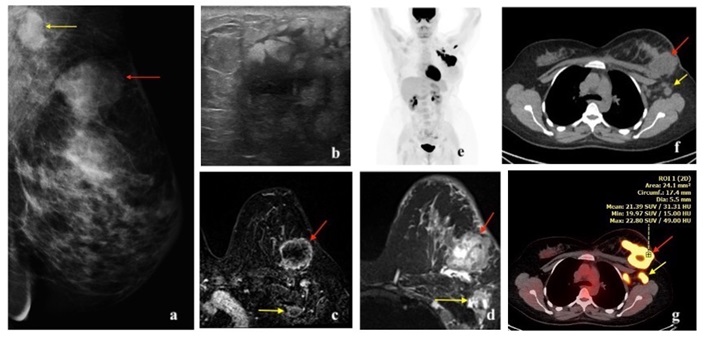
HER2 enriched cancers constitute 15% of invasive breast cancers [3]. The 5-year survival rate is approximately 40% [20]. HER2 amplified breast cancers have unique biological and clinical characteristics [12]. They are more often multifocal, multicentric and have nodal involvement at the time of presentation [3]. They are more likely to have visceral and brain metastases [21,31]. HER2 cancer on mammogram can present as a mass with microcalcifications and spiculated margins or as a group of calcifications (pleomorphic morphology being more common followed by amorphous and punctate) [22,23,32]. Calcifications are a characteristic feature of HER2 cancers, seen in over 75% of the cases [22,32]. On ultrasound, a hypoechoic irregular mass with non-circumscribed margins, internal echogenic foci or calcifications and mixed posterior features can be seen [22-24]. On MRI, HER2 cancer can present as a heterogeneously enhancing mass with spiculated or irregular margins and washout kinetics or less commonly as an area of non-mass enhancement [33]. With regards to 18F-FDG PET/CT, studies have shown association of higher SUVmax in HER2 positive tumors, more so with ER and PR negative status [16, 17, 26, 27, 28] (Figures 6,7,8).
Imaging features of triple-negative cancers
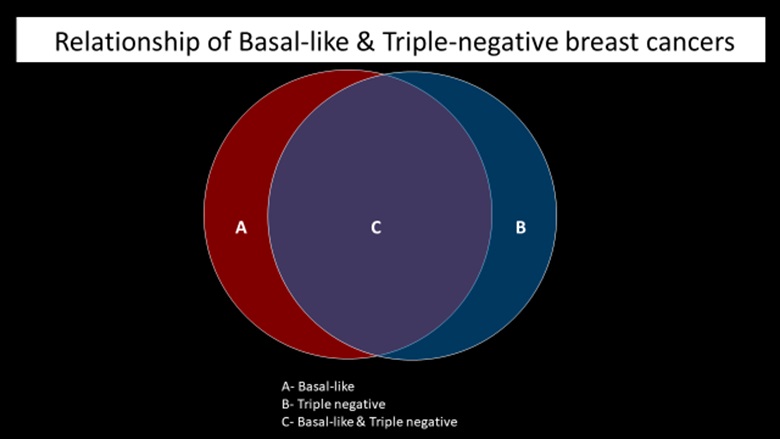
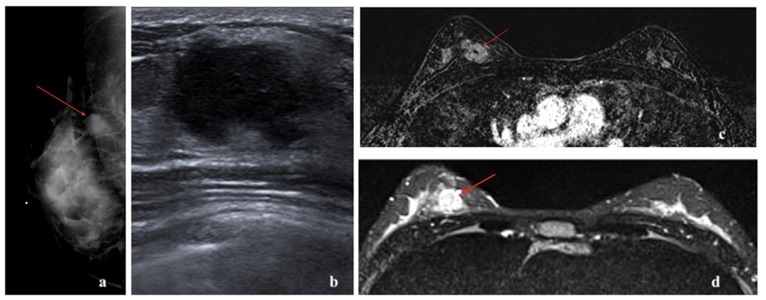
Triple-negative breast cancer (TNBC) refers to the tumors in which the cancer cells do not express estrogen or progesterone receptors and also do not express HER2 protein. TNBC cells test “negative” on all three biomarker tests. These cancers tend to be more common in women younger than age 40, in African American women, or women with BRCA1 mutation. Compared with the other types of breast cancer, triple-negative tumors are often larger at the time of diagnosis, involve lymph nodes more frequently, and are more often high-grade tumors. Approximately 70-80% triple negative breast cancers are basal-like [34]. Some rare histologic types of breast cancer, such as adenoid cystic and secretory carcinomas, are also characteristically triple-negative cancers; yet are associated with a better prognosis than the more common triple-negative infiltrating ductal carcinoma (Figure 9).
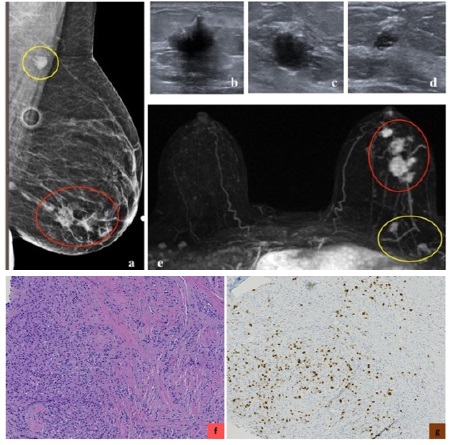
Basal-like cancers are more common in young African American women and can have an association with BRCA1 gene mutation. They constitute 15% of invasive breast cancers [3]. They can present as interval cancers. They can metastasize to lungs and brain and have the worst five-year survival rate among all molecular classes [20]. However, they do respond well to chemotherapy. The risk of recurrence is highest one to four years after diagnosis, with rare chances of recurrence thereafter. Basal-like cancer on mammogram can present as a high- density mass with indistinct margins or can be round or oval and mimic benign processes. Microcalcifications are uncommon. On ultrasound, basal-like cancer can present as either a complex cystic-solid mass with non-circumscribed margins or as a round or oval markedly hypoechoic to anechoic mass with circumscribed margins and posterior acoustic enhancement with fibroadenoma-like appearance [22-24,35]. On MRI, basal-like cancer can present as a round or oval T2 hyperintense mass with circumscribed or irregular margins. On dynamic contrast enhanced imaging, they can demonstrate rim enhancement or have enhancing internal septae with variable kinetics. Non-mass enhancement is typically not a feature [25]. Triple-negative cancers have been reported to show higher 18F-FDG uptake than luminal A and B types [16,36-38] (Figures 10,11,12).
In summary, each molecular class of breast cancer can have a characteristic appearance on imaging. However, it is important to know that in practice there may be heterogeneity in their imaging appearances and overlapping imaging features may be present among these molecular classes.
Molecular subtypes and breast cancer treatment perspectives
TNM (notation using tumor, lymph node and metastasis) staging is the standard breast cancer staging tool. The AJCC is one of the major bodies governing guidelines for cancer staging. The 8th and latest edition became effective in the United States as of January 1, 2018. This version incorporates the use of biomarkers in countries where they are widely available to designate prognostic staging [39].
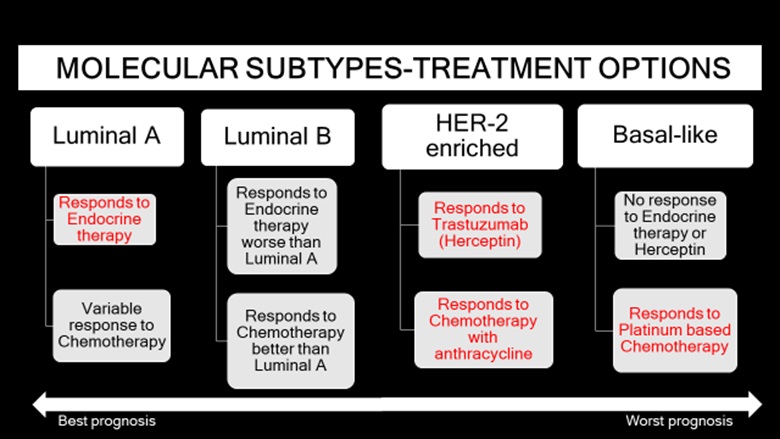
The presence of a specific biomarker in a breast cancer case is predictive of a positive response with targeted therapy. Hence, molecular classification plays a significant role in the dynamic model of breast cancer treatment by driving the choice of therapy. The decision for optimal breast cancer treatment depends on multiple factors including age of the patient, size of the tumor, extent of the tumor (whether the tumor is multifocal or multicentric or locally advanced), lymph node status, grade of the tumor and receptors for estrogen, progesterone and HER 2. The overall prognosis of invasive non-metastatic breast cancer depends on nodal status, histologic grade, tumor size [4]. Across all molecular classes, stage at diagnosis is the strongest predictor of survival. Between the molecular classes, luminal A has the best survival rates, followed by luminal B, HER2-enriched and basal-like in that order, from best to worst prognosis [20,40].
Treatment modalities include surgery, adjuvant or neoadjuvant chemotherapy, endocrine therapy (selective estrogen receptor modulators like tamoxifen and aromatase inhibitors like anastrozole and letrozole) and radiation therapy. Surgical treatment choices include lumpectomy or mastectomy, which can be simple or radical or modified radical. Luminal cancers respond well to endocrine therapy, with better response in luminal A subtype than luminal B subtype. Luminal cancers respond variably to taxane-based chemotherapy, with better response in luminal B subtype than luminal A subtype. Cyclindependent kinase-4/6 (CDK-4/6) inhibitors, example palbociclib, ribociclib and abemaciclib, are being used to treat metastatic HER2 negative luminal cancers. HER2 cancers respond to chemotherapy with anthracycline and have relative resistance to hormonal agents. HER2-amplified tumors have an increased sensitivity to doxorubicin possibly due to co-amplification of the topoisomerase-2 gene which is near the HER2 locus on chromosome 17 and is the target of the drug [12,41]. Targeted chemotherapy with HER-2 targeted antibody drug, trastuzumab, also known as herceptin, has improved prognosis. Since the introduction of trastuzumab therapy, there has been a 52% reduction in disease recurrence and a 33% reduction in the death rate [42]. Basal-like cancers are sensitive to platinumbased chemotherapy (example carboplatin). Poly-adenosine diphosphate ribose polymerase (PARP) inhibitors are only used to treat metastatic breast cancer in patients with BRCA1/2 mutation, as the tumor cells are sensitive to DNA damage involving the PARP enzyme [3,13,14]. Immunotherapy drugs can be used for treatment of metastatic triple negative breast cancers that express programmed cell death protein 1 (PD-L1) (Figure 13).
Tumor multigene next-generation sequencing (NGS) which allows fast and affordable sequencing of a high number of nucleotides, can be used to test for genomic mutations in metastatic breast cancers, like BRCA1/2, PIK3CA, AKT1, PTEN, ERBB2, ESR1 and NF1 mutations [43]. NGS is a powerful tool for selection of effective medications and treatment strategies for patients in the era of personalized medicine.
Conclusion
Molecular stratification of breast cancer is a crucial parameter in identifying appropriate treatment plan and for risk scoring. As key contributors in the care of breast cancer patients, it is important for radiologists to be familiar with molecular subtypes of breast cancer and to recognize their specific imaging features when present. Some of the individual decisive features are shape, density, margins of a mass and presence or absence of calcifications on mammogram, shape, margins, orientation and posterior features on ultrasound, shape, margins, T2 signal, enhancement pattern and wash-out kinetics of a mass on MRI and SUVmax on 18F-FDG PET/CT.
It is the combined morphologic picture on imaging that can stand out as a thumbprint for each molecular class in certain patients. However, there may be considerable heterogeneity and overlap in their imaging appearances and further research is needed to validate specific imaging biomarkers. Nevertheless, by understanding the effect of all prognostic factors for breast cancer, specifically the molecular classes, radiologists along with a multidisciplinary team, can achieve individualized patient care and improve patient outcomes
Glossary of abbreviations
CC: Craniocaudal; MLO: Mediolateral oblique; MRI: Magnetic Resonance Imaging; DCE: Dynamic Contrast Enhanced; STIR: Short Tau Inversion Recovery; MIP: Maximum Intensity Projection; PET: Positron Emission Tomography; CT: Computed Tomography; FDG: Fluorodeoxyglucose; SUVmax: maximum Standardized Uptake Value; ER: Estrogen Receptor; PR-Progesterone Receptor; HER2: Human Epidermal Growth Factor Receptor 2; H&E: hematoxylin and eosin
References
- American Cancer Society. Breast cancer facts & figures 2017- 2018. Atlanta: American Cancer Society. Inc. 2017.
- Trop I, LeBlanc SM, David J et al. Molecular Classification of Infiltrating Breast Cancer: Toward Personalized Therapy. Radiographics. 2014; 34(5): 1178-1195.
- Eliyatkın N, Yalcın E, Zengel B, Aktas S, Vardar E. Molecular Classification of Breast Carcinoma: From Traditional, OldFashioned Way to A New Age, and A New Way. J Breast Health 2015; 11(2): 59-66.
- Tirada N, Aujero M, Khorjekar G et al. Breast Cancer Tissue Markers, Genomic Profiling, and Other Prognostic Factors: A Primer for Radiologists. Radiographics. 2018; 38: 1902-1920.
- Munoz M, Fernandez-Acenero MJ, Martin S, Schneider J. Prognostic significance of molecular classification of breast invasive ductal carcinoma. Arch Gynecol Obstet. 2009; 280(1): 43-48.
- Perou CM, Sorlie T, Eisen MB et al. Molecular portraits of human breast tumors. Nature 2000; 406(6797): 747-752.
- Anders CK, Carey LA. Understanding and treating triple-negative breast cancer. Oncology (Williston Park). 2008; 22(11): 1233- 1239.
- Goldhirsch A, Wood WC, Coates AS, Gelber RD, Thurlimann B, Senn HJ. Strategies for subtypes—dealing with the diversity of breast cancer: highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann Oncol. 2011; 22(8): 1736-1747.
- Goldhirsch A, Winer EP, Coates AS, et al; Panel members. Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol. 2013; 24(9): 2206-2223.
- Coates AS, Winer EP, Goldhirsch A, et al. Tailoring therapiesimproving the management of early breast cancer: St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2015. Ann Oncol. 2015; 26(8): 1533-1546.
- Konecny G, Pauletti G, Pegram M, et al. Quantitative association between HER-2/neu and steroid hormone receptors in hormone receptor-positive primary breast cancer. J Natl Cancer Inst. 2003; 95(2): 142-153.
- Gutierrez C, Schiff R. HER 2: Biology, Detection, and Clinical Implications. Arch Pathol Lab Med. 2011; 135(1): 55-62.
- Correa GF, Reis-Filho JS. Microarray-based gene expression profiling as a clinical tool for breast cancer management: are we there yet? Int J Surg Pathol. 2009; 17: 285-302.
- Schnitt SJ. Will molecular classification replace traditional breast pathology? Int J Surg Pathol. 2010; 18: 162S-166S.
- Moncayo VM, Aarsvold JN, Alazraki NP. Lymphoscintigraphy and Sentinel Nodes. J Nucl. Med 2015; 56(6): 901-907.
- Arslan E, Cermik TF, Trabulus FDC, Talu ECK, Basaran S. Role of 18F-FDG PET/CT in evaluating molecular subtypes and clinicopathological features of primary breast cancer. Nucl Med Commun. 2018; 39(7): 680-690.
- Buck A, Schirrmeister H, Kuhn T et al. FDG uptake in breast cancer: correlation with biological and clinical prognostic parameters. Eur J Nucl Med Mol Imaging. 2002; 29(10): 1317- 1323.
- Lee SS, Bae SK, Park YS et al. Correlation of molecular subtypes of invasive ductal carcinoma of breast with glucose metabolism in FDG PET/CT: based on the recommendations of the St. Gallen Consensus Meeting 2013. Nucl Med Mol Imaging. 2017; 51(1): 79-85.
- Aogi K, Kadoya T, Sugawara Y et al. Utility of 18F FDG-PET/CT for predicting prognosis of luminal-type breast cancer. Breast Cancer Res Treat. 2015; 150(1): 209-217.
- Sorlie T, Perou CM, Tibshirani R et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA. 2001; 98(19): 10869- 10874.
- Bertos NR, Park M. Breast cancer: one term, many entities? J Clin Invest. 2011; 121(10): 3789-3796.
- Ko ES, Lee BH, Kim HA, Noh WC, Kim MS, Lee SA. Triple-negative breast cancer: correlation between imaging and pathological findings. Eur Radiol. 2010; 20(5): 1111-1117.
- Rashmi S, Kamala S, Murthy SS, Kotha S, Rao YS, Chaudhary KV. Predicting the molecular subtype of breast cancer based on mammography and ultrasound findings. Indian J Radiol Imaging. 2018; 28(3): 354-361.
- Wu T, Li J, Wang D et al. Identification of a correlation between the sonographic appearance and molecular subtype of invasive breast cancer: A review of 311 cases. Clinical Imaging. 2019; 53: 179-185.
- Uematsu T, Kasami M, Yuen S. Triple-negative breast cancer: correlation between MR imaging and pathologic findings. Radiology. 2009; 250(3): 638-647.
- Ueda S, Tsuda H, Asakawa H et al. Clinicopathological and prognostic relevance of uptake level using 18F-fluorodeoxyglucose positron emission tomography/computed tomography fusion imaging (18F-FDG PET/CT) in primary breast cancer. Jpn J Clin Oncol. 2008; 3894): 250-258.
- Kitajima K, Fukushima K, Miyoshi Y et al. Association between 18F-FDG uptake and molecular subtype of breast cancer. Eur J Nucl Med Mol Imaging. 2015; 42(9): 1371-1377.
- Higuchi T, Nishimukai A, Ozawa H et al. Prognostic significance of preoperative 18F-FDG PET/CT for breast cancer subtypes. Breast. 2016; 30: 5-12.
- Reddel RR, Sutherland RL. Tamoxifen stimulation of human breast cancer cell proliferation in vitro: A possible model for tamoxifen. Eur J Cancer Clin Oncol. 1984; 20(11): 1419-1424.
- Quon A, Gambhir SS. FDG-PET and Beyond: Molecular Breast Cancer Imaging. J Clin Oncol. 2005; 23(8): 1664-1673.
- Gabos Z, Sinha R, Hanson J et al. Prognostic significance of human epidermal growth factor receptor positivity for the development of brain metastasis after newly diagnosed breast cancer. J Clin Oncol. 2006; 24(36): 5658-5663.
- Wang Y, Ikeda DM, Narasimhan B et al. Estrogen receptornegative invasive breast cancer: imaging features of tumors with and without human epidermal growth factor receptor type 2 overexpression. Radiology. 2008; 246(2): 367-375.
- Youk JH, Son EJ, Chung J, Kim JA, Kim EK. Triple-negative invasive breast cancer on dynamic contrast-enhanced and diffusionweighted MR imaging: comparison with other breast cancer subtypes. Eur Radiol. 2012; 22(8): 1724-1734.
- Ballinger T, Kremer J, Miller K. Triple Negative Breast CancerReview of Current and Emerging Therapeutic Strategies. Oncology & Hematology Review. 2016; 12(2): 89-94.
- Schrading S, Kuhl CK. Mammographic, US, and MR imaging phenotypes of familial breast cancer. Radiology. 2008; 246(1): 58-70.
- Groheux D, Giacchetti S, Moretti JL et al. Correlation of high 18F-FDG uptake to clinical, pathological and biological prognostic factors in breast cancer. Eur J Nucl Med Mol Imaging. 2011; 38(3): 426-435.
- Keam B, Im SA, Koh Y et al. Early metabolic response using FDG PET/CT and molecular phenotypes of breast cancer treated with neoadjuvant chemotherapy. BMC Cancer. 2011; 11:452.
- Ekmekcioglu O, Aliyev A, Yilmaz S et al. Correlation of 18F-fluorodeoxyglucose uptake with histopathological prognostic factors in breast carcinoma. Nucl Med Commun. 2013; 34(11): 1055-1067.
- Giuliano AE, Edge SB, Hortobagyi GN. Eight edition of the AJCC cancer staging manual: breast cancer. Ann Surg Oncol. 2018; 1783-1785.
- Fallahpour S, Navaneelan T, De P, Borgo A. CMAJ Open. 2017; 5(3): E734-E739.
- Villman K, Sjostrom J, Heikkila R, et al. TOP2A and HER2 gene amplification as predictors of response to anthracycline treatment in breast cancer. Acta Oncol. 2006; 45(5): 590-596.
- Romond EH, Perez EA, Bryant J, et al. Trastuzumab plus adjuvant chemotherapy for operable HER2- positive breast cancer. N Engl J Med. 2005; 353(16): 1673-1684.
- Mosele F, Remon J, Mateo J et al. Recommendations for the use of next generation sequencing for patients with metastatic cancers: a report from the precision ESMO Precision Medicine Working Group. Ann Oncol. 2020; 31(11): 1491-1505.