Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
The role of cardiac magnetic resonance for arrhythmic risk stratification in a patient with myocarditis
Giuseppe Fede*; Giuseppe Abate; Giovanni Tasca; Nicoletta Guccione; Paola Belluardo; Carmelo Di Tommasi; Salvatore Solarino; Giuseppe Sulsenti; Maria Luisa Guarrella; Guglielmo Piccione; Sabina Ficili
Department of Cardiovascular Disease, Ospedale Maggiore, Modica, Italy
*Corresponding Author : Giuseppe Fede
Department of Cardiovascular Disease, Ospedale
Maggiore, Via Aldo Moro 1, 97015 - Modica, Italy
Email: g_fede@tiscali.it
Received : Dec 24, 2020
Accepted : Jan 18, 2021
Published : Jan 21, 2021
Archived : www.jcimcr.org
Copyright : © fede G (2021).
Abstract
Myocarditis is an inflammatory disease of cardiac muscle with a variable clinical presentation, ranging from asymptomatic cases to different degrees of left ventricular systolic dysfunction up to heart failure and dilated heart disease. Ventricular arrhythmias (VA) can occur in patients with myocarditis and implantable cardioverter defibrillator (ICD) may be indicated in patients with life-threatening VA who are not in the acute phase of myocarditis and who are receiving optimal medical therapy. Reduced left ventricular ejection fraction (LVEF) below 35%, which is used as the main criterion for stratifying the risk of sudden cardiac death (SCD), has low sensitivity and low specificity for arrhythmic risk stratification in patients with myocarditis. Myocardial scar is the main determinant for VA in these patients. Cardiac magnetic resonance imaging (CMR), using late gadolinium enhancement(LGE), has an excellent ability to determinate the extension and characterization of myocardial scar, indeed CMR can potentially improve SCD risk stratification and indication for ICD implantation in patients with myocarditis.
We present a case of a 36 years-old male presenting to the Emergency Department with a monomorphic sustained ventricular tachycardia in whom MRI revealed myocardial-pericardial recurrent inflammatory involvement and worsening disease progression. ICD was implanted in consideration of the high risk of life-threatening arrhythmias.
Keywords: Miocarditis, Ventricular arrhythmias, Sudden cardiac death, Cardiac magnetic resonance; Implantable cardioverter defibrillator.
Abbreviations: VA: Ventricular arrhythmias; SCD: Sudden cardiac deaths; ICD: Implantable cardioverter-defibrillator; DCM: Dilated cardiomyopathy; LVEF: Left ventricular ejection fraction; CMR: Cardiac magnetic resonance imaging; LGE: Late gadolinium enhancement; ECG:12-lead electrocardiogram; VT: Ventricular tachycardia; RBBB: Right bundle branch block; LV: Left ventricular; DE: Delayed enhancement
Citation: Fede G, Abate G, Tasca G, Guccione N, Belluardo P, et al. The role of cardiac magnetic resonance for arrhythmic risk stratification in a patient with myocarditis. J Clin Images Med Case Rep. 2021; 2(1): 1008.
Introduction
Myocarditis is an inflammatory disease of cardiac muscle caused by infectious and noninfectious conditions with a variable clinical presentation, ranging from asymptomatic or oligosymptomatic cases to different degrees of left ventricular systolic dysfunction up to heart failure and dilated heart disease [1,2]. Ventricular arrhythmias (VA) may occur in patients with myocarditis: while polymorphic VA are more common in the acute phase, monomorphic VA are more frequent in the chronic phase [3]. In a minority of cases, research has demonstrated fatal VA and sudden cardiac deaths (SCD) correlated with myocarditis [3-5]. Bradyarrhythmia and new onset of heart block can also happen in rare cases of myocarditis (eg, Lyme disease, sarcoidosis, giant cell myocarditis) [6].
Implantable cardioverter-defibrillator (ICD) implantation can be beneficial in patients with life-threatening VA or with dilated cardiomyopathy (DCM) and high risk of SCD [7]. Current risk stratification of primary prevention ICD implantation relies on a reduced left ventricular ejection fraction (LVEF) below 35% [7]. Although LVEF was an excellent predictor of total mortality and nonSCD mortality in patients with DCM, a huge number of studies have showed that LVEF is an unreliable surrogate to predict SCD [8,9]. In fact, the results demonstrated an unacceptable number of SCD [10,11] in patients with DCM and LVEF above 35%.
Myocardial scar is the main determinant for Vain ischemic and non-ischemic DCM [12]. Cardiac magnetic resonance imaging (CMR), using late gadolinium enhancement (LGE), has an excellent ability to determinate the extension and characterization of myocardial scar, indeed CMR can potentially improve SCD risk stratification and indication for ICD implantation [13,14].
Case presentation
A 36 years-old male was admitted to the Emergency Department complaining chest pain and palpitations arised playing soccer. Six months before he had suffered from acute pericarditis treated with non steroidal anti-inflammatory drugs and corticosteroid. He did not make any mention about other previous pathologies and there was not a family history of sudden cardiac death or cardiovascular diseases. He has rarely smoked and he occasionally drinked alcoholic beverage.
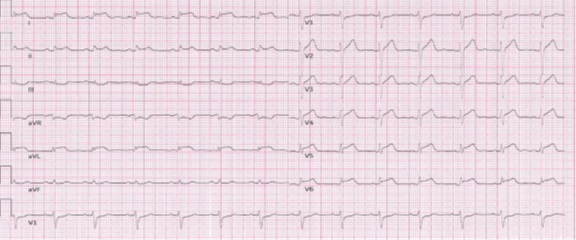
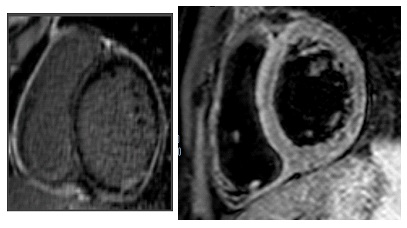
After his admission to the hospital a physical examination revealed a heart rate of 160 bpm and a normal blood pressure (125/70 mmHg), without other signs of heart failure. A 12-lead electrocardiogram (ECG) showed a monomorphic ventricular tachycardia (VT) with right bundle branch block (RBBB) morphology and left axis deviation (Figure 1). The VT was not stopped by either endovenous lidocaine or amiodarone; it was interrupted with endovenous metoplolol. Blood tests made known elevated high-sensitive troponin T levels (2,3 ng/mL, upper limit: 0.014 ng/ mL). Cardiotropic viral serology and autoantibody serum tests were negative. A trans-toracic echocardiogram showed mild left ventricular (LV) systolic dysfunction,with an LV ejection fraction (LVEF) of 48%, mild LV dilation (end-diastolic volume: 120 mL) and LV wall motion abnormalities of the posterior-inferior and lateral walls. Two days after his admission his ECG showed a ST-segment elevation in lead D1-D2-aVL-V5-V6 and reciprocal ST depression in lead D3 and aVR. A coronary angiography ruled out the coronary artery disease. CMR revealed mild LV systolic dysfunction (LVEF=47%), edema of inferior basal LV wall, severe hypokinesia of inferior basal LV wall associated with circumferential subepicardial delayed enhancement (DE) and intramyocardial DE of the septum, as a result of myocardial-pericardial recurrent inflammatory involvement. The patient was discharged on beta-blockers and ace-inhibitor with a diagnosis of recurrent myocardial pericarditis.
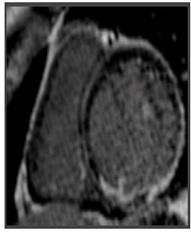
At 3 and 6 months follow up, the ventricular ectopic burden on 24 h-Holter ECG monitoring progressively increased to 20% and 35%, respectively. Moreover, a sustained monomorphic VT was registered at the Holter ECG monitoring (ventricular rate of 180 bpm, RBBB morphology). The patient was re-admitted for further tests. When the patient was admitted to the hospital he was haemodynamic stable without heart failure symptoms. A second CMR revealed LV dilation and a further reduction in LV systolic function (LVEF=40%); an almost transmural extent of DE in the inferior basal wall, subepicardial DE in lateral wall, and a persistence of intramyocardial DE in the interventricular septum were found, suggesting worsening disease progression. In consideration of the clinical-instrumental evolution and the high risk of lifethreatening arrhythmias although optimal medical therapy, it was suggested an ICD implantation.
Discussion
The case in this report could represent different stages of myocarditis: acute pericarditis, acute active myocarditis with mild impaired LV systolic function and monomorphic VT, chronic myocarditis with worsening disease progression characterized by LV dilatation, further impairment of LV systolic function and VT recurrence.
Acute myocarditis is often a transient condition followed by complete recovery, however VA can occur in the acute phase of the disease. Usually polymorphic VT are seen in acute myocarditis and monomorphic VT are more common in the chronic phase [3]. Symptomatic nonsustained VT can be treated with antiarrhythmic drugs whereas sustained VT requires urgent cardioversion followed by electrocardiographic monitoring and antiarrhythmic drugs in order to prevent the recurrence of arrhythmia [7]. ICD implantation is not indicated in the acute myocarditis because arrhythmia risk may resolve. A wearable cardioverter-defibrillator has a potential role in patients with acute myocarditis and high risk for SCD [15]. Conversely ICD implantation may be indicated in patients with life-threatening VA who are not in the acute phase of myocarditis and who are receiving optimal medical therapy. However to date, there are no specific indications for ICD implantation in patients with myocarditis, and LVEF is used as the main criterion for stratifying the risk of SCD in these patients [7].
Numerous investigations have proved that a reduced LVEF has low sensitivity and low specificity for the prediction of SCD [7]. In fact the majority of SCD cases occur in patients with preserved or moderately reduced LVEF [10,11]. Moreover relatively few patients with reduced LVEF will benefit from an ICD implantation because they will never experience a life-threatening arrhythmic event [16,17], therefore they are exposed to unnecessary procedural risks with useless healthcare costs [18].
Myocardial scar is the main substrate for re-entrant VT [19]. Diffuse fibrosis, commonly found in patients with DCM, can cause electrophysiological derangements leading to ventricular fibrillation. Both myocardial scar and diffuse fibrosis can be studied using CMR. LGE on CMR is able to detect myocardial scar with good accuracy [20] and is a robust predictor of VA and SCD in patients with DCM [21]. T1 mapping on CMR can quantified diffuse fibrosis and it is associated with worse outcomes in patients with DCM [22]. Thus, CMR using LGE and T1 mapping is a powerful tool for stratifying arrhythmic risk and indication for primary ICD in patients with DCM.
Conclusion
In conclusion CMR plays a pivotal role in patients with myocarditis [23]: it enables detection features of active infection as hyperemia, edema and accompanying pericardial effusion, it allows to quantify ventricular size and function (regional and global wall motion abnormalities, LVEF quantification), and more importantly, it is able to detect the presence of myocardial scar and the extension of myocardial fibrosis that are both associated with arrhythmic risk [21].
Future randomized clinical trials should investigate the role of MRI for arrhythmic risk stratification in patients with myocarditis. It can improve patients selection for ICD implantation and SCD prevention, bridging a gap still present in the current international guidelines [7].
References
- Canter CE, Simpson KE. Diagnosis and treatment of myocarditis in children in the current era. Circulation. 2014; 129(1): 115-128.
- Felker GM, Thompson RE, Hare JM, Hruban RH, Clemetson DE, Howard DL et al. Underlying causes and long-term survival in patients with initially unexplained cardiomyopathy. N Engl J Med 2000; 342(15): 1077-1084.
- Peretto G, Sala S, Rizzo S, Palmisano A, Esposito A, De CF et al. Ventricular Arrhythmias in Myocarditis: Characterization and Relationships With Myocardial Inflammation. J Am CollCardiol 2020; 75(9): 1046-1057.
- Eckart RE, Scoville SL, Campbell CL, Shry EA, Stajduhar KC, Potter RN et al. Sudden death in young adults: a 25-year review of autopsies in military recruits. Ann Intern Med. 2004; 141(11): 829-834.
- Maron BJ, Carney KP, Lever HM, Lewis JF, Barac I, Casey SA et al. Relationship of race to sudden cardiac death in competitive athletes with hypertrophic cardiomyopathy. J Am Coll Cardiol 2003; 41(6): 974-980.
- Cooper LT, Jr., Berry GJ, Shabetai R. Idiopathic giant-cell myocarditis- -natural history and treatment. Multicenter Giant Cell Myocarditis Study Group Investigators. N Engl J Med 1997; 336(26): 1860- 1866.
- Al-Khatib SM, Stevenson WG, Ackerman MJ, Bryant WJ, Callans DJ, Curtis AB et al. 2017 AHA/ACC/HRS Guideline for Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2018; 72(14): e91-e220.
- Pontone G, Guaricci AI, Andreini D, Solbiati A, Guglielmo M, Mushtaq S et al. Prognostic Benefit of Cardiac Magnetic Resonance Over Transthoracic Echocardiography for the Assessment of Ischemic and Nonischemic Dilated Cardiomyopathy Patients Referred for the Evaluation of Primary Prevention Implantable CardioverterDefibrillator Therapy. Circ Cardiovasc Imaging. 2016; 9(10).
- Wu KC, Calkins H. Powerlessness of a Number: Why Left Ventricular Ejection Fraction Matters Less for Sudden Cardiac Death Risk Assessment. Circ Cardiovasc Imaging. 2016; 9(10).
- Wellens HJ, Schwartz PJ, Lindemans FW, Buxton AE, Goldberger JJ, Hohnloser SH et al. Risk stratification for sudden cardiac death: current status and challenges for the future. Eur Heart J 2014; 35(25): 1642-1651.
- Stecker EC, Vickers C, Waltz J, Socoteanu C, John BT, Mariani R et al. Population-based analysis of sudden cardiac death with and without left ventricular systolic dysfunction: two-year findings from the Oregon Sudden Unexpected Death Study. J Am CollCardiol 2006; 47(6): 1161-1166.
- Horowitz LN, Josephson ME, Harken AH. Epicardial and endocardial activation during sustained ventricular tachycardia in man. Circulation 1980; 61(6): 1227-1238.
- Lehrke S, Lossnitzer D, SchÃb M, Steen H, Merten C, Kemmling H et al. Use of cardiovascular magnetic resonance for risk stratification in chronic heart failure: prognostic value of late gadolinium enhancement in patients with non-ischaemic dilated cardiomyopathy. Heart. 2011; 97(9): 727-732.
- Iles L, Pfluger H, Lefkovits L, Butler MJ, Kistler PM, Kaye DM et al. Myocardial fibrosis predicts appropriate device therapy in patients with implantable cardioverter-defibrillators for primary prevention of sudden cardiac death. J Am Coll Cardiol. 2011; 57(7): 821-828.
- Prochnau D, Surber R, Kuehnert H, Heinke M, Klein HU, Figulla HR. Successful use of a wearable cardioverter-defibrillator in myocarditis with normal ejection fraction. Clin Res Cardiol 2010; 99(2): 129-131.
- Tung R, Zimetbaum P, Josephson ME. A critical appraisal of implantable cardioverter-defibrillator therapy for the prevention of sudden cardiac death. J Am Coll Cardiol 2008; 52(14): 1111- 1121.
- Ghanbari H, Dalloul G, Hasan R, Daccarett M, Saba S, David S et al. Effectiveness of implantable cardioverter-defibrillators for the primary prevention of sudden cardiac death in women with advanced heart failure: a meta-analysis of randomized controlled trials. Arch Intern Med 2009; 169(16): 1500-1506.
- Maisel WH, Moynahan M, Zuckerman BD, Gross TP, Tovar OH, Tillman DB et al. Pacemaker and ICD generator malfunctions: analysis of Food and Drug Administration annual reports. JAMA 2006; 295(16): 1901-1906.
- Kober L, Thune JJ, Nielsen JC, Haarbo J, Videbæk L, Korup E et al. Defibrillator Implantation in Patients with Nonischemic Systolic Heart Failure. N Engl J Med. 2016; 375(13): 1221-1230.
- Iles LM, Ellims AH, Llewellyn H, Hare JL, Kaye DM, McLean CA et al. Histological validation of cardiac magnetic resonance analysis of regional and diffuse interstitial myocardial fibrosis. Eur Heart J Cardiovasc Imaging 2015; 16(1): 14-22.
- Di MA, Anguera I, Schmitt M, Klem I, Neilan TG, White JA et al. Late Gadolinium Enhancement and the Risk for Ventricular Arrhythmias or Sudden Death in Dilated Cardiomyopathy: Systematic Review and Meta-Analysis. JACC Heart Fail. 2017; 5(1):28-38.