Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Acute thrombectomy for saddle pulmonary embolus: Case presentation and review of management
Rajeev Shukla 1,2*; Adrian Pakavakis4; Prasanth Sadasivan Nair1; Anna Shukla3; Julian A Smith1; Prashant Joshi1
1 Department of Cardiothoracic Surgery, Monash Medical Centre, Clayton, VIC Australia.
2 Department of Surgery, University of Melbourne, Melbourne, VIC Australia.
3 School of Health Sciences, University of Melbourne, Melbourne, VIC Australia.
4 Department of Intensive Care Medicine, Monash Medical Centre, Clayton, VIC Australia.
*Corresponding Author : Rajeev Shukla
Department of Cardiothoracic Surgery, Monash Medical Centre, Clayton, VIC Australia.
Email: drshuklara@gmail.com
Received : Feb 11, 2021
Accepted : Mar 09, 2021
Published : Mar 11, 2021
Archived : www.jcimcr.org
Copyright : © Shukla R (2021).
Abstract
Pulmonary embolism is the third leading cause of cardiovascular death after myocardial infarction and stroke [1,2,4,5]. Anticoagulation is the primary choice of treatment for the majority of patients presenting with acute pulmonary embolism [2,4]. However, a greater risk of mortality in patients with right ventricular (RV) dysfunction may open the door to more aggressive treatment modalities [2]. We present the case of a patient who was diagnosed with a post-operative saddle pulmonary embolus that failed initial treatment with anticoagulation and required emergency thrombectomy to prevent mortality. A brief overview of treatment options is highlighted.
Keywords: Pulmonary embolism management, Pulmonary embolism guidelines, Saddle pulmonary embolism, Catheter based intervention.
Citation: Shukla R, Pakavakis A, Nair PS, Shukla A, Smith JA, Joshi P. Acute thrombectomy for saddle pulmonary embolus: case presentation and review of management. J Clin Images Med Case Rep. 2021; 2(2): 1023.
Introduction
Pulmonary embolism (PE) has a variable presentation ranging from incidental discovery in asymptomatic patients to severe haemodynamically instability, which therefore makes the diagnosis clinically challenging [1,2,3]. Mortality rates three months after PE are linked to the size of the embolus and the degree of RV strain and have been reported as high as 17% [3]. The mortality rates of patients presenting in cardiogenic shock can be as high as 30% [6]. PE is a preventable disease; with early recognition and the initiation of appropriate treatment, mortality can be prevented and associated morbidity significantly improved. Treatment modalities are initiated with the aid of risk stratification and deviation from this can result in potentially dire consequences.
Case presentation
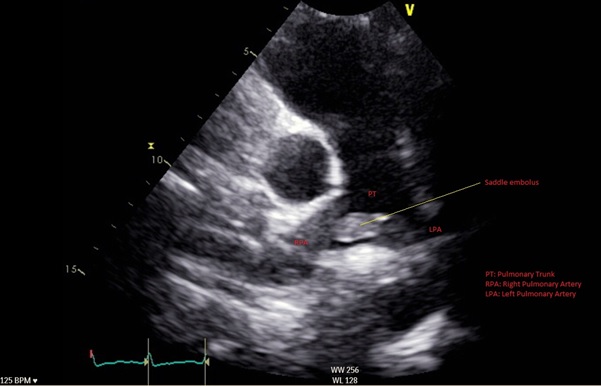
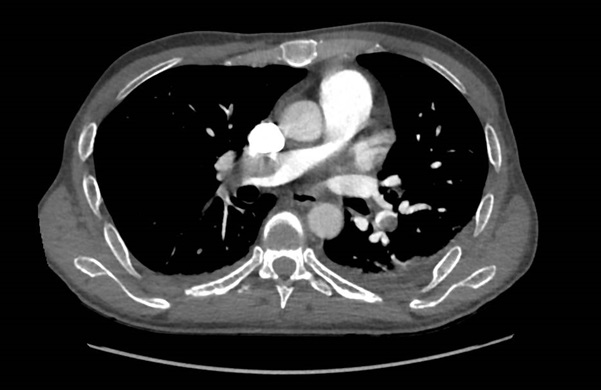
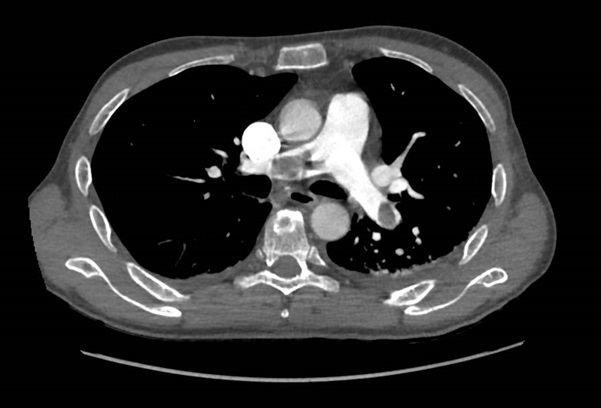
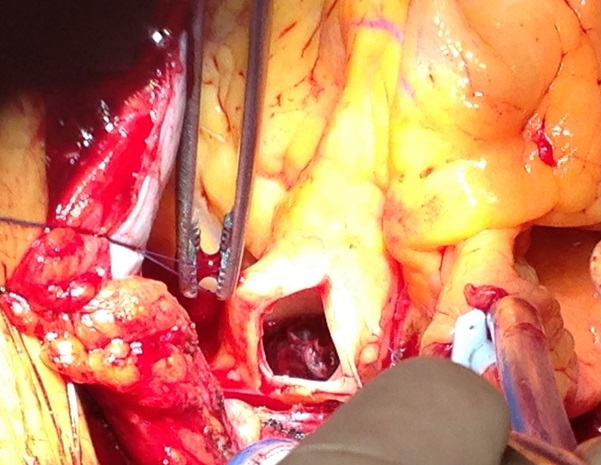
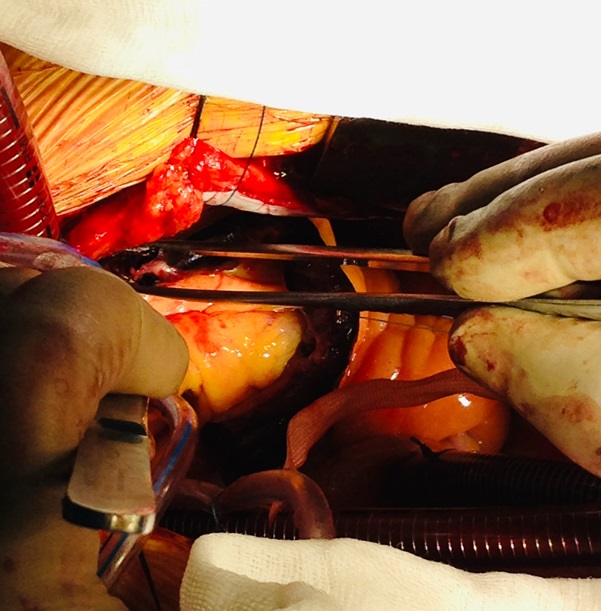
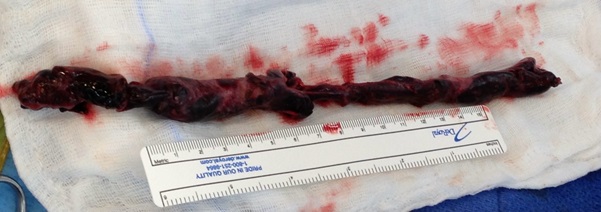
A 46-year-old man was admitted for management of an acute exacerbation of Crohn’s disease. His medical history included deep vein thrombosis six weeks earlier with obstructive thrombus in one of the commitant peroneal veins in the midcalf extending to 10 cm but with no extension above the knee. During the hospital admission the patient required a semi-elective laparoscopic total colectomy. After an initial uncomplicated recovery, the patient developed sudden onset hypoxia (SaO2 70% RA), sinus tachycardia (HR 135) and hypotension (BP 90/70 mmHg) on the second post-operative day. The patient required low dose inotropic support with noradrenaline to maintain haemodynamic stability. Electrocardiography demonstrated sinus tachycardia with S1Q3T3 features. A transthoracic echocardiogram (TTE) demonstrated a large saddle PE (Figure 1), dilated and severely hypokinetic right ventricle, dilated inferior vena cava (IVC) with no collapse during respiration and moderate pulmonary hypertension (PA systolic pressure 50 mmHg). An urgent computed tomography pulmonary angiogram (CTPA) was obtained and revealed a massive saddle pulmonary embolus (Figure 2) extending from the main pulmonary trunk across the bifurcation of the pulmonary trunk into both right and left main pulmonary arteries (Figure 3). Initially the patient was commenced on a therapeutic heparin infusion, however, continued haemodynamic instability warranted acute surgical pulmonary embolectomy. The patient was urgently transferred to the operating theatre where arterial and central venous lines were inserted. Following the induction of general anaesthesia the patient was intubated and ventilated. The surgical approach required median sternotomy and subsequent administration of heparin for preparation of cardiopulmonary bypass, which was achieved by placing cannulae in the ascending aorta and both cavae. A dose of antegrade cold blood cardioplegia was delivered on applying the cross clamp across the aorta, which achieved instantaneous electromechanical arrest. A vertical pulmonary arteriotomy was performed revealing a large thrombus extending into the left and right pulmonary arteries (Figure 4). The entire thrombus was carefully removed en-bloc (Figure 5 and 6). On completion of the embolectomy, the pulmonary artery was closed in two layers of 5-0 prolene. Intraatrial and intraventricular extension of the thrombus was excluded by perfoming a small right atriotomy, which was subsequently closed in two layers of 5-0 prolene. The patient was weaned from cardiopulmonary bypass and heparin reversed with protamine. Routine closure of the sternum and soft tissues completed the surgery. The patient was transferred to the intensive care unit in a haemodynamically stable condition on a low dose of noradrenaline. The post bypass transoesophageal echocardiogram showed improved contractility of the right ventricle with overall reduction in diameter. The patient was extubated on day 1 and made an uneventful recovery. The patient subsequently had an inferior vena cava filter inserted two days after embolectomy and was anticoagulated with warfarin. An ultrasound during this procedure revealed a left iliac vein clot which was presumed to be the source of the pulmonary embolus. Repeat TTE one week post embolectomy revealed normal RV ejection fraction with upper normal RV size. The IVC was mildly dilated with satisfactory inspiratory collapse. Pulmonary artery (PA) pressures were unobtainable. The patient was discharged from hospital thirteen days following embolectomy. Three months post discharge the patient suffered a small intracranial bleed withoutneurological injury. At seven years, the patient is alive and doing well with no residual cardiac dysfunction.
Discussion
The annual incidence of pulmonary embolism has been reported to be between 39-115/100,000 population globally and is the third leading cause of cardiovascular mortality behind myocardial infarction and stroke [1,2,4,5]. Diagnosis can often be challenging due to the spectrum of presentations ranging from asymptomatic to cardiovascular collapse and death [1-3]. Health organisations world-wide have begun to adopt multidisciplinary pulmonary embolism response teams (PERTs) who are tasked with convening in real-time to aid clinical decision regarding management - taking into account risk factors and risk stratification scores, such as the pulmonary embolism severity index (PESI) to assess risk of early mortality [1,2,5]. The PESI score stratifies patients into 5 levels of risk from very low to very high. It has been extensively validated and shown to be reliable in identifying patients at low risk of early mortality [1,5]. The AHA and ESC guidelines both support the use of risk stratification scores such as PESI or Geneva Pulmonary Embolism Score to help categorise patients into levels of risk and determine the need for further investigations and the final treatment pathway [5,8]. However, it is important to note that different risk stratification models assess different aspects of risk. The AHA/ESC categorise patients into high, intermediate and low risk of mortality resulting from PE in 30 days. In comparison the PESI scheme predicts the mortality in 30 days due to any cause [2,5]. Hence risk stratification should aid in clinical decision making but not be used to define absolute treatment modalities. ESC guidelines recommend early use of echocardiography even in patients determined as low-risk PE due to the significant increase in mortality in the presence of RV dysfunction [5].
Treatment
The treatment phases for acute PE can be divided into (1) haemodynamic and respiratory support and (2) intervention of acute pulmonary embolism.
(1) Haemodynamic and respiratory support
Severe PE compromises both circulation and gaseous exchange and presents as hypoxaemia and RV failure. The ventilation and perfusion mismatch that is observed in such cases requires careful, deliberate interventions to prevent further haemodynamic deterioration. RV dilatation occurs as a result of PA obstruction causing a sudden rise in PA pressures. The developing RV failure further compromises forward flow from RV to LA and consequently a fall in cardiac output. The complex physiological changes require judicious and careful initiation of all supportive interventions. Supplemental oxygen is preferred via non-invasive routes in the first instance. Mechanical ventilation should be reserved for patients who are unable to tolerate non-invasive oxygenation and the use of anaesthetic induction agents which can cause hypotension should be avoided. Positive-pressure ventilation should be selected at the lowest tolerable pressures to prevent further reduction in venous return. Volume replacement should be given judiciously in order to prevent over distention of a failing RV, thereby compromising cardiac output. Such replacement should be guided by invasive CVP monitoring or TTE. Inotropic use has been shown to be effective in the haemodynamically unstable patient by improving coronary perfusion [5].
(2) Intervention of acute pulmonary embolism
Anticoagulation: Both the ESC and AHA recommend that in the absence of absolute contraindication, therapeutic anticoagulation with low molecular weight heparin (LMWH) or fondaparinux is commenced while concomitantly performing appropriate investigations for all risk categories of PE. AHA also recommend the use of unfractionated heparin (UFH), however, the ESC guidelines tend not to prefer UFH due to it carrying a higher risk of bleeding and heparin-induced thrombocytopaenia. ESC recommend reserving UFH use to patients with severe renal impairment (CrCl <30 ml/min), severe obesity or imminent haemodynamic decompensation requiring reperfusion intervention [5,8].
Thrombolysis: The ESC and AHA recommend thrombolysis for intermediate and high risk PE. Studies have shown a faster improvement in RV function, reduction in PA pressure and pulmonary obstruction when thrombolysis is commenced within 48 hours of onset of symptoms, although intermediate-risk patients experienced a higher risk of severe extracranial and intracranial bleeding. An overall reduction in mortality rates of 50-60% have been reported for the latter category of patients. Early thrombolysis and its effect on long term sequalae of acute PE such as chronic pulmonary hypertension remain unknown, and results from the PEITHO trial confirm no role for thrombolysis for intermediate PE for the purpose of preventing long term sequalae [5,8].
Catheter Based Interventions: These interventions are proposed for patients who are categorised as high or intermediate risk PE with absolute or relative contraindications to systemic thrombolysis, failed thrombolysis or at risk of significant clinical deterioration prior to the thrombolytic effect [1,5,7,8]. Two types of intervention exist:
Embolectomy – Involves a catheter being navigated to the site of clot burden and retrieval of the clot by aspiration. Studies have shown these interventions to significantly reduced RV/LV pressure ratio but only produce modest reduction in PA pressure. No intracerebral bleeding events have been reported [7].
Catheter directed thrombolysis – Involves the delivery of thrombolytics locally to the site of clot burden, thereby excluding the use of systemic thrombolytics. This can be achieved by use of a low-power, high-frequency ultrasound catheter. The ULTIMA trial demonstrated improvement in RV function and PA pressure in the short-term, however, the long-term benefit remains unclear. Bleeding rates are higher than those observed by systemic anticoagulation but lower than systemic thrombolysis [5,7,8].
The AHA and ESC recommend the use of catheter based interventions in high risk patients in whom thrombolysis has failed or is contraindicated. Given the lack of prospective, randomised trials examining catheter-based interventions, Theroux et al propose that patients in the high and intermediate risk PE category be considered on a case-by-case basis with discussion at the PERT team level [5,7,8].
Surgical embolectomy: is reserved for patients in the high or intermediate risk PE categories with absolute contraindications to thrombolysis or at risk of rapid clinical deterioration prior to the effect of systemic thrombolysis or as a rescue intervention where thrombolysis has failed. Studies have shown that prompt surgical intervention significantly improves RV function and PA pressure with a lower risk of major bleeding when compared with thrombolysis. No difference in 30 day mortality was found between surgical embolectomy and thrombolysis (13% and 15%), however, stroke and re-intervention rates within 30 days was higher with thrombolysis [5,7,8].
Conclusion
We present the case of a 46-year-old man who had successful emergency surgical embolectomy for a high risk saddle pulmonary embolism. RV dysfunction and PA pressure returned to normal post procedure. The patient is alive at 7 years and his echocardiogram at 5 years post embolectomy remains stable with mildly dilated right ventricle and normal right ventricular function. Both the AHA and ESC guidelines make recommendations aimed at improving the morbidity and mortality of patients presenting with acute, high and intermediate risk PE, which include the early involvement of the PERT team to aid real-time clinical assessment and decision for treatment options and the early use of echocardiography to assess RV dysfunction – even in low risk PE [1,2,5]. Thrombolysis, catheter based and surgical interventions for treatment of acute PE are endorsed by AHA and ESC. Both catheter-based and surgical interventions require specialist expertise and therefore outcomes are variable and reflective of this [5,7,8]. In our centre we offer both services, however, we have demonstrated that with early surgical intervention by an experienced team, excellent outcomes can be achieved without the risk of stroke, bleeding or re-intervention that is inherent with thrombolytic interventions.
References
- Giri J, Sista AK, Weingberg I et al. Interventional therapies for acute pulmonary embolism: Current status and principles for the development of novel evidence. A scientific statement from the American Heart Association. Circulation. 2019; 140(20): e774- e801.
- Tice C, Seigerman M, Fiorilli P et al. Management of acute pulmonary embolism. Curr Cardiovasc Risk Rep. 2020; 14(24): 1-11.
- Cooper JM, Beckman JA. Massive pulmonary embolism: A remarkable case and review of treatment. Vascular Medicine. 2002; 7: 181-185.
- Namana V, Siddiqui S, Balasubramanian R et al. Saddle pulmonary embolism: Right ventricular strain an indicator for early surgical approach. Oxford Medical Case Reports. 2016; 6: 130-134.
- Konstantinides SV, Meyer G, Becattini C et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). European Heart Journal. 2020; 41: 543-603.
- Bêlohlávek J, Dytrych V and Linhart A. Pulmonary embolism, part I: Epidemiology, risk factors and risk stratification, pathophysiology, clinical presentation, diagnosis and nonthrombotic pulmonary embolism. Exp Clin Cardiol. 2013; 18(2): 129-138.
- Theroux C, Aliotta J, Mullin C J. High risk pulmonary embolism: Current evidence-based practices. R I Med J. 2019; 102(10): 43-47.
- Jaff MR, McMurtry SM, Archer LS et al. AHA Scientific Statement: Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis and chronic thromboembolic pulmonary hypertension. Circulation. 2011; 123: 1788-1830.