Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Idiopathic normal pressure hydrocephalus presenting with paroxysmal paraparesis as a cardinal symptom: A case presentation
Andreas Eleftheriou1 *; Christina Koumantzia2
1 Department of Neurology and Department of Clinical and Experimental Medicine, Linköping University, Linköping, Sweden.
2 Department of Radiology in Linköping, and Department of Health, Medicine and Caring Sciences, Linköping University, Linköping, Sweden
*Corresponding Author : Andreas Eleftheriou
Department of Neurology, Institution of Clinical
and Experimental Medicine, Linköping University,
Linköping, Sweden.
Email: andreas.eleftheriou@regionostergotland.se
Received : Feb 05, 2021
Accepted : Mar 15, 2021
Published : Mar 17, 2021
Archived : www.jcimcr.org
Copyright : © Eleftheriou A (2021).
Abstract
Idiopathic normal pressure hydrocephalus (iNPH) is a gradually progressive disease affecting the elderly population. The diagnosis of probable iNPH is based on clinical history, physical findings, brain imaging. INPH involves non-obstructive enlargement of the cerebral ventricles combined with one or more symptoms of cognitive decline, impaired gait and balance, and urinary urgency. Our case describes an unusual coexistence of radiological and clinical iNPH symptoms with paroxysmal paraparesis.
Keywords: Idiopathic normal pressure hydrocephalus, Paraparesis, Water-hammer.
Citation: Eleftheriou A, Koumantzia C. Idiopathic normal pressure hydrocephalus presenting with paroxysmal paraparesis as a cardinal symptom: A case presentation. J Clin Images Med Case Rep. 2021; 2(2): 1028.
Introduction
Idiopathic normal pressure hydrocephalus (iNPH) is a gradually progressive disease with higher prevalence in the elderly population. In a recent population-based study, the prevalence of iNPH among individuals 80 years and older, was 5.9% to 8.9 [1,2]. The diagnosis of probable iNPH is based on clinical history, physical findings, brain imaging with Computed Tomography (CT) or/and Magnetic Resonance Imaging (MRI), and physiological criteria according to the International iNPH guidelines from 2005 and the Japanese guidelines from 2008 and 2012 [3-5]. In order to be diagnosed as having probable iNPH a patient needs to have clinical symptoms such as impaired gait and/or balance combined with either cognitive decline or/and urinary urgency [6]. Nevertheless, in the literature there are case reports describing a lot of other symptoms in patients with possible iNPH, such as neuropsychiatric symptoms [7], parkinsonian symptoms [8], depression [9], schizophrenia [8], ALS [10] and seizures [11]. Herein, we report an unusual manifestation of possible iNPH disease with paroxysmal paraparesis in the lower extremities.
Case presentation
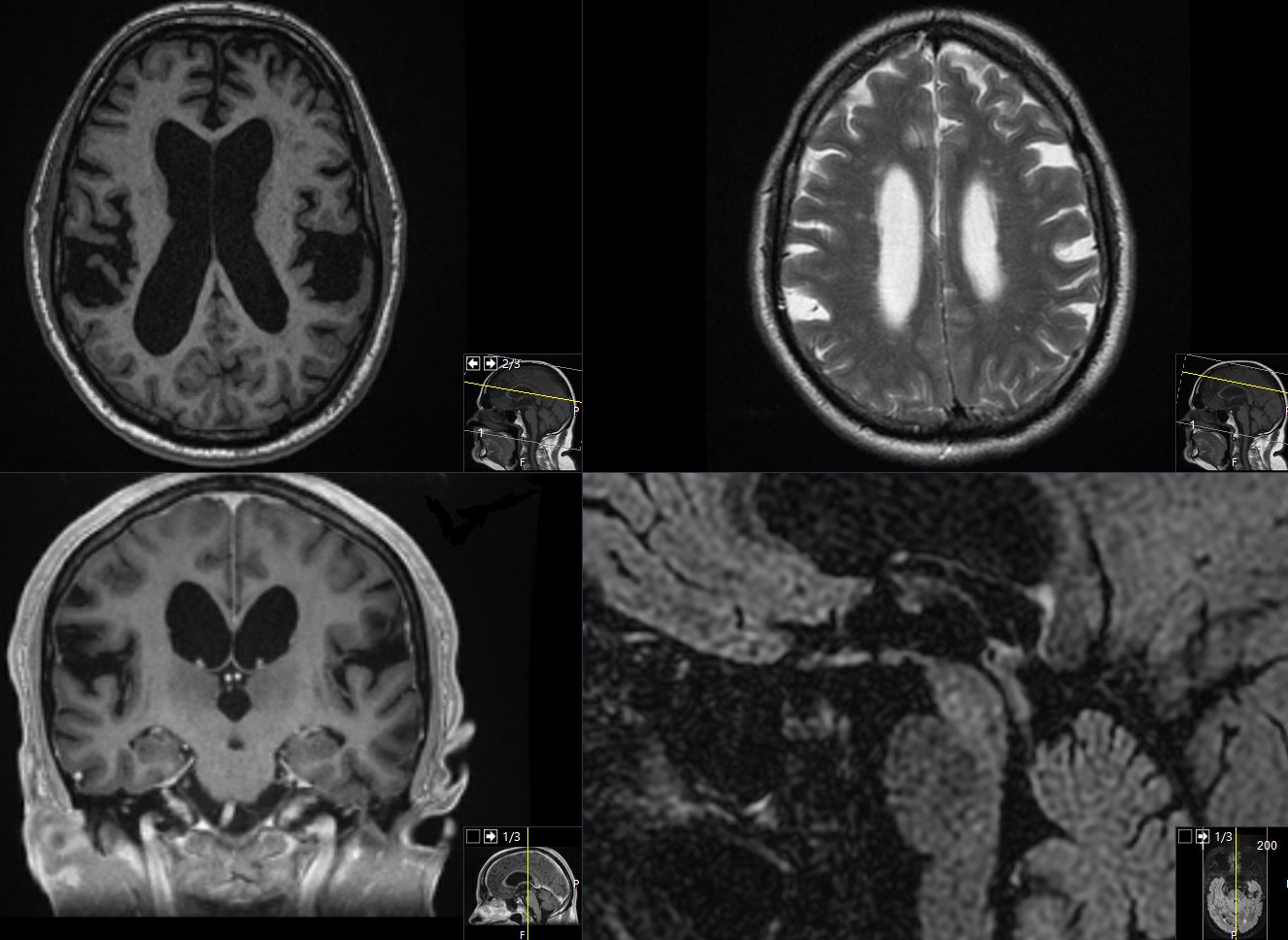
A 79-year-old Caucasian man, non-smoker, with hypertension, hyperlipidemia and sleep apnea was referred to our outpatient department at Linköping university hospital, because of episodes of paroxysmal abrupt paraparesis in the last two years. The patient experienced four episodes characterized by an acute weakness of lower extremities, after almost 30 minutes of normal exercise, without unconsciousness, seizures or other cardiorespiratory symptoms. The first time he was admitted to the cardiology department. A 24-hour-telemetry showed no arrhythmia. The orthostatic hypotension test such as extensive blood tests were normal. The Electroencephalogram (EEG), the Electroneurography (ENG) and the Electromyography (EMG) were normal. The brain MRI revealed an obvious ventriculomegaly disproportionate to cerebral atrophy, Evans index was at 0.37, corpus callosum thinning and elevation with a callosal angle at 68o [12], widening of temporal horns (without hippocampal atrophy) and widening of third ventricle at 11 mm, there was an aqueductal flow void, narrowing of sulci- and subarachnoid spaces over the high convexity and midline surface of the brain and ballooning of frontal horns. There was no vascular microangiopathy at the periventricular white matter (Figure 1). The radiological imaging fulfilled the criteria for possible iNPH diagnosis according to the International criteria from 2005 [6]. The MRI of whole spinal cord was normal.
Almost one year later, the patient developed urinary urgency daytime. Neither sleep disorder nor olfactory dysfunction were noticed. Romberg test, start-stop test, reflexes, limp-power test, ataxia test, Grasset test were normal under neurological investigation. Gait test was compatible to iNPH disease with decreased step-height and length, decreased cadence, increased trunk sway during walking, turned-out toes on walking, widened standing base, turning bloc and retropulsion. A physiotherapist evaluation was performed and the patient needed 11 seconds (sec) for Timed Up and Go (TUG)-time, 15 steps for TUG-step test, 10 sec for 10 meters walk time-test (10mwt) and 17 steps for 10 meters walk step-test (10 mws). Cognitive evaluation from iNPH-team’s occupational therapist showed psychomotor slowing with increased response latency, a Ray Auditory Verbal Learning Test score with 33 words, impairment to maintenance attention, decreased fine motor accuracy and spread with both hands, a Stoop Color-Word test with a time of 81 sec and Stoop interference with a time of 139 sec and MMSE 30/30.
A CSF tap test was performed in recumbent position. Once CSF was obtained, a spinal fluid manometer (Optidynamic, Mediplast, Italy) was connected to measure the CSF pressure in cm H2 0. The pressure was measured during a period of one minute to avoid artificially elevated levels. The patient was relaxed and had his neck to a neutral position and his legs extended. The CSF tap test showed a lumbar pressure of 22 cm H2 0, 50 ml CSF were drained. After the CSF tap test, physiotherapist’s evaluation showed TUGt 9 sec, TUGs 13 steps, 10 wmt 9 sec and 10 mws 16 steps. We noticed a qualitative and a quantitative improvement of patient’s status through video analysis of all gait controls from the first investigation until the last one in our department. CSF biomarkers showed β-amyloid at 470 ng/L (normal >620 ng/L), amyloid β 42/40 at 0.88 (normal >0.61), tau at 160 (normal <479 ng/L), phosphorylated tau 21 ng/L (normal <61 ng/L) and GFAp at 490 ng/L (normal <1250 ng/L). CSF was without pleocytosis, albumin ratio and glucose level were normal, Angiotensin Converting Enzyme was normal; there were no intrathecal oligoclonal immunoglobulin G bands, Borrelia burgdorferi specific antibodies were negative in the CSF and blood. No malign cells were identified in the cytological examination. Toxoplasma-DNA, Cytomegalovirus, Epstein-Barr virus, Herpes simplex virus, Human alphaherpesvirus-3 serology were normal. The patient was regarded to the neurosurgery department as a candidate for shunt operation.
Discussion
Acute flaccid paralysis (AFP) is a clinical syndrome characterized by rapid onset of weakness [13]. The differential diagnosis of AFP is wide [14]. In our case, given that MRI of whole spinal cord was normal, diagnoses such as acute myelopathy, trauma or spinal cord compression were excluded. Acute hypokalemic periodic paralysis, as well as thyrotoxic periodic paralysis were easily ruled out with blood sample analysis. Given that our patient’s medical history was free from psychiatric disorders as well as alcohol abuse, psychogenic problems were not possible underlying cause of the patient’s symptomatology. Atonic seizures should always be included in differential diagnosis of AFP, but in our case the normal EEG made this diagnosis less possible [15]. Neurologic disorders associated with viruses were easily excluded based on both medical history of our patient and negative laboratory tests. Given that the ENG and EMG were normal, diseases such as acute motor axonal neuropathy and peripheral neuropathies were excluded from the differential diagnosis. Disorders of the muscle such as inflammatory polymyositis associated with viral, parasitic, or bacterial infections were excluded with laboratory tests, while genetic tests in order to exclude muscular dystrophies were not performed [14].
Several possible mechanisms have been proposed to explain the pathophysiology of iNPH, namely tissue distortion due to raised intracranial pressure, interstitial oedema with stagnation of fluid causing decreased clearance of toxic metabolites, and impaired cerebral blood flow in the subcortical areas resulting in defective regional auto-regulation and ischemia [16]. The so-called “water-hammer” effect of the CSF pulse has been considered as a pathogenic factor in the development of normal-pressure hydrocephalus. Based on the “water-hammer” effect, each cardiac pulse could affect the CSF flow between the 3rd and 4th ventricle at the level of cerebral aqueduct of Sylvius, something that could periodically cause block to the fibers of the corticospinal pathway through the mechanical pressure against the red nuclei which are situated within the tegmentum of the midbrain, just behind the 4th ventricle [17].
As our patient was a clear responder to CSF tap test, the probability that the patient suffered from iNPH was high. Our case describes an unusual coexistence of radiological and clinical iNPH symptoms with paraparesis. Taking into consideration that the causes of AFP named above were excluded in our case, our hypothesis is that the “water-hammer” effect could possibly contribute to paraparesis pathogenesis in our patient with iNPH, but this remains theoretical, and further studies are required.
Our patient underwent iNPH investigation and was found eligible for shunt operation, which still is not performed. It is of great interest if the patient’s paraparesis should disappear postoperatively, something that will be evaluated at follow-up.
Conclusion
Our case describes an unusual coexistence of radiological and clinical iNPH symptoms with paroxysmal paraparesis. We could consider paraparesis as the cardinal symptom in our patient with iNPH according to our hypothesis based on the “water-hammer” effect, but this pathophysiological mechanism remains unstudied and requires research.
Additional information
Ethics approval and consent to participate: There is no need for ethics approval.
Availability of data and material: We used the data from our Cambio COSMIC Healthcare System, which is a digital comprehensive healthcare system installed in all clinics in our region. Radiological material obtained through Sectra Image Display System 7.
Competing interests: The authors declare that they have no competing interests.
Authors’ contributions: Andreas Eleftheriou and Christina Koumantzia were the authors of this manuscript. Andreas Eleftheriou was the neurologist who performed the neurological examination.
Conflict of interest: The authors have no conflict of interest to disclose.
Acknowledgements: Thanks to physiotherapist Johanna Rydja and occupational therapist Katarina Owen for their assessments of motor and cognitive function, respectively.
References
- Andersson J, et al. Prevalence of idiopathic normal pressure hydrocephalus: A prospective, population-based study. PLoS One, 2019: 14(5); e0217705
- Jaraj D, et al. Prevalence of idiopathic normal-pressure hydrocephalus. Neurology, 2014: 82(16); 1449-54.
- Ishikawa M, et al. Guidelines for management of idiopathic normal pressure hydrocephalus. Neurol Med Chir (Tokyo), 2008: 48 Suppl; S1-23.
- Marmarou A, et al. Guidelines for management of idiopathic normal pressure hydrocephalus: progress to date. Acta Neurochir Suppl. 2005: 95; 237-40.
- Mori E, et al. Guidelines for management of idiopathic normal pressure hydrocephalus: Second edition. Neurol Med Chir (Tokyo). 2012: 52(11); 775-809.
- Relkin N, et al. Diagnosing idiopathic normal-pressure hydrocephalus. Neurosurgery, 2005. 57(3 Suppl): p. S4-16; discussion ii-v
- Chatziioannidis S, et al. Psychotic symptoms in normal pressure hydrocephalus. Psychiatriki, 2013. 24(3): 217-24.
- Molde K, Soderstrom L, Laurell K. Parkinsonian symptoms in normal pressure hydrocephalus: A population-based study. J Neurol. 2017: 264(10); 2141-2148.
- Israelsson H, et al. Symptoms of depression are common in patients with idiopathic normal pressure hydrocephalus: The INPH-CRasH Study. Neurosurgery. 2016: 78(2); 161-8.
- Eleftheriou A, Nilsson M, Lundin F. A patient with idiopathic normal pressure hydrocephalus and amyotrophic lateral sclerosis. Coincidence or a genetic link between the diseases? EJMCR. 2020: 4(5); 157-160.
- Eleftheriou A, Amezcua S, Nilsson M. Idiopathic normal pressure hydrocephalus presenting with epileptic seizure as a cardinal symptom: A case presentation. Interdisciplinary Neurosurgery. 2020: 19; 100618.
- Kockum K, et al. The idiopathic normal-pressure hydrocephalus Radscale: a radiological scale for structured evaluation. Eur J Neurol. 2018: 25(3); 569-576.
- Growdon JH, Braunwald E, Wilson JD, eds. Harrison’s principles of internal medicine. New York, NY: McGraw- Hill Book Company, 1994: 115-25.
- Marx A, Glass JD, Sutter RW. Differential diagnosis of acute flaccid paralysis and its role in poliomyelitis surveillance. Epidemiol Rev. 2000: 22(2); 298-316.
- Satow T, et al. Partial epilepsy manifesting atonic seizure: report of two cases. Epilepsia. 2002: 43(11); 1425-31.
- Keong NC, et al. Imaging normal pressure hydrocephalus: Theories, techniques and challenges. Neurosurg Focus. 2016: 41(3); E11.
- Zisimopoulou V, et al. Vertebrobasilar dolichoectasia induced hydrocephalus: The water-hammer effect. Clin Pract. 2015: 5(2); 749.