
Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Distinctive COVID-19 manifestations and re-exposure to SARS CoV 2 in clinical laboratory personnel under regular monitoring by RT-PCR and serology
Fábio de Oliveira Martinez Alonso1; Bruno Duarte Sabino1; Marianna Tavares Venceslau2; Rafael Brandão Varella3*
1Contraprova Diagnósticos, Rio de Janeiro, Brazil.
2Departament of Preventive Medicine, University Hospital Clementino Fraga Filho, Federal University of Rio de Janeiro, Brazil
3Department of Microbiology and Parasitology, Biomedical Institute, Fluminense Federal University, Brazil.
*Corresponding Author : Rafael Brandão Varella
Laboratory of Virology, Biomedical Institute-UFF, Brazil. 24210-130
Email: rvarella@id.uff.br
Received : Feb 22, 2021
Accepted : Mar 20, 2021
Published : Mar 22, 2021
Archived : www.jcimcr.org
Copyright : © Varella RB (2021).
Abstract
Clinical laboratory personnel (CLP) present a unique opportunity to investigate distinct forms of COVID-19 as they are under constant testing for SARS CoV 2 infection. SARS CoV 2 RNA and antibodies were routinely investigated over a 5-month period in 26 professionals from a clinical laboratory in RJ, Brazil. Of them, three (11.5%) CLP presented the following peculiar COVID-19 manifestations: 2/26 (7.7%) had SARS CoV 2 antibodies without RNA detection during the follow-up, with a possible re-exposure in one case, and 1/26 (3.8%) a confirmed reinfection with RNA detection, and possibly a third re-exposure. Based on a long follow-up of SARS CoV 2 infection in CLP, this study showed that cases of COVID-19 without RNA detection are not common, but it does indicate the risk of re-exposure after the fall of antibody levels. Although scarcely reported, the investigation of less frequent forms of COVID-19 is relevant, given the lack of knowledge of its impact on the pandemic.
Keywords: SARS CoV 2, Healthcare workers, COVID-19, Antibody detection, qRT-PCR.
Citation: Alonso FM, Sabino BD, Venceslau MT, Varella RB. Distinctive COVID-19 manifestations and re-exposure to SARS CoV 2 in clinical laboratory personnel under regular monitoring by RT-PCR and serology. J Clin Images Med Case Rep. 2021; 2(2): 1032.
Introduction
Tracing COVID-19 cases and contacts has been acknowledged as one of the pivotal strategies for controlling the pandemic [1]. The use of real-time reverse-transcription PCR (qRT-PCR) is considered the main tool for identifying acute infection, specially within the first week of symptom onset [2], while serology is useful after 10-14 days of infection to investigate previous exposure to SARS CoV 2 [3].
Whereas qRT-PCR is considered the gold standard for SARSCoV-2 diagnosis [4], different reports have pointed out negative results in individuals who developed clinical symptoms, characteristic CT images and antibodies for COVID-19 infection [5] Although it is still poorly reported, its impact on epidemiology is not clear but should be considered, given its particular impact on the most susceptible populations. Another feature of COVID-19 pandemic is the decrease in SARS-CoV-2 antibody titers over time, potentially exposing individuals to re-infections [6].
The possibility of identifying these nuances during the pandemic is limited, since the general population is tested in a timely manner. Meantime, health professionals constitute a unique population to investigate different COVID-19 presentations, effects of prolonged exposure and adaptive immunological response, due to their constant exposure to infected patients [7], clinical samples, and routine testing. In this report, we present three distinctive COVID-19 cases in a cohort of CLP under regular testing.
Case description
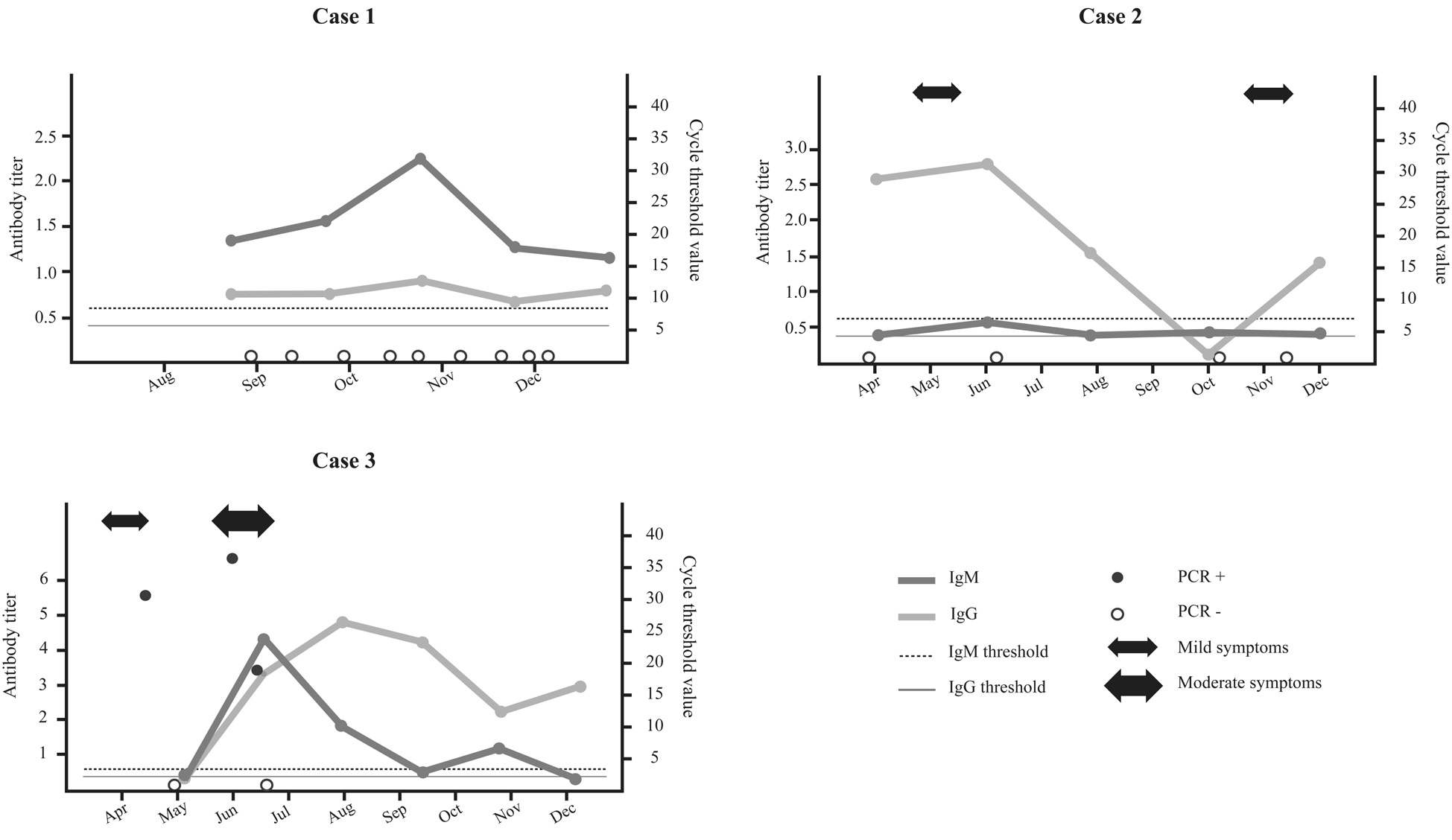
Twenty-six Laboratory staff of the private clinical laboratory Contraprova Ltd. located in Niteroi-RJ, Brazil, were routinely tested for SARS-CoV-2 infection. For such, respiratory material was collected and submitted to qRT-PCR for SARS-CoV-2 [Allplex™2019‐nCoV Assay (Seegene Inc., Seoul, Korea)]. ELISA test (Vircell, Spain) was used to investigate SARS-CoV-2 specific IgM and IgG antibodies. Of them, 3 (11.5%) individuals, all adults under 40s without comorbidities, presented distinct COVID-19 features. Figure 1 illustrates the cases described below:
Case #1: female, 26 years old, followed since August 2020 and without detection of SARS-CoV-2 RNA during testing. Serology pointed to sustained low IgG titers and higher IgM titers, peaking in October. She never showed symptoms of COVID-19, but had constant close contact with her mother, deceased from COVID19.
Case #2: female, 34 years old, followed since April 2020 also without any detectable SARS-CoV-2 RNA, although a 3-month period of testing was missing. IgG levels peaked in July, becoming undetectable in October. She reported mild COVID-19 symptoms in May 2020 along with close family members, and again in November 2020, coinciding with re-detectable IgG levels in November. IgM levels never turned positive.
Case #3: male, 25 years old, tested positive for SARS-CoV-2 RNA in April, negative in May and positive again in June. He had mild symptoms in April and more severe symptoms in June, characterizing a possible re-infection. Although SARS-CoV-2 RNA was detectable since April, IgG and IgM antibodies only became detectable in late June, coinciding with the second stronger symptomatic period. After its peak in June (IgM) and July (IgG), when antibody titers started to decline, IgM and IgG levels raised again in August (IgM) and October (IgG), possibly indicating another episode of SARS-CoV-2 infection, although without subsequent PCR detection or COVID-19 symptoms.
Discussion
In this paper, we presented three distinctive cases of COVID-19 out of 26 SARS CLP under regular monitoring. The frequency of patients without RNA detection with reagent ELISA varies significantly between studies, with values ranging from 34% [8] to 66.7% [4] being reported. Such values are higher than those presented in this study (7.7%), although such variations may be related to the methodology used, sampling, and frequency of testing.
Although negative PCR in ELISA positive individuals have been described before, the follow-up is scarcely reported. Indeed, many factors may contribute to negative PCR results, such as viral loads under detection limit [4], genetic mutations [9], inadequate or late sampling [5]. However, given the regularity of testing and the use of three different targets (genes E, N and S) in qRT-PCR methodology, we could infer that most of the samples were negative for SARS CoV 2 RNA or under detection limit, which was recently demonstrated with the use of the digital PCR [8].
Although Cases 1 and 2 never presented detectable SARS CoV 2 RNA, the context of infection and the antibody response differed. Case 1 probably had a constant contact with SARS CoV 2 through her severely COVID-19 infected mother, which probably prevented IgG levels to lower, but not enough to produce detectable PCR and clinical manifestations. Case 2, in turn, reported two episodes of COVID-like syndrome with an evident IgG response following each episode. Curiously, when IgG levels turned undetectable by October, Case 2 reported another COVID-like syndrome, with another IgG response, indicating a possible re-infection. The transmission capacity of both cases was not determined, but its potential has been demonstrated previously [4].
Case 3, on the other hand, developed differently, and was partially discussed in a previous paper [10]. Nonetheless, the continued monitoring indicates that after the fall in serum antibody levels from July to undetectable in September (IgM), Case 3 has probably been re-exposed to the virus for a third time, judging by the increase of IgG and IgM titers in December, although PCR remained negative. Differently from Case 2, he never experienced clinical symptoms after the second episode.
One common feature of all three cases is the rise of IgG titers in December, which coincides with an exponential increase in cases that have occurred since mid-November in the state [11]. It is plausible that these individuals were exposed in this wave of cases, which may have caused this effect on the level of antibodies. Case 3, who experienced more severe symptoms in the second episode, with lung injury, may have developed a stronger immunity that prevented a new clinical manifestation, unlike Case 2, who had milder symptoms.
This study has some limitations. Being asymptomatic, Cases 1 and 2 were not submitted do CT scan to investigate typical COVID19 lesions. It is possible that the interval between sample collections, especially in Cases 2 and 3, could have missed a potential RNA detection. Also, SARS CoV 2 genetic mutations in Cases 1 and 2 were not investigated. Despite these setbacks, to our knowledge this is one of the longest follow-up of distinct cases of COVID-19, whose frequency in general population and potential impact of transmission are unknown.
Acknowledgements: This paper was supported by LaboratórioContraprovaAnálises, Ensino e Pesquisas LTDA and by the National Council for Scientific and Technological Development – CNPq (Varella, RB).
Ethical approval: The study was approved by the University Hospital Ethical Committee of the Fluminense Federal University (register 30926020.2.0000.5243).
References
- Jin Y, Wang M, Zuo Z, Fan C, Ye F, Cai Z, et al. Diagnostic value and dynamic variance of serum antibody in coronavirus disease 2019. Int J Infect Dis. 2020; 94: 49-52.
- Walsh KA, Jordan K, Clyne B, Rohde D, Drummond L, Byrne P, et al. SARS-CoV-2 detection, viral load and infectivity over the course of an infection. J Infect. 2020; 81(3): 357-371.
- Kellam P, Barclay W. The dynamics of humoral immune responses following SARS-CoV-2 infection and the potential for reinfection. J Gen Virol. 2020; 101(8): 791-797.
- Cao G, Tang S, Yang D, Shi W, Wang X, Wang H, et al. The Potential Transmission of SARS-CoV-2 from Patients with Negative RT-PCR Swab Tests to Others: Two Related Clusters of COVID-19 Outbreak. Jpn J Infect Dis. 2020; 73(6): 399-403.
- Mallett S, Allen AJ, Graziadio S, Taylor SA, Sakai NS, Green K, et al. At what times during infection is SARS-CoV-2 detectable and no longer detectable using RT-PCR-based tests? A systematic review of individual participant data. BMC Med. 2020; 18(1): 346.
- Iyer AS, Jones FK, Nodoushani A, Kelly M, Becker M, Slater D, et al. Dynamics and significance of the antibody response to SARS-CoV-2 infection. medRxiv [Preprint]. 2020.
- Korth J, Wilde B, Dolff S, Anastasiou OE, Krawczyk A, Jahn M, et al. SARS-CoV-2-specific antibody detection in healthcare workers in Germany with direct contact to COVID-19 patients. J Clin Virol. 2020; 128: 104437.
- Alteri C, Cento V, Antonello M, Colagrossi L, Merli M, Ughi N, et al. Detection and quantification of SARS-CoV-2 by droplet digital PCR in real-time PCR negative nasopharyngeal swabs from suspected COVID-19 patients. PLoS One. 2020; 15(9): e0236311.
- Li Z, Li Y, Chen L, Li S, Yu L, Zhu A, et al. A Confirmed Case of SARS-CoV-2 Pneumonia with Routine RT-PCR Negative and Virus Variation in Guangzhou, China. Clin Infect Dis. 2020; 9: ciaa941.
- Alonso FOM, Sabino BD, Guimarães MAAM, Varella RB. (2020). Recurrence of COVID-19 with SARS-CoV-2 RNA reversion to positive and late antibody response: A case report from Brazil. J Med Virol. 2021; 93(2): 655-656.
- Coronavirus panel of the state of Rio de Janeiro [accessed in 15 January]. Available at: http://painel.saude.rj.gov.br/ monitoramento/covid19.html#.
