Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Acute myeloid leukemia presenting with multiple breast and skin myeloid sarcomas: A case report
Yumna Khatib1; Heba Farfoura1; Hussam Haddad2 ; Lina Mrie3; Kamal Al-Rabie3; Mohammad Ma’koseh3*
1Department of Radiology, King Hussein Cancer Center, Amman, Jordan.
2Department of Pathology, King Hussein Cancer Center, Amman, Jordan.
3 Department of Medical Oncology, King Hussein Cancer Center, Amman, Jordan.
*Corresponding Author : Mohammad Ma’koseh
King Hussein Cancer Center, Department of Medical
Oncology, Queen Rania Al-Abdulla Street, Amman
11941, Jordan.
Email: Maakoseh727@yahoo.com
Received : Feb 25, 2021
Accepted : Apr 07, 2021
Published : Apr 09, 2021
Archived : www.jcimcr.org
Copyright : © Ma’koseh M (2021).
Abstract
We report a case of 63-year old female who presented to breast imaging unit with multiple bilateral breast masses. Her past history was significant for left thigh mass. Mammogram and breast ultrasound revealed multiple bilateral breast masses. Ultrasound guided true-cut biopsy from right breast mass was consistent with myeloid sarcoma. Bone marrow biopsy showed acute myeloid leukemia (FAB-M5B) evolving on the background of Chronic Myelomonocytic Leukemia (CMML).
Keywords: Myeloid Sarcoma; Breast; Chronic Myelomonocytic Leukemia.
Citation: Ma’koseh M, Khatib Y, Farfoura H, Haddad H, Mrie L, et al. Acute myeloid leukemia presenting with multiple breast and skin myeloid sarcomas: A case report. J Clin Images Med Case Rep. 2021; 2(2): 1045.
Introduction
Myeloid sarcoma (MS), also known as granulocytic sarcoma or chloroma is a rare manifestation that is characterized by of 1 or more tumor myeloid masses occurring at an extra medullary site [1]. In the most recent World Health Organization classification of myeloid neoplasms and acute leukemia, myeloid sarcoma was classified as independent subtype of Acute Myeloid Leukemia (AML) [2].
Breast MS rarely presents as breast mass with or without bone marrow involvement before or with the initial diagnosis of AML. More commonly, it presents in patients with previous diagnosis of AML, myelodysplastic or myeloproliferative neoplasms [3-6].
The diagnosis may be challenging, as breast imaging lacks specificity and few reports focusing on radiographic description have been published so far [7-9]. Pathologically, It may present with infiltrative pattern similar to invasive lobular carcinoma or lymphoma [9,10] and the diagnosis is usually confirmed with immunohistochemistry [11,12].
Myeloid sarcoma is relatively more common in patients with AML with prominent monocytic differentiation as well as in patients with Chronic Myelomonocytic Leukemia (CMML) [13,14].
Here we report a case who presented with breast mass and was found to have bilateral multicenteric breast MS. Further evaluation showed evidence of CMML with transformation to AML.
Case report
The patient is a 63-year old female who presented to early detection clinic as she felt a mass in the right breast for one month. On the same time, she could palpate a mass in the left thigh. Initially, her primary care physician evaluated the left thigh mass, and she was re-assured and considered to have a lipoma. There was no nipple discharge or pain.
On examination, two palpable right breast masses each measuring about 3 X 4 cm, left thigh mass measuring about 2 X 2 cm and multiple skin nodules on the back (Figure 1).
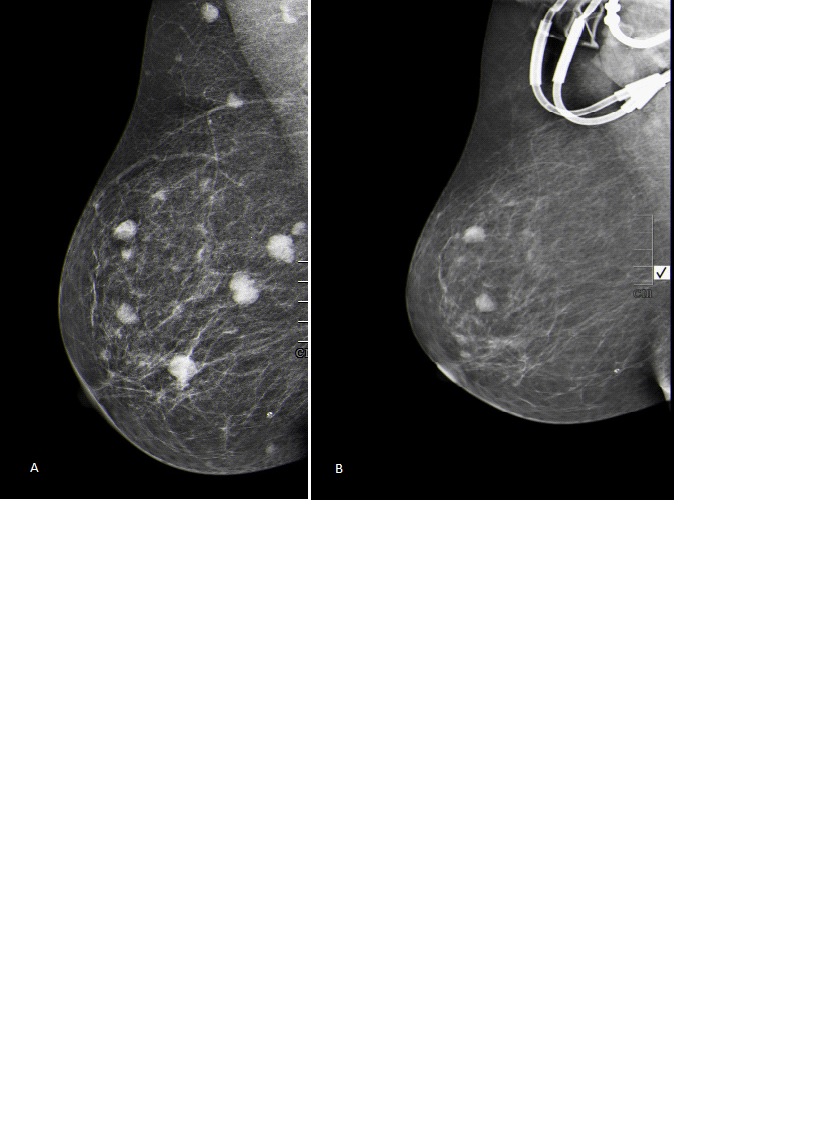
Breast ultrasound showed multiple heterogeneous hypoechoic masses the largest one was located in the right breast and was measuring 10 X 11 X 12 mm. Breast mammogram was done and revealed multiple bilateral round dense masses in both breasts. There was no evidence of clustered micro-calcifications (Figure 2).
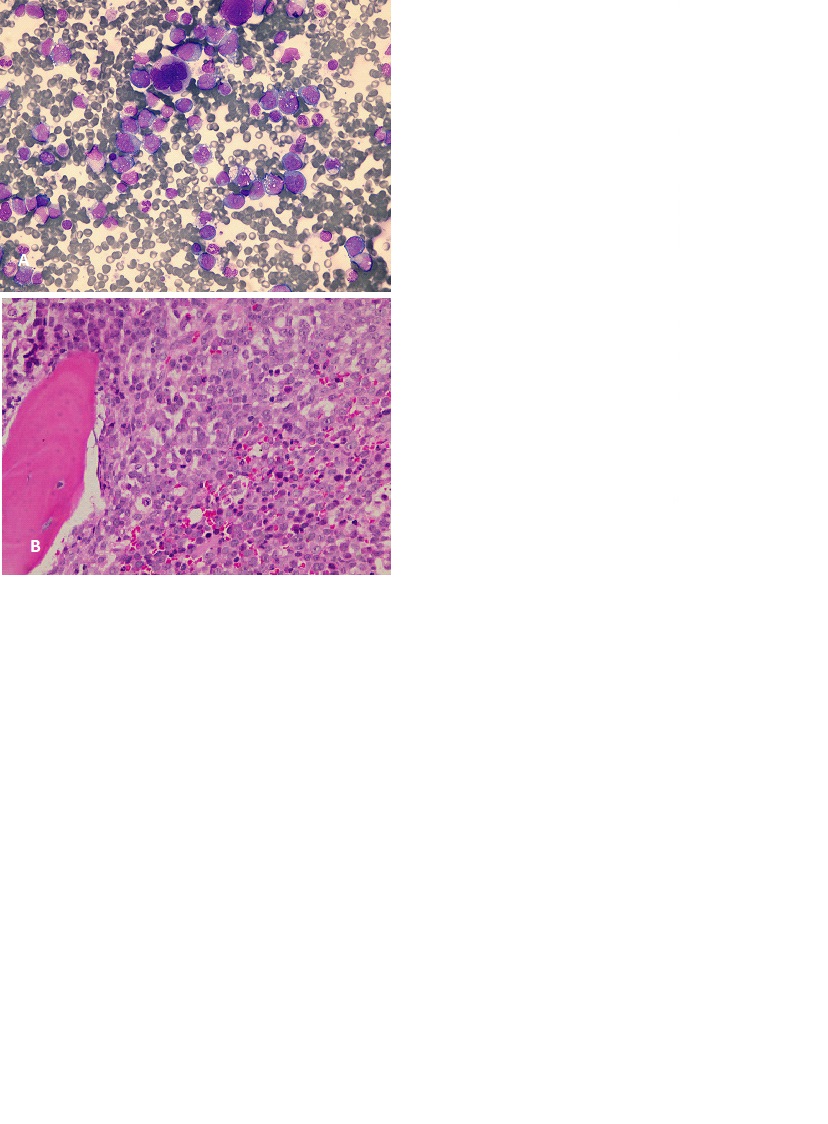
Complete blood count was significant for white blood cell count of 33 X 109 /L with 34.7% monocytes. Peripheral blood smear showed neutrophilic leukocytosis, monocytosis and few circulating blasts. Right breast true-cut biopsy showed tumor cells which were positive for CD43, LCAm[focal], CD68 and lysozyme (Figure 3). They were negative for CD3, CD20, CD79a, CD30, Tdt, CD34, MPO, CD117, S100, GATA-3, Synaptophysin, CK7, CK-MNF and E-cadherin. These findings were consistent with myeloid sarcoma, monocytic type.
Bone marrow aspirate and biopsy showed that 30% of cells were large in size, had large nuclei, prominent nucleoli, and abundant finely granular vacuolated cytoplasm. There were large number of megakaryocytes with dysplastic changes (Figure 4). Lysozyme was strongly positive in neoplastic cells. CD34 and CD61 immunostains were positive in scattered cells. Flowcytometry was done on bone marrow aspirate, and showed that 32% of cells were expressing CD117, CD64, CD4, CD11b, CD11c, CD15, HLADR and MPO consistent with acute monocytic leukemia.
Bone marrow cytogenetics were normal. Fluorescent in situ hybridization (FISH) was negative for Mixed Lineage Leukemia (MLL) gene re-arrangement. Whole body Positron Emission Tomography/Computed Tomography (PET-CT) scan showed increased uptake in the bone marrow, both breast as well as multiple subcutaneous soft tissue nodules all over the body, most prominent in both thighs.
Cerebrospinal fluid cytology showed no malignant cells. The patient was started on systemic chemotherapy combination of idarubicin and cytarabine (5+2 protocol).
21 days after starting the chemotherapy, follow-up bone marrow biopsy showed residual 15% blasts, with dysplastic changes in granulocytes. Bilateral breast mammogram was repeated and showed that bilateral breast masses almost disappeared and were significantly smaller (Figure 2). CT scan of the chest, abdomen and pelvis showed marked improvement in the subcutaneous and bilateral breast nodules. The patient was admitted for re-induction, but unfortunately she developed severe pneumonia and sepsis and she died.
Discussion
Myeloid sarcomas are rare extra-medullary tumors composed of malignant myeloid precursors. They may affect 2.5-9% of patients with AML and may involve any part of the human body; most common sites are skin, lymph nodes, soft tissues, bones, the CNS, and the urogenital tract [1,3,15]. Breast involvement is rare, occurring in around 8% of MS cases [3]. In two recent reviews, breast involvement in AML most commonly occurred after therapy or stem cell transplant [about 50% of cases], while 21% were diagnosed on presentation [16,17]. Interestingly, 90% were less than 50 years old [16]. In patients diagnosed on presentation of AML, involvement of other extra medullary sites was reported in 8 of 23 cases [16].
Although skin involvement is common in MS, rapidly progressing multiple skin nodules seen in our patient is rare [18]. Clinical findings mostly include painless palpable mass. Bilateral involvement was reported in 26.6% of cases while 41% had multiple lesions [17]. Radiological findings are often non-specific. Mammographic findings usually include hyper dense, non-calcified intra-mammary masses with hazy border [7,17]. On ultrasound, lesions are mostly hypo echoic with microlobulations or speculated margins. Sometimes it can show prominent vascularity on color doppler [8,15]. Final diagnosis is usually made with histological examination. Immunohistochemistry shows CD68-KP1 as the most commonly expressed marker, then myeloperoxidase, CD117, CD99, CD68/PG-M1, lysozyme, CD34, terminal deoxynucleotidyl transferase, CD56, CD61, CD30, glycophorin A, and CD4 [14]. The most common cytogenetic and biochemical abnormalities include t [8,21], inv [16], and cytoplasmic expression of NPM and FLT3-ITD [11,14].
The prognostic significance of patients developing MS in the context of AML is debatable. Historically, they were associated with worse prognosis and short survival [11], but recent studies reported that presence of MS was not associated with worse outcome, with 5-years survival rates of 20-30%, which is not much different from AML in general [19,20]. Other factors like age, cytogenetic and molecular profile play more important role in determining the prognosis. Furthermore, prognostic significance of MS in the presence of other poor risk features is not fully understood [20].
Generally, the treatment of AML on top of CMML presenting with MS includes the use of standard induction chemotherapy with consideration of consolidation allogeneic stem cell transplant. Radiotherapy can be considered if the response to chemotherapy is not complete and on relapse for rapid symptom relief, but shouldn’t be used as sole treatment even in patients with isolated MS as it didn’t delay transformation to AML [14,21]. A regimen of 24Gy in 12 fractions was found to effective and not toxic [22].
Conclusion
In conclusion, MS of breast is uncommon presentation of AML. Diagnosis may be challenging, but early recognition and initiation of treatment is important. Treatment includes systemic chemotherapy and consolidation according to the standard risk stratification of AML.
References
- Campidelli C, Agostinelli C, Stitson R, Pileri SA. Myeloid sarcoma: extramedullary manifestation of myeloid disorders. Am J Clin Pathol. 2009; 132: 426-37.
- Daniel A. Arber, Attilio Orazi, Robert Hasser jian et al, The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016; 127: n2391- 405.
- Liu PI, Ishimaru T, McGregor DH, Okada H, Steer A. Autopsy study of granulocytic sarcoma (chloroma) in patients with myelogenous leukemia, Hiroshima-Nagasaki 1949–1969. Cancer. 1973; 31: 948–55.
- Yilmaz AF, Saydam G, Sahin F, Baran Y. Granulocytic sarcoma: A systematic review. Am J Blood Res. 2013.
- Vela-Chávez TA, Arrecillas-Zamora MD, Quintero-Cuadra LY, Fend F. Granulocytic sarcoma of the breast without development of bone marrow involvement: a case report. Diagn Pathol. 2009; 4: 2.
- Thachil J, Richards RM, Copeland G. Granulocytic sarcoma – a rare presentation of a breast lump. Ann R Coll Surg Engl. 2007; 89: W7–9.
- Kinoshita T, Yokokawa M, Yashiro N. Multicentric granulocytic sarcoma of the breast: Mammographic, sonographic and MR findings. Clin Imaging; 2006; 30: 271–4.
- Ahrar K, McLeary MS, Young LW, Masotto M, Rouse GA. Granulocytic sarcoma [chloroma] of the breast in an adolescent patient: ultrasonographic findings. J Ultrasound Med. 1998; 17: 383–4.
- Furebring-Freden M, Martinsson U, Sundstrom C. Myelosarcoma without acute leukemia: Immunohistochemical and clinicopathologic characterization of eight cases. Histopathology. 1990; 16: 243–250.
- Rosen PP. Rosen’s Breast Pathology. Rosen’s breast pathology. 3 rd edition. Wolters Kluwer/Lippincott Williams & Wilkins; 2008; 1116.
- Avni B, Koren-Michowitz M. Myeloid sarcoma: current approach and therapeutic options. Ther Adv Hematol. 2011; 2: 309–16.
- Traweek ST, Arber DA, Rappaport H, Bryrnes RK. Extramedullary myeloid cell tumors: An immune histochemical and morphologic study of 28 cases. Am J Surg Pathol. 1993; 17: 1011–1019.
- Elenitoba-Johnson K, Hodges GF, King TC, Wu CD, Medeiros LJ. extramedullary myeloid cell tumors arising in the setting of chronic myelomonocytic leukemia a report of two cases. Arch Pathol Lab Med. 1996; 120: 62–67.
- Magdy M, Karim NA, Eldessouki I, Gaber O, Rahouma M, Ghareeb M. Myeloid sarcoma. Oncology Research and Treatment. 2019; 42: 219-24.
- Fernandes Vieira V, Vo QD, Bouquet de la Jolinière J, Khomsi F, Feki A, Hoogewoud HM , Granulocytic Sarcoma Presenting as a Palpable Breast Lump. Front Surg. 2017; 23: 67.
- Cunningham I. A clinical review of breast involvement in acute leukemia. Leuk Lymphoma. 2006; 47: 2517–26.
- Surov A, Wienke A, Abbas J. Breast leukemia: An update. Acta Radiol. 2012; 53: 261–6.
- Nakagawa S, Tagami H, Ichinohasama R, Aiba S. Rapidly growing cobblestone-like nodules as a manifestation of myeloid sarcoma. Acta Derm Venereol. 2008.
- Pileri SA, Ascani S, Cox M, Campidelli C, Bacci F, Piccioli M et al. Myeloid sarcoma: Clinicopathologic, phenotypic and cytogenetic analysis of 92 adult patients. Leukemia. 2007; 21: 340-350.
- Bakst RL, Tallman MS, Douer D, Yahalom J, et al. How I treat extramedullary acute myeloid leukemia. Blood. 2011; 118: 3785- 93.
- He J, Ye X, Li L, Zhu J, Zhang J, Xie W, et al. Clinical characteristics and prognosis of nonleukemic myeloid sarcoma. The American Journal of the Medical Sciences. 2014; 347: 434-8.
- Bakst R, Wolden S, Yahalom J. Radiation therapy for chloroma (granulocytic sarcoma). International Journal of Radiation Oncology* Biology* Physics. 2012; 82: 1816-22.