Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
A life-threatening presentation of primary plasma cell leukemia
Roma S Fourmanov1*; Annemiek Joosen2; Lidwine Tick3; Heleen S de Lil3
1 Resident Internal Medicine/Hematology, Máxima Medical Center in Veldhoven/Eindhoven, The Netherlands.
2 Clinical Chemist, Máxima Medical Center in Veldhoven/Eindhoven, The Netherlands.
3 Hematologist Máxima Medical Center in Veldhoven/Eindhoven, The Netherlands
*Corresponding Author : Roma S Fourmanov
Resident Internal Medicine/Hematology, Máxima
Medical Center in Veldhoven/Eindhoven, The
Netherlands.
Email: roma.fourmanov@mmc.nl
Received : Mar 31, 2021
Accepted : Apr 28, 2021
Published : Apr 30, 2021
Archived : www.jcimcr.org
Copyright : © Fourmanov RS (2021).
Abstract
Background: Multiple myeloma is a relatively common type of plasma cell dyscrasia, in which monoclonal plasma cells proliferate. This frequently leads to anemia, renal failure, hypercalcemia and bone lesions. Primary plasma cell leukemia is a much rarer type of plasma cell dyscrasia, with measurable plasma cells in the blood circulation and usually more acute presenting signs.
Case: A 57-year-old woman presented to the emergency department with dyspnea. Because of hypoxemia due to a hemoglobin concentration of 3.1 g/dL (1,9 mmol/L), asystole occurred, and cardiopulmonary resuscitation had to be started. The severe anemia turned out to be due to a primary plasma cell leukemia. Palliative treatment was started with combination chemotherapy with VTD (bortezomib, thalidomide and dexamethasone) with a very good partial response, after which she proceeded to an autologous stem cell transplantation with high dose melphalan conditioning.
Conclusion: Primary plasma cell leukemia is a plasma cell dyscrasia with both resemblances and differences from the better-known multiple myeloma. It is less common, but presenting signs often are more acute and more severe. Currently there is no curative treatment.
Keywords: Plasma cell leukemia; Hematological emergency; Multiple myeloma; VTD.
Citation: Fourmanov RS, Joosen A, Tick L, Lil HSD. A life-threatening presentation of primary plasma cell leukemia. J Clin Images Med Case Rep. 2021; 2(2): 1094.
Introduction
In plasma cell dyscrasia proliferation of monoclonal plasma cells occurs and often leads to organ damage, directly or through the production of monoclonal proteins. Primary plasma cell leukemia is a rare type of plasma cell dyscrasia, with measurable plasma cells in the blood circulation and typically an aggressive clinical course. The more prevalent multiple myeloma is a much better-known entity with characteristic renal failure, hypercalcemia, anemia and bone lesions, also known as the ‘CRAB-criteria’. These symptoms can also occur in plasma cell leukemia; however, presentation is often more acute. We present a case of plasma cell leukemia in which the patient had very severe anemia without renal insufficiency, hypercalcemia or bone lesions.
Case report
A 57-year-old woman was taken to the emergency department by ambulance. Because of an abnormal ECG there was suspicion of an acute coronary syndrome. She reported shortness of breath since a couple of weeks. Her medical history included an ischemic stroke and alcohol abuse. Physical examination revealed an ill, tachypnoeic and pale patient with cold extremities. Laboratory results showed macrocytic anemia with a hemoglobin concentration of 3.1 g/dL (1.9 mmol/L) and a severe metabolic lactic acidosis. Even before red cell transfusion could be started, asystole occurred most likely due to hypoxemia secondary to the severe anemia. Cardiopulmonary resuscitation was started and red cells were transfused. This led to return of spontaneous circulation, after which she was admitted to the intensive care unit.
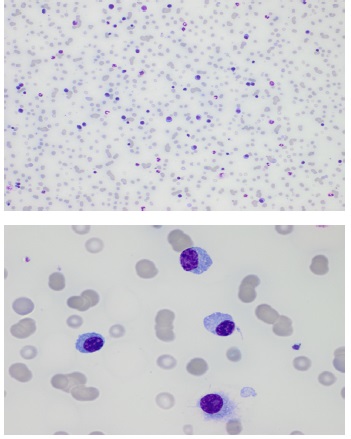
Blood results revealed thrombocytopenia, leukocytosis and erythroblastosis. In addition, there was low haptoglobin with low reticulocyte count, at first attributed to folic acid deficiency in combination with possible Coombs negative hemolysis. The white blood cell differential count revealed an impressive percentage of 53% plasma cells (Figure 1). Furthermore, an IgA lambda monoclonal protein of 51,2 g/L was found. An overview of blood results is shown in Table 1.
Table 1: Most relevant blood results at presentation.
|
Patient |
Reference range |
Unit |
CRP |
4.0 |
<5 |
mg/L |
Leukocytes |
31.3 |
4.0 – 10.0 |
x 109/L |
Neutrophils % |
25 |
40 - 75 |
% |
Lymfocytes % |
20 |
20 - 45 |
% |
Monocytes % |
2 |
2 - 10 |
% |
Eosinophils % |
0 |
1 - 5 |
% |
Plasmacells % |
53 |
<1 |
% |
Neutrophils # |
7.8 |
1.5 – 7.5 |
x 109/L |
Lymfocytes # |
6.3 |
1 – 3.5 |
x 109/L |
Monocytes # |
0.6 |
<1 |
x 109/L |
Erytroblasts # |
5 |
0 |
/100 leukocytes |
Commentary on differential count |
Rouleaux formation, hypogranular platelets, a single blast |
|
|
Platelet count |
52 |
150 – 400 |
x 109/L |
Hemoglobin |
3.1 |
12.1 – 16.1 |
g/dL |
MCV |
136 |
80 - 100 |
fL |
Reticulocytes |
45 |
25 - 120 |
x 109/L |
LDH |
510 |
<247 |
U/L |
Haptoglobin |
<0.20 |
0.37 – 2.21 |
g/L |
Folic acid |
4.0 |
>8.0 |
nmol/L |
Vitamin B12 |
570 |
145 - 569 |
pmol/L |
Creatinin |
149 |
49 - 90 |
µmol/L |
Ionized calcium |
1.00 |
1.15 – 1.30 |
mmol/L |
M-protein |
51.2 |
Not present |
g/L |
Free light chain kappa |
4.57 |
6.7 – 22.4 |
mg/L |
Free light chain lambda |
398 |
8.3 - 27.0 |
mg/L |
Free light chain ratio |
0.011 |
0.31 – 1.56 |
|
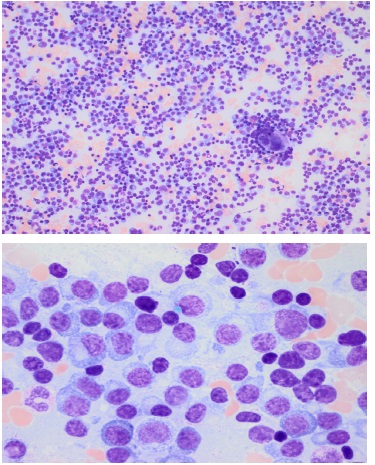
These findings were diagnostic for primary plasma cell leukemia. Renal function was slightly affected but recovered after resuscitation, calcium concentration was normal, and a CT-scan showed no lytic bone lesions. Thoracic and abdominal CT did show hepato-splenomegaly. A bone marrow aspirate was conducted and revealed 70% pathological plasma cells (Figure 2), cytogenetic analysis showed chromosome 1q21 copy number gain and loss of 13q14.
After short admission to the intensive care unit, the patient was transferred to the hematology ward for further recovery. She had not suffered any brain damage.
During admission, the leukocyte count normalized and the percentage of plasma cells in blood decreased to 30%. Possibly the initial leukocytosis was partly caused by the stress response during cardiopulmonary resuscitation, but nevertheless, even after normalization of the leukocyte count, diagnostic criteria for a primary plasma cell leukemia were still met.
Additionally, liver enzymes and coagulation tests were elevated without improvement after administration of vitamin K. APTT normalized after APTT mixing test. Factor II, V, VII and X were low, in line with a liver synthesis disorder. This was explained by post-alcoholic liver cirrhosis, Child-Pugh C, in combination with shock liver after cardio-pulmonary resuscitation; other causes were excluded. Abdominal CT revealed collateral blood vessels around her stomach and distal esophagus, but gastroscopy showed no signs of esophageal varices. The earlier described thrombocytopenia, low haptoglobin concentration and hepato-splenomegaly, which were explained by the plasma cell leukemia, might also have been secondary to liver cirrhosis.
Because of the liver cirrhosis, the patient did not qualify for intensive treatment in the HOVON 129 study, in which treatment is started with induction chemotherapy with carfilzomib and lenalidomide followed by serially autologous and allogeneic stem cell transplants. Inpatient treatment with high-dose dexamethasone was started, followed by outpatient palliative systemic chemotherapy with VTD courses (bortezomib, thalidomide and dexamethasone). After 2 courses a very good partial response had occurred with a decline of the IgA lambda M-protein to <2 g/L. After 4 courses of VTD the patient proceeded to consolidation treatment with an autologous stem cell transplant with high-dose melphalan conditioning. She is currently recovering after the transplant and doing well.
Discussion
Plasma cell leukemia is a rare disorder with resemblances to the much better-known multiple myeloma. Both disorders belong to the group of plasma cell dyscrasia, in which clonal plasma cells proliferate producing a monoclonal paraprotein, involving heavy or light chains [1]. In multiple myeloma this proliferation is limited to plasma cells in the bone marrow, whereas in plasma cell leukemia plasma cells are also located in the peripheral blood. Multiple myeloma can transform into plasma cell leukemia, called secondary plasma cell leukemia, whereas when it originates spontaneously it is called primary plasma cell leukemia [2]. The incidence of plasma cell leukemia in Europe is around four cases in 10 million inhabitants per year, whereas the incidence of multiple myeloma is seven cases in 100.000 inhabitants per year [3].
Clinical presentation
A patient with multiple myeloma typically presents with ‘CRAB’ criteria: HyperCalcemia, Renal failure, Anemia and Bone lesions. These are also the presenting symptoms in plasma cell leukemia, although lytic bone lesions and hypercalcemia occur less often. Moreover, the concentration of Lactate DeHydrogenase (LDH) is often high at presentation, and hepatosplenomegaly is quite common [4]. Presentation and natural course are often more severe and acute in plasma cell leukemia compared to multiple myeloma. This is clearly illustrated by this case report.
Diagnosis
The diagnosis of plasma cell leukemia can be made if the absolute count of monoclonal plasma cells in peripheral blood exceeds 2 × 109 /L or 20 % [5]. In primary plasma cell leukemia, there is no previous history of multiple myeloma. In secondary plasma cell leukemia, the patient was previously diagnosed with multiple myeloma; this often occurs in the setting of endstage relapsed/refractory disease.
Treatment and prognosis
Primary plasma cell leukemia has a poor prognosis; historically median survival was only eight months [6]. After introduction of bortezomib containing chemotherapy regimens combined with autologous stem cell transplant prognosis improved. This treatment is comparable to the treatment of multiple myeloma. In a recent Greek cohort, overall survival in primary plasma cell leukemia was 18 months, but 48 months in patients who received a bortezomib based therapy and an autologous stem cell transplant [7]. In comparison, the five-year survival rate of patients with multiple myeloma is 55%.
The current EMN12 study, which is taking place in seven European countries including the Netherlands, aims to investigate whether survival can be further improved in patients with minor comorbidities who are treated with a carfilzomib containing regimen followed by first an autologous stem cell transplant and then an allogeneic stem cell transplant [8].
Conclusion
Primary plasma cell leukemia is a rare form of plasma cell dyscrasia with an aggressive natural course. It has many resemblances with the better-known and more common multiple myeloma, though the presentation is usually more acute and the natural course more aggressive. Also, the concentration of LDH is increased more often and hepato-splenomegaly occurs more often. Prognosis has historically been very poor but has improved with implementation of bortezomib-based treatment regimens and autologous stem cell transplant. Currently new chemotherapy regimens, followed by multiple stem cell transplants, are investigated with the aim of improving future treatment outcomes.
References
- Craig FE, Foon KA. Flow cytometric immunophenotyping for hematologic neoplasms. Blood. 2008; 111: 3941.
- Fernández de Larrea C, Kyle RA, Durie BG, Ludwig H, Shah JJ et al. Plasma cell leukemia: consensus statement on diagnostic requirements, response criteria and treatment recommendations by the International Myeloma Working Group. Leukemia. 2013; 27: 780.
- Sant M, Allemani C, Tereanu C, De Angelis R, Capocaccia R et al. Incidence of hematologic malignancies in Europe by morphologic subtype: Results of the HAEMACARE project. Blood. 2010; 116: 3724.
- Tiedemann RE, Gonzalez-Paz N, Kyle RA, Santana-Davila R, PriceTroska T et al. Genetic aberrations and survival in plasma cell leukemia. Leukemia. 2008; 22: 1044.
- McKenna RW, Kyle RA, Kuehl WM, Harris NL, Coupland RW, Fend E. Plasma cell neoplasms. In: World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues, IARC Press, Lyon. 2017; 250.
- Ramsingh G, Mehan P, Luo J, Vij R, Morgensztern D. Primary plasma cell leukemia: A Surveillance, Epidemiology, and End Results database analysis between 1973 and 2004. Cancer. 2009; 115: 5734.
- Katodritou E, Terpos E, Delimpasi S, Kotsopoulou M, Dimopoulos MA et al. Real-world data on prognosis and outcome of primary plasma cell leukemia in the era of novel agents: A multicenter national study by the Greek Myeloma Study Group. Blood Cancer J. 2018; 8: 31.
- Donk van de NW, Sonneveld P, Zweegman S, Vellenga E, Lokhorst HM et al. The first HOVON and European Myeloma Network trial for primary plasma cell leukemia: HOVON 129. Ned Tijdschr Hematol. 2015; 12:190-193.