Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
T2* relaxometry maps of the uterus – Future prospectives with oncology implications
Roxana M Pintican1*; Reka David Kerekes2; Vlad Bura2; Florin Stamatian3; Tunde Kovacs4; Iris Morar5; Viorica Nagy6; Radu Fechete7; Angelica Chiorean8
1 “Iuliu Hatieganu” University of Medicine and Pharmacy, Cluj-Napoca, Romania, Radiology and Medical Imaging, County Emergency Hospital Radiology Department, Cluj-Napoca, Romania.
2 Radiology and Medical Imaging, County Emergency Hospital Radiology Department, Clinicilor Str. 3-5, 400009, Cluj-Napoca, Romania.
3 “Iuliu Hatieganu” University of Medicine and Pharmacy, Cluj-Napoca, Romania, Chief of the Obstetrics and Gynecology Department, County Emergency Hospital, Clinicilor Str. 3-5, 400009 ,Cluj-Napoca, Romania.
4 Obstetrics and Gynecology Department at IMOGEN Research Center, County Emergency Hospital, L. Pasteur Str., 400349, Cluj-Napoca, Romania.
5 Physicist, IMOGEN Research Center, County Emergency Hospital, L. Pasteur Str., 400349, Cluj-Napoca, Romania.
6 “Iuliu Hatieganu” University of Medicine and Pharmacy, Cluj-Napoca, Romania; “Ion Chiricuta” Oncology Institute, Republicii Str 34-36, 400015, Cluj-Napoca, Romania.
7 professor at Technic University, Physics and Chemistry Department, G Baritiu Str. 25, 400027, Cluj-Napoca, Romania.
8 Iuliu Hatieganu” University of Medicine and Pharmacy, Department of Radiology, Cluj-Napoca, Romania.
*Corresponding Author : Roxana M Pintican
“Iuliu Hatieganu” University of Medicine and
Pharmacy, Department of Radiology, Cluj-Napoca,
Romania; Radiology and Medical Imaging, County
Emergency Hospital Radiology Department, Clinicilor
Str. 3-5, 400009 ,Cluj-Napoca, Romania.
Email: roxana.pintican@gmail.com
Received : Mar 22, 2021
Accepted : Apr 29, 2021
Published : May 04, 2021
Archived : www.jcimcr.org
Copyright : © Pintican RM (2021).
Abstract
T2* relaxometry mapping has found diverse applications in neuroradiology but was insufficiently evaluated in pelvis MRI. Further, staging of the uterine-related cancers may be sometimes a cumbersome.
Our aim is to present the T2* relaxometry maps respectively relaxometry and proton density values in patients with endometrial and cervical cancer, together with their postoperative outcomes.
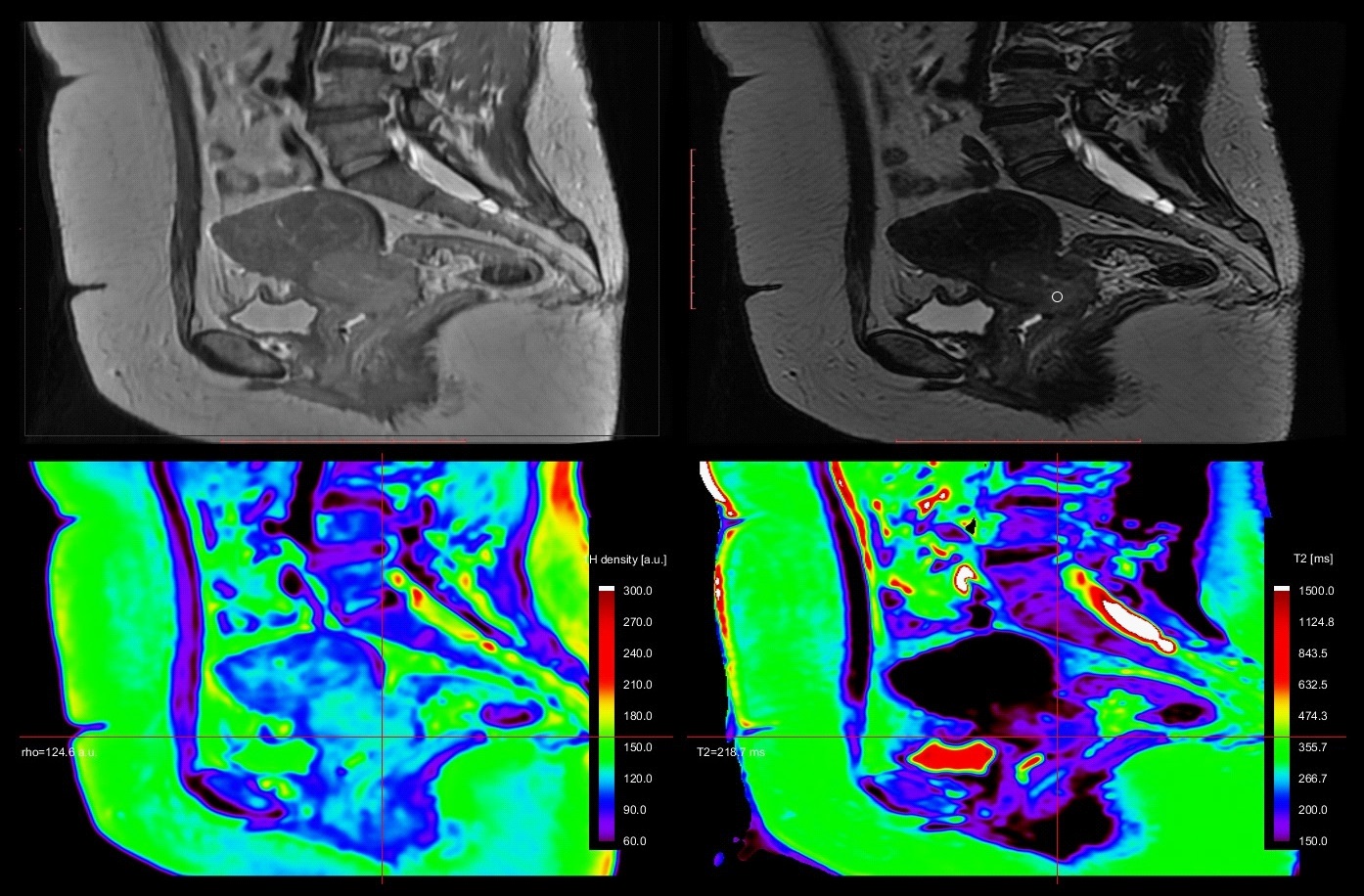
Three patients with uterine-related cancers underwent a 3T pelvic MRI examination. The protocol included a large field-of-view (FOV = 28 cm), a short (TE=30 ms) and a long (TE = 200 ms) echo time sequences. T2* maps were obtained by the means of post-processing methods.
Parametric maps showed same Relaxation Time (RT) between endometrial cancer and adjacent metastatic lymphadenopathy, respectively different RT between endometrial and cervical cancer.
In conclusion, we presented the T2* relaxometry values of endometrial and cervical cancer, and discuss their possible implications in FIGO staging and patient management.
Keywords: T2 relaxometry; T2 mapping; MRI; Endometrial cancer; Cervical cancer; FIGO staging.
Citation: Pintican RM, Kerekes RD, Bura V, Stamatian F, Kovacs T, et al. T2* relaxometry maps of the uterus – Future prospectives with oncology implications. J Clin Images Med Case Rep. 2021; 2(3): 1102.
Introduction
In the era of multiparametric Magnetic Resonance Imaging (MRI), the T2 relaxometry is gaining more and more credibility and usefulness. T2 parametric maps found varied application in neuroradiology, being able to improve detection of epileptogenic zones in hippocampus, as a neuroimaging marker in Friedreich’s ataxia and even in infectious diseases such as cysticercosis [1-3].
Cardiac and musculoskeletal imaging likewise, proved to be enriched by using T2 parametric maps, in the characterization of acute myocardial infarction or cartilage damage after ACL reconstruction [4,5].
Regarding the branch of women imaging, the relaxometry maps are promising in both lesion detection and characterization. Studies concluded that T2 relaxation time was significantly lower in invasive breast cancers than DCIS (ductal carcinoma in situ) and more than that, patients with invasive type had lower T2 time in higher histologic grade [6]. Furthermore, T2 maps revealed shorter time for responders versus non-responders after neoadjuvant chemotherapy, thus being useful in appreciating tumor apoptosis [7]. A recent paper, highlighted a new method of MRI fingerprinting technique, which allows breast lesions diagnostic using volumetric quantification of T1 and T2 relaxation time [8].
T2 relaxometry studies focused on the pelvis area, are still at the beginning. Imaoka et al have previously demonstrated the layered uterine structure and Ghosh et al emphasized the use of the T2 maps in diagnostic of endometrial cancer and adenomyosis, with a protocol that included multiple TE (echo time) per patient /acquisition [9,10].
To the best of our knowledge, there are no MRI relaxometry studies on cervical cancer, or related to endometrial or cervical cancer staging. Furthermore, MRI was recently included as a mandatory pre-operative diagnostic tool in patients with endometrial cancer, allowing patient stratification and guiding the surgical planning [11].
Subsequently, our aim is to evaluate oncologic patients using a T2 relaxometry map of the whole pelvis, obtained with only two different TE’s. Related to future prospective with oncology implication, we will exhibit the T2 relaxometry map of cancer area, lymph nodes and nearby structures with pathology correlation.
Case series
Three oncologic patients (two with endometrial cancer and one with cervical cancer) underwent a 3T MRI (Discovery MR750w, GE) examination using a body coil transmission and a 16-channel phased-array receiver. The MRI protocol included 2 different TE (TE1 =30 ms and TE2 =200 ms), with a long repetition time (TR=5000 ms) and a large field-of-view (FOV=28 cm). We decided on using only 2 different TE’s, due to the short examination time, and also because it minimize the motion artifacts.
An in-house dedicated software written in ProcessingTM was used to calculate the T2 relaxation time for each manually defined areas of interest. T2 relaxometry and proton density maps of the entire pelvis were obtained.
We analyzed the pathologic endometrium and cervix, pelvic lymphadenopathies, and a section of small bowel and rectum that were related to the cancer area. In concordance with international oncology recommendations, FIGO staging system was used for staging endometrial and cervical cancer patients [12,13].
Case 1
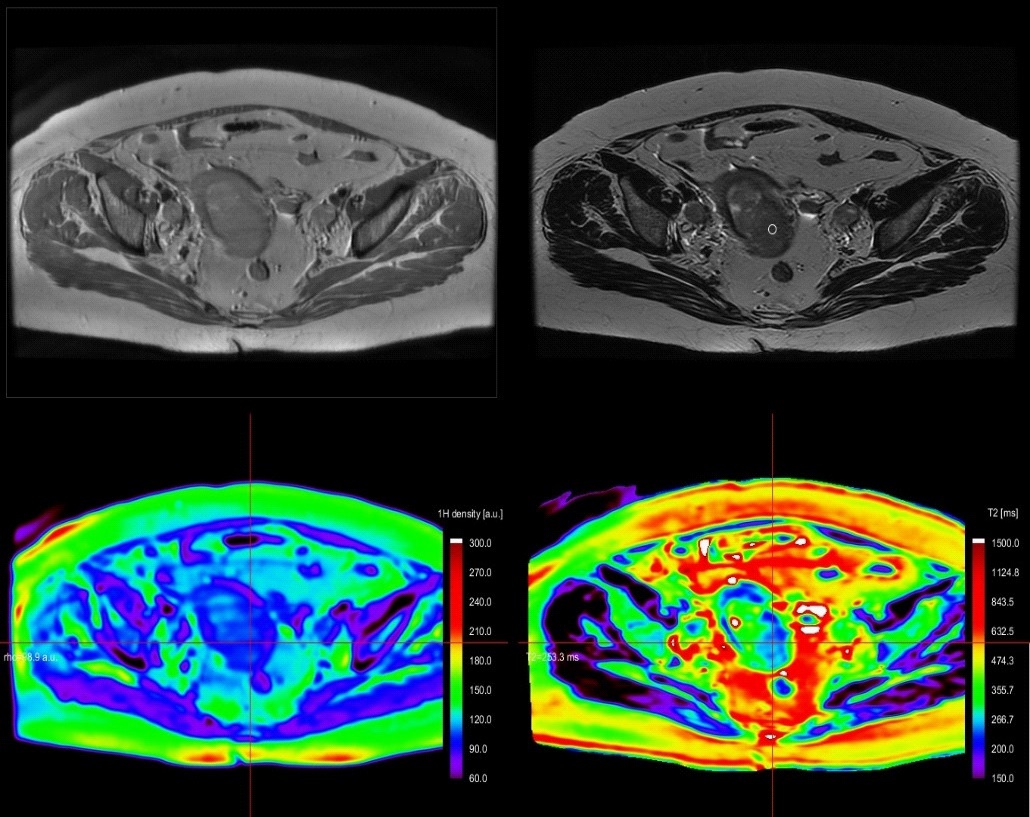
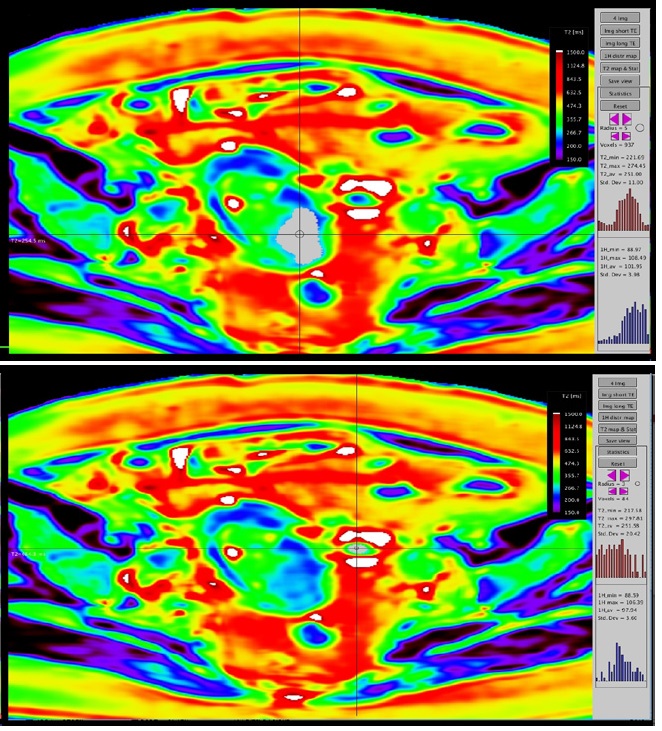
A 67y old patient, with endometrial curettage positive for cancer. While bilateral iliac lymph nodes have suspicious T2 characteristics (round shape, inhomogeneous), the left parauterine lymph node has uncertain features (oval, homogeneous) on both T2 and DWI (no restricted diffusion). On T2* relaxometry map, the lymph node exhibits similar relaxation time as the endometrial cancer. The postoperative pathology revealed positive endometrial cancer cells, grade 2 within all 3 lymph nodes and a stage IIIC1 was attributed to the patient (Figure 1 & 2).
Case 2
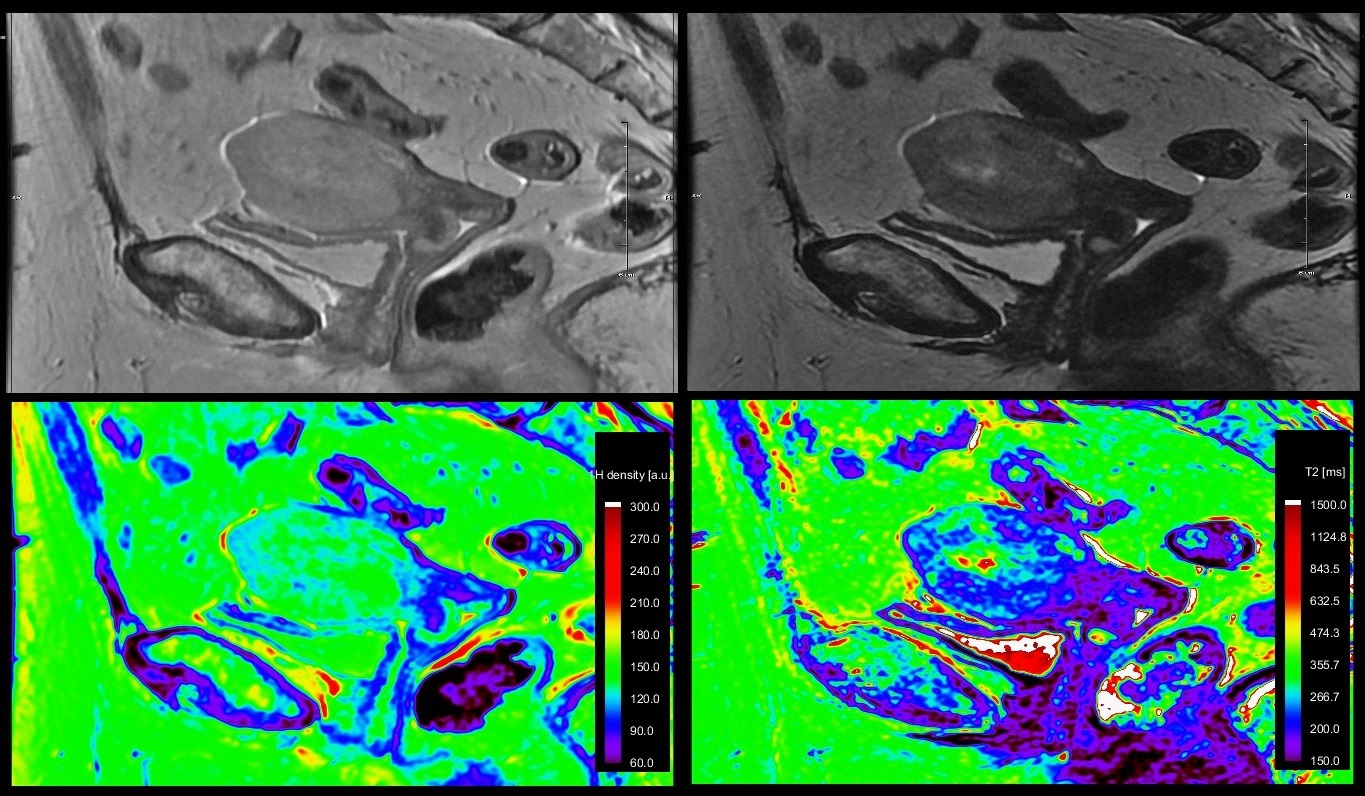
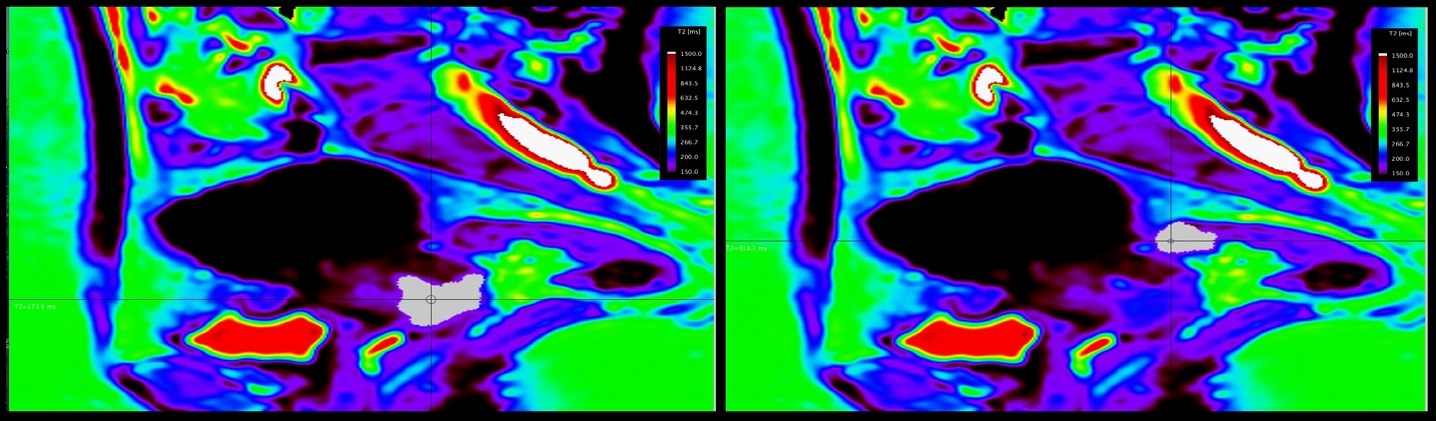
A 71y old patient with known endometrial cancer. On MRI images, we easily highlighted the endometrial cancer invading the myometrium, but we were not able to exclude an extension into uterine serosa or into a segment of small bowel loop situated nearby. A cleavage plan between the structures could not be revealed on standard T1, T2 sequences and a possible stage IV disease was suggested. The T2* maps showed different relaxation time for endometrial cancer and small bowel. Intraoperative examination of the segment suggested tumor-free small bowel with no further resection needed; a FIGO stage II was attributed (Figure 3 & 4).
Case 3
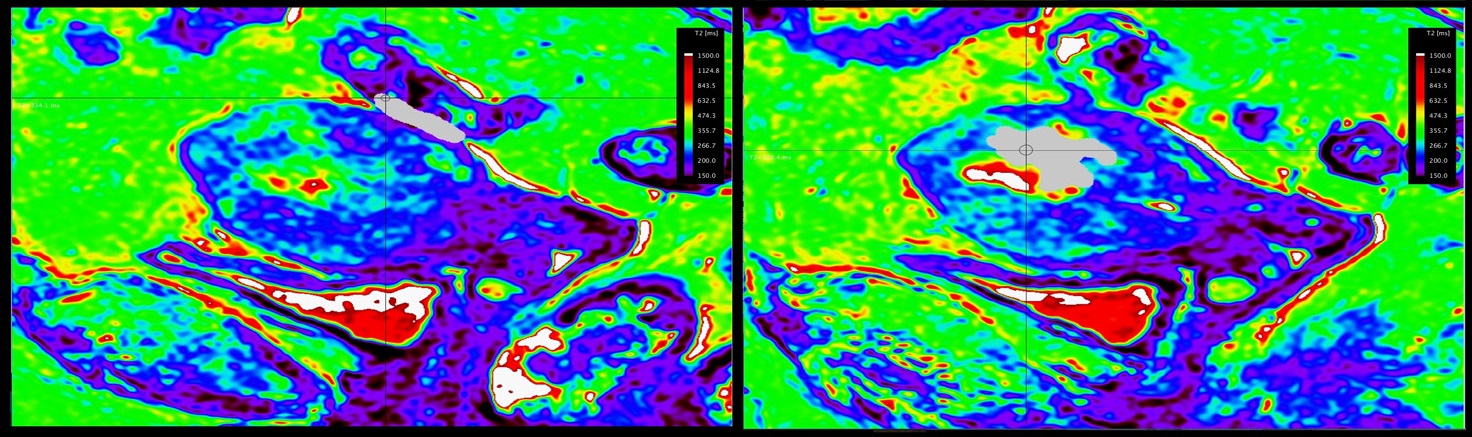
A 51y old patient with proven cervical cancer. Pelvic MRI was performed for staging purposes. The cervical cancer invades the upper two-third of vagina and has bilateral parametrial involvement. However, the rectum invasion could not be excluded on standard MRI sequences, so the stage IV could not be excluded. On T2* relaxation maps, cervical cancer and the rectum located adjacent to the tumor presented with different value of relaxation times. Intraoperative examination of the rectum excluded a tumoral invasion, and pathology concluded a stage IIB cervical cancer (Figure 5 & 6).
Discussions
We obtained T2 relaxometry maps of endometrial and cervical cancer patients, in axial and sagittal planes, with a larger FOV and shorten acquisition time than previously mentioned [9,10]. We demonstrated T2 relaxometry values of endometrial and cervical cancer, metastatic lymphadenopathy, small bowel and rectum (Table 1).
Table 1: Most relevant blood results at presentation.
Area of interest |
T2 min (ms) |
T2 max (ms) |
T2 av (ms) |
Std dev |
1H min |
1H max |
1H av |
std dev |
Endometrial cancer |
212.05 |
581.21 |
334.96 |
53.64 |
104.32 |
153.6 |
127.13 |
7.71 |
Cervix cancer |
138.48 |
239.85 |
196.87 |
15.7 |
107.62 |
130.38 |
123.8 |
3.5 |
Small bowel loop |
112.77 |
307.32 |
180.58 |
29.52 |
65.81 |
163.85 |
105.48 |
15.79 |
Rectum |
139.51 |
187.65 |
160.64 |
10.21 |
97.14 |
157.95 |
114.41 |
9.43 |
In first case, the T2* relaxometry values matched the value of the primary tumour (endometrial cancer). This observation could result in a first future oncology prospective, implying that the role of T2 relaxometry maps in identifying metastatic lymph nodes should be investigated and perhaps it may help the radiologist reach a higher confidence in reporting them. In addition, a recent paper addressed the noninvasive characterization of lymph nodes using a multigradient echo signal decay technique, and obtained promising results [14].
In second and third cases, T2* relaxometry values of cancers were different from that of non-cancerous small bowel and rectum confirmed by the intraoperative examination. A second research direction may be suggested related to parametric maps in patients with doubtful tumor invasion (primary tumor, relapse, adhesions on a postoperative pelvis, or just the lack of the cleavage plan between structures).
Differentiating between endometrial and cervical origin before surgery is critical as it has major implications for patient management. Currently, on MRI, the origin is subjectively established by: 1- the location where the tumour seems to be centered or 2 - the location of the bulk (>50% of the tumour mass) in cases where the tumour extends across the corpus or cervix. Accordingly, we observed different T2* relaxometry values for endometrial and cervical cancer (~300 ms versus 196 ms). A third prospective and future research direction may result from analyzing the current technique on being able to differentiate between endometrial and cervical cancer, based on a cut-off T2* value, especially in patients where biopsy is not possible (eg. cervical stenosis).
Limitations
First, a too small number of patients have prevented us from establishing a cut-off T2* relaxometry value for each pelvic structure (bladder, vagina, etc.) and/or cancer type. Second, DWI/ADC map were inconclusive for the first out of the three cases and contrast-enhanced sequences were not available for any of the cases.
Emphasizing that the small number of patients didn’t allow us to make a final conclusion supported by statistical analysis, the present article proposes a number of future oncology prospective that could lead to at least two innovative research directions: 1) establishing cut-off values that could be further used to early diagnose the endometrial and cervical cancer or establishing tumor’s origin in selected cases, to identify malignant lymphadenopaties and tumor invasion in the nearby organs; 2) comparing T2* maps with the DWI/ADC and/or contrastenhanced sequences regarding lymphadenopathy characterization and tumor-invasion. Further studies on larger cohort of patients are needed in order to conclude on and extrapolate the above mentioned concepts and innovative technique.
References
- Winston GP, Vos SB, Burdett JL et al. Automated T2 relaxometry of the hippocampus for temporal lobe epilepsy. Epilepsia. 2017; 58: 1645-1652.
- Bonilha da Silva C, Bergo FPG, D'Abreu A, et al. Dentate nuclei T2 relaxometry is a reliable neuroimaging marker in Friedreich's ataxia. Eur J Neurol. 2014; 21: 1131-1136.
- De Souza A, Nalini A, Saini J et al. T2 relaxometry helps prognosticate seizure outcome in patients with solitary cerebral cysticercosis. J Neurol Sci. 2017; 15: 376: 1-6.
- Mayr A, Klug G, Feistritzer HJ et al. Myocardial edema in acute myocarditis: Relationship of T2 relaxometry and late enhancement burden by using dual-contrast turbo spin-echo MRI. Int J Cardiovasc Imaging. 2017; 33: 1789-1794.
- Bae JH, Hosseini A, Wang Y et al. Articular cartilage of the knee 3 years after ACL reconstruction. A quantitative T2 relaxometry analysis of 10 knees. Acta Orthop. 2015; 86: 605-10.
- Seo M, Ryu JK, Jahng GH et al. Estimation of T2* Relaxation Time of Breast Cancer: Correlation with Clinical, Imaging and Pathological Features. Korean J Radiol. 2017; 18: 238–248.
- Liu L, Yin B, Geng DY, et al. Changes of T2 Relaxation Time From Neoadjuvant Chemotherapy in Breast Cancer Lesions. Iran J Radiol. 2016; 13: e24014.
- Chen Y, Panda A, Pahwa S et al. Three-dimensional MR Fingerprinting for Quantitative Breast Imaging. Radiology. 2018; 30: 180836.
- Imaoka I, Nakatsuka T, Araki T et al. T2* relaxometry mapping of the uterine zones. Acta Radiol. 2012; 53: 473-437.
- Ghosh A, Singh T, Bagga R et al. T2 relaxometry mapping in demonstrating layered uterine architecture: Parameter optimization and utility in endometrial carcinoma and adenomyosis: A feasibility study. Br J Radiol. 2018; 91: 20170377.
- Nougaret S, Horta M, Sala E, Lakhman Y, Thomassin-Naggara I, et al. Endometrial Cancer MRI staging: Updated Guidelines of the European Society of Urogenital Radiology. Eur Radiol. 2019; 29: 792-805.
- Creasman W. Revised FIGO staging for carcinoma of the endometrium. Int J Gynaecol Obstet. 2009; 105: 109.
- Bhatla N, Berek JS, Fredes MC, et al. Revised FIGO staging for carcinoma of the cervix uteri. International Journal of Gynecology & Obstetrics. 2019; 145: 129–35.
- Santiago I, Santinha J, Ianus A, Galzerano A, Theias R, et al. Susceptibility Perturbation MRI Maps Tumor Infiltration into Mesorectal Lymph Nodes. Cancer Res. 2019; 79: 2435-2444.
- Vargas HA, Akin O, Zheng J, Moskowitz C, Soslow R, et al. The value of MR imaging when the site of uterine cancer origin is uncertain. Radiology. 2011; 258: 785-792.