Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Spinal intradural extra-arachnoid inflammatory myofibroblastic tumor (IMT): Case report and review of the literature
Chuan Cheng1; kun Wang2; Cao Yang2; Xinghuo Wu2
1Computer Management Center, Wuhan Union Hospital, Tongji Medical, College, Huazhong University of Science and Technology, Wuhan, Hubei, China.
2Department of Orthopaedic Surgery, Wuhan Union Hospital, Tongji Medical, College, Huazhong University of Science and Technology, Wuhan, China.
*Corresponding Author : Xinghuo Wu
Department of Orthopaedic Surgery, Wuhan Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, 1277 Jiefang Avenue, Wuhan 430022, Hubei, China.Department of Orthopaedic Surgery, Wuhan Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, 1277 Jiefang Avenue, Wuhan 430022, Hubei, China.
Email: wuxinghuo@163.com
Received : Apr 12, 2021
Accepted : May 10, 2021
Published : May 13, 2021
Archived : www.jcimcr.org
Copyright : © Wu X (2021).
Abstract
In recent years, molecular genetics studies have highlighted the pathogenesis of IMT, more and more studies have been conducted to explore ALK gene. Literaturelly, the expression of ALK gene in IMT is very rare in central nervous system, while is relative common in non-central nervous system. The present case is the first reported patient with spinal intradural extra-arachnoid Inflammatory Myofibroblastic Tumor (IMT) with overexpressed ALK1. Preoperatively, the case was initially diagnosed as spinal lymphoma. ALK abnormalities was determined by immunohistochemistry and Fluorescence in Situ Hybridization (FISH). Spinal IMT has no definite features either clinically or in the imaging manifestations. The disease should be considered when excluding other possible diseases.
Keywords: Inflammatory myofibroblastic tumor; Intradural; ALK; Spine.
Citation: Cheng C, Wang K, Yang C, Wu X. Spinal intradural extra-arachnoid inflammatory myofibroblastic tumor (IMT): Case report and review of the literature. J Clin Images Med Case Rep. 2021; 2(3): 1136.
Introduction
Inflammatory Myofibroblastoma (IMT) is also known as plasma cell granuloma, fibroxanthogranuloma, myofibroblastoma, myxoid hamartoma, inflammatory fibrosarcoma and inflammatory pseudotumor [1]. Under the premise of a large number of clinical and pathological observations, the features of inflammatory pseudotumor were recognized through the collaborative research of immunohistochemistry, flow cytometry and cytogenetics. Inflammatory pseudotumor has been identified as a true tumor, due to the characteristics of local invasive growth, vascular infiltration, local recurrence, and even malignant changes. Thus, it is recommended to be named as inflammatory myofibroblastoma (IMT) by the World Health Organization international histological classification of soft tissue.
IMT can occur in any part of the body at any age. The most common sites of IMT are in the lungs and soft tissue, while IMT in the central nervous system is rare. Spinal IMT has no definite features either clinically or in the imaging manifestations. The disease should be considered when excluding other possible diseases. The present case is the first reported patient with spinal intradural extra-arachnoid Inflammatory Myofibroblastic Tumor (IMT) with overexpressed ALK1.
Clinical presentation
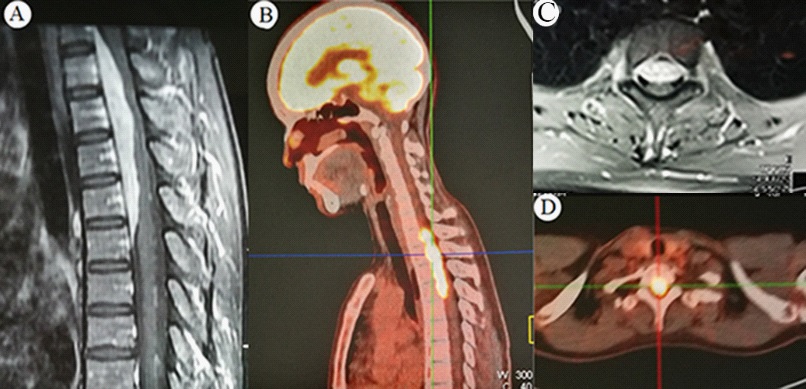
Informed consent was obtained from the patient. A 42-year-old female had a 3-week history of progressive radicular pain in her shoulder and back, add-on abdominal zonesthesia, and distal weakness. Her neurological examination showed no motor and sensory deficits. She also had a history of fever, with a temperature of 38-38.6oC. Biological examination revealed a serous inflammatory reaction (ESR 62 mm/hour;). Serum levels of RF (Rheumatoid Factors) IgA (-) and RF IgG(-) are normal, with an increased level of RF IgG (181.1 RU/ml). T-spot testing for tuberculosis was negative. Magnetic Resonance Imaging (MRI) of the thoracic spine revealed an extradural mass compressing spinal cord at the T-1 to T-5 level with the size of 8.7 X 0.5 cm. Her MRI showed isointense on T1-weighted images, homogeneously hyperintense on intravenous contrast administration, and slightly hyperintense on T2-weighted images Preoperative MRI showed no bony destruction and a normal fatty marrow. PET examination suggested a spinal cord soft tissue lesion (T1-T5) with abnormally increased metabolism, and the disease was diagnosed as a neoplastic lesion (lymphoma?) (Figure 1).
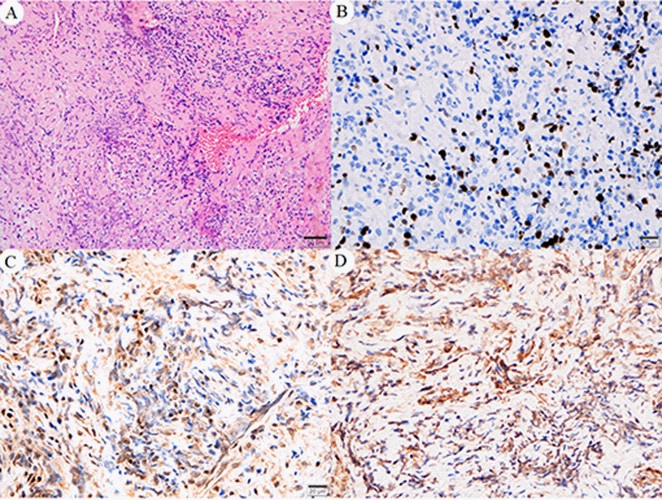
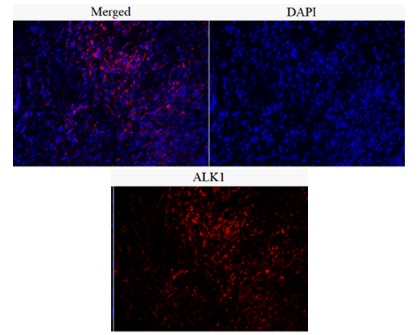
Preoperatively, the mass was initially diagnosed as epidural. However, following a T-1 to T-5 laminectomy, an intradural extra-arachnoid mass was found and removed. Histopathological examination of the extra-arachnoid specimen revealed nodular proliferation of spindle cells with dense infi ltration of lymphoid cells in fi brous tissue (Figure 2). Immunohistochemical examination showed negative for antibodies EMA, S-100, PR, GFAP, PCK, CD34, and SSTR2. The cells were immunopositive for KI67, α-SMA, and anaplastic lymphoma kinase (ALK1). ALK abnormalities was further determined by fluorescence in situ hybridization (FISH) (Figure 3). IgG4 immunostain indicated IgG4: IgG ratio <40%. Finally, the disease was considered as spinal intradural extra-arachnoid Inflammatory Myofibroblastic Tumor (IMT).
Postoperatively, there was a temporary dysfunction of urinary and lower limbs. After initiation of corticosteroid therapy, the patient’s neurological function gradually improved. Indeed, after 6 weeks, she was able to walk alone, and her urinary function returned to normal.
Table 1: Characteristics of reported cases of spinal Infl ammatory myofi broblastic tumor.
Discussion
After IMT was defi ned in the 2002 World Health Organization classifi cation of soft tissue tumors, increasingly cases have been reported in various organs but spinal involvement of IMT is very rare. The Location of spinal IMT exists in epidural, intradural extramedullary, and intramedullary. The present case revealed the involvement of spinal IMT in intradural extra-arachnoid. Literaturelly, 17 cases of intraspinal IMT have been reported [1-17] (Table 1). All but one reported cases occurred in adults aged 22–60 years’ old. Lee et al. reported a case of IMT in a 3-yearold female patient [18]. Of the 17 lesions, two were in the lumbar spine [13,16], the other 15 lesions were in cervical and/or thoracic spine. The locations of lesions in relation to the spinal cord, 6 cases were Extradural (ED), 6 cases were intradural extramedullary (IDEM), 4 cases were Intramedullary (IM), and the other 1 case was ED and IDEM. The present case is the first reported lesion in intradural extra-arachnoid. All patients underwent surgical resection, alone or with steroid therapy, radiotherapy or physiotherapy. In addition, reoperation was reported in only one case [2], with lesions in both ED and IDEM. Eventually, 9 of the patients recovered completely, the other 8 patients didn’t gain fully recovery.
Inflammatory Myofibroblastoma (IMT) has a benign clinical course. Rapid development of modern image diagnosis technologies have made the diagnosis of the intraspinal lesions possible, such as MRI and PET. Enhanced Magnetic Resonance Imaging (MRI) is an effective diagnostic method for spinal lesions, such as extradural lymphoma, metastasis, or myeloma, and so on. MRI signs of these disease are all present as inhomogeneous hyperintensity on T2-weighted and low signal intensity on T1-weighted images, thus it’s difficult to differentiate IMT from the mentioned tumors on the basis of imaging findings. However, there is a slight difference in the MRI imaging of IMT, characterized by no bony destruction and a normal fatty marrow [19,20]. Although IMT has no specific clinical and radiological presentation, but we should keep in mind to differentiate intraspinal mass lesions. However, MRI Characteristics of spinal IMT are not specific to differentiate from other spinal mass, and surgical resection and pathological examination is the best way to achieve a precise diagnosis [20,21]. Pathologically, IMT is mainly composed of myofibroblast spindle cells, often accompanied by plasma cells or lymphocyte infiltration. The diagnosis of the disease depends on histopathological examination, characterized by infi ltration of infl ammatory cells, nodular fasciitis, and/or dense plate-like collagen resembling desmoid [22]. Immunostaining can help confirm the diagnosis by determining myofi broblast marker.
In recent years, molecular genetics studies have highlighted the pathogenesis of IMT, ALK gene fusions have been confirmed in approximately 50% of the disease. Gene fusions are important genomic events in human tumor, supporting the neoplastic nature of IMT [23,24]. The rearrangement and expression of Anaplastic Lymphoma Kinase (ALK) gene and the fusion of ALK gene with rb-2 protein gene have been confirmed in IMT, confirming the nature of neoplastic hyperplasia. The ALK locus mutation (2p23 to 5q35) and overexpression is a typical feature of anaplastic large cell lymphoma. As literature reported, the expression of ALK gene in IMT is very rare in central nervous system, while is relative common in non-central nervous system.
Surgical resection is the recommended effective treatment for IMT, other treatments include steroid therapy, radiotherapy, and chemotherapy. However, chemo- and radiotherapy have not been proven as effective treatments, which should be considered in cases with ALK overexpression [23-25]. According to the literature review, there were 17 reported cases of this disease, but only 5 of them were reported as IMT, and the other 12 were all reported as inflammatory pseudotumor in the early stage.
Conclusion
So far, it’s the fi rst case of IMT in the IDEA space of the thoracic spine, confirmed by modern radiological, histological and immunohistochemical methods. The recommended treatment is surgical excision with steroid - assisted therapy.
References
- Roberts G, Farrell M, Allcutt D. Spinal inflammatory pseudotumours. Br J Neurosurg 2001; 15: 197-8.
- Zemmoura I, Hamlat A, Morandi X. Intradural extramedullary spinal inflammatory myofibroblastic tumor: Case report and literature review. Eur Spine J. 2011; 20: S330-335.
- Eimoto T, Yanaka M, Kurosawa M, Ikeya F. Plasma cell granuloma (inflammatory pseudotumor) of the spinal cord meninges: report of a case. Cancer. 1978; 41: 1929-1936.
- Hsieh PC, Lin CN. Multicentric plasma cell granuloma of spinal cord meninges. Clin Orthop Relat Res. 1995; 188-192.
- Lee M, Epstein FJ, Rezai AR, Zagzag D. Nonneoplastic intramedullary spinal cord lesions mimicking tumors. Neurosurgery. 1998; 43: 788-794.
- Gilliard C, De Coene B, Lahdou JB, Boutsen Y, Noel H, Godfraind C. Cervical epidural pseudotumor and multifocal fibrosclerosis. Case report and review of the literature. J Neurosurg 2000; 93: 152-156.
- Hsiang J, Moorhouse D, Barba D. Multiple plasma cell granulomas of the central nervous system: case report. Neurosurgery. 1994; 35: 744-747.
- Roberts GA, Eldridge PR, Mackenzie JM. Case report: inflammatory pseudotumour of the spine, with literature review. Br J Neurosurg 1997; 11: 570-572.
- Kilinc M, Erturk IO, Uysal H, Birler K, Evrenkaya T, et al. Multiple plasma cell granuloma of the central nervous system: A unique case with brain and spinal cord involvement. Case report and review of literature. Spinal Cord. 2002; 40: 203-206.
- Aizawa T, Sato T, Tanaka Y, Kishimoto K, Watanabe M, et al. Intramedullary plasma cell granuloma in the cervicothoracic spine. Case report. J Neurosurg. 2002; 97: 235-238.
- Lacoste-Collin L, Roux FE, Gomez-Brouchet A, et al. Inflammatory myofibroblastic tumor: A spinal case with aggressive clinical course and ALK overexpression. Case report. J Neurosurg. 2003; 98: 218-221.
- Brandsma D, Jansen GH, Spliet W, Van Nielen K, Taphoorn MJ. The diagnostic difficulties of meningeal and intracerebral plasma cell granulomas--presentation of three cases. J Neurol 2003; 250: 1302-1306.
- Jeon YK, Chang KH, Suh YL, Jung HW, Park SH. Inflammatory myofibroblastic tumor of the central nervous system: clinicopathologic analysis of 10 cases. J Neuropathol Exp Neurol 2005; 64: 254-259.
- Seol HJ, Kim SS, Kim JE, Lee SH, Won JY. Inflammatory pseudotumor in the epidural space of the thoracic spine: a case report and literature review of MR imaging findings. AJNR Am J Neuroradiol 2005; 26: 2667-2670.
- Boutarbouch M, Arkha Y, Rifi L, Derraz S, El Ouahabi A, El Khamlichi A. Intradural cervical inflammatory pseudotumor mimicking epidural hematoma in a pregnant woman: Case report and review of the literature. Surg Neurol. 2008; 69: 302-305.
- Yoon SH, Kim KJ, Chung SK, et al. Inflammatory myofibroblastic tumor in the intradural extramedullary space of the lumbar spine with spondylolisthesis: Case report and review of the literature. Eur Spine J. 2010; 19: S153-157.
- Mankotia DS, Garg K, Nambirajan A, et al. Primary spinal extradural inflammatory myofibroblastic tumor: A rare cause of paraparesis. Neurol India. 2016; 64: 1333-1335.
- Adams RF, Anslow P. The natural history of transdural herniation of the spinal cord: Case report. Neuroradiology. 2001; 43: 383-387.
- Baker LL, Goodman SB, Perkash I, Lane B, Enzmann DR. Benign versus pathologic compression fractures of vertebral bodies: assessment with conventional spin-echo, chemical-shift, and STIR MR imaging. Radiology. 1990; 174: 495-502.
- Campi A, Pontesilli S, Gerevini S, Scotti G. Comparison of MRI pulse sequences for investigation of lesions of the cervical spinal cord. Neuroradiology. 2000; 42: 669-675.
- Dietemann JL, Thibaut-Menard A, Warter JM, et al. MRI in multiple sclerosis of the spinal cord: evaluation of fast short-tan inversion-recovery and spin-echo sequences. Neuroradiology 2000; 42: 810-813.
- Despeyroux-Ewers M, Catalaa I, Collin L, Cognard C, Loubes-Lacroix F. Inflammatory myofibroblastic tumour of the spinal cord: Case report and review of the literature. Neuroradiology 2003; 45: 812-817.
- Coffin CM, Patel A, Perkins S, Elenitoba-Johnson KS, Perlman E, Griffin CA. ALK1 and p80 expression and chromosomal rearrangements involving 2p23 in inflammatory myofibroblastic tumor. Mod Pathol. 2001; 14: 569-576.
- Cook JR, Dehner LP, Collins MH, et al. Anaplastic lymphoma kinase (ALK) expression in the inflammatory myofibroblastic tumor: A comparative immunohistochemical study. Am J Surg Pathol 2001; 25: 1364-1371.
- Chun YS, Wang L, Nascimento AG, Moir CR, Rodeberg DA. Pediatric inflammatory myofibroblastic tumor: anaplastic lymphoma kinase (ALK) expression and prognosis. Pediatr Blood Cancer. 2005; 45: 796-801.