Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
COVID-19 in a pediatric cancer patient with T-lymphoblastic lymphoma: Response to convalescence plasma and subsequent induction of SARS-CoV-2 specific humoral and cellular immunity despite strong B-cell lymphopenia
Arne Simon1*; Tabea Reinhardt2; Sascha Meyer2; Yeliz Baltaci1; Jürgen Rissland3; Hermann Eichler4; Verena Klemis5; Martina Sester5; Norbert Graf1
1Pediatric Oncology and Hematology, Saarland University and Children’s Hospital Medical Center, Saarland University Hospital, Homburg, Germany.
2General Pediatrics and Neonatology, Saarland University and Children’s Hospital Medical Center, Saarland University Hospital, Homburg, Germany.
3Institute of Virology, Saarland University and Children’s Hospital Medical Center, Saarland University Hospital, Homburg, Germany.
4Institute of Clinical Hemostaseology and Transfusion Medicine, Saarland University and Children’s Hospital Medical Center, Saarland University Hospital, Homburg, Germany.
5Department of Transplant and Infection Immunology, Saarland University and Children’s Hospital Medical Center, Saarland University Hospital, Homburg, Germany.
*Corresponding Author : Arne Simon
Children’s Hospital Medical Center, Pediaric Oncology and hematology, Kirrberger Str. Building 0966421
Homburg, Saar Germany.
Email: Arne.Simon@uks.eu
Received : Apr 13, 2021
Accepted : May 10, 2021
Published : May 13, 2021
Archived : www.jcimcr.org
Copyright : © Simon A (2021).
Abstract
We report on an 11-year old pediatric cancer patient with T-lymphoblastic lymphoma who acquired a severe SARS-CoV-2 pneumonia immediately after the start of oral maintenance treatment with mercaptopurine. He was admitted to our hospital on day 7 after onset of symptoms (day 5 after diagnosis). Due to respiratory failure, he was transferred to the pediatric intensive care unit (from day 8 for 5 days) for high-flow-nasal cannula respiratory support. Treatment consisted of intravenous methylprednisolone and ceftriaxone. Because anti-SARS-CoV-2 antibodies were not detectable, he received two units of COVID-19 convalescence plasma containing high-titer neutralizing anti-SARS-CoV-2 antibodies (day 8 and day 9). Anti-SARS-CoV-2 antibodies increased to a maximum of 725 IU/L on day 10. Clinically, this was accompanied by a gradual decrease in oxygen demand and an overall clinical improvement. Remarkably, the patient started to mount a humoral immune response with continuously increasing anti-SARS-CoV-2 antibodies from day 15 up to 35.698 AU/ml on day 51 with extremely low (< 0.4%; < 0.01 x 109/L) detectible CD19-positive B-lymphocytes in his peripheral blood. Moreover, a strong SARS-CoV-2 specific CD4 and CD8 T cell response was induced with a predominance of CD8 T cells towards NCAP. This finding indicates that passive immunization facilitated active induction of both humoral and cellular immunity. This also suggests that active immunization may be feasible and effective in pediatric cancer patients despite B-lymphocytopenia.
Citation: Simon A, Reinhardt T, Meyer S, Baltaci Y, Rissland J, et al. COVID-19 in a pediatric cancer patient with T-lymphoblastic lymphoma: Response to convalescence plasma and subsequent induction of SARS-CoV-2 specific humoral and cellular immunity despite strong B-cell lymphopenia. J Clin Images Med Case Rep. 2021; 2(3): 1138.
Background
Although there are a number of reports on SARS-CoV-2 infections in pediatric cancer patients, it is still very difficult to define their particular risk to acquire severe COVID-19 after SARS-CoV-2 infection. Even during intensive chemotherapy, many severely immunocompromised pediatric cancer patients show only mild clinical symptoms after SARS-CoV-2 infection. Nonetheless, some severe COVID-19 cases have been described in this vulnerable pediatric population [1-3].
Case report
Our 11-year old male patient (44 kg, 155 cm) started with oral maintenance treatment (mercaptopurine, 25 mg/day) on February 1, 2021 for his high-risk T-lymphoblastic lymphoma treatment. Previous testing for SARS-CoV-2 on admission and during intensive chemotherapy was always negative in the patient and his mother. Due to the risk to acquire a SARS-CoV-2 infection outside of his home, he received home and digital schooling and did not meet with his friends or classmates. He and his mother were highly disciplined concerning social distancing and hand hygiene and always wore a medical mask when going outside.
On February 08 (formerly referred to as day 1), he visited our outpatient pediatric cancer complaining about a mild dry cough for 2 days, a reduced general condition, abnormal fatigue without fever and a loss of appetite due to a loss of taste. His clinical examination did not yield any abnormalities with regard to lung auscultation; he was eupneic with a normal respiratory rate and a transcutaneously measured oxygen saturation (SO2) of 94%. A PCR test of a nasopharyngeal swab yielded a positive result for SARS-CoV-2 [COBAS, cycle threshold (ct) of 30, no variant of concern). The source of this infection remained unclear; PCR-testing of all closed contacts of his household showed negative results for SARS-CoV-2.
Oral maintenance chemotherapy was stopped. On his next outpatient visit (day 3), he demonstrated no clinical deterioration, and no aggravation of symptoms and normal SO2. Peripheral blood cell count showed leukocytopenia (leukocyte count: 2.3 x 109/L) with 2 x 109/L neutrophils and 0.18 x 109/L lymphocytes, Hb 9.8 mg/dl and platelets at 132 x 109/L. Lymphocyte subpopulation analysis revealed no B-lymphocytes, 0.06 x 109/L (60/μl) TCD3/CD8 lymphocytes and 0.109 x 109/L (109/μl) TCD3/CD4 lymphocytes. Ferritin in serum was elevated at 3243 ng/ml, troponin T and pro-BNP were normal. Due to a C-reactive protein (CRP) of 101 mg/L (procalcitonin, PCT 0.37 ng/ml), antibiotic treatment was started with oral Sultamicillin.
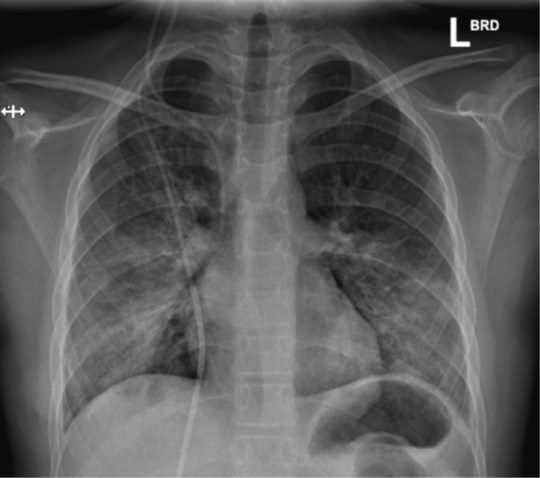
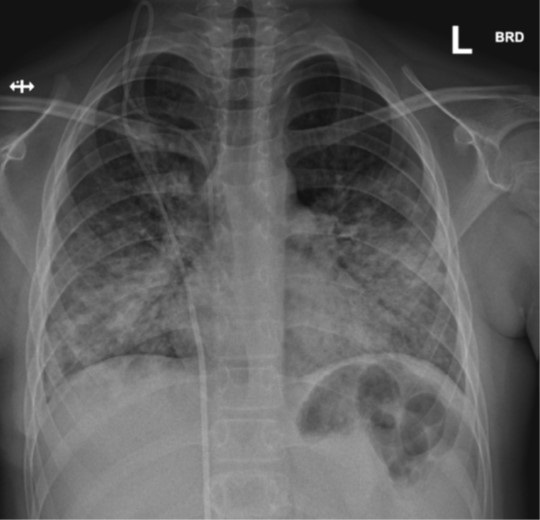
On day 5, his clinical examination was still unremarkable without dyspnea or tachypnea, but SO2 was only 88%, which resulted in hospital admission. On this day, he became pyrexic (39°C). Intravenous treatment with ceftriaxone and methylprednisolone (2 mg/kg/day) was started. A chest X-ray on day 6 and day 14 showed alveolar and peri-bronchial consolidations (Figure 1&2). Blood cultures taken on admission yielded no growth. Further diagnostic investigations (including PCR-based testing for 13 other respiratory viruses, atypical bacterial respiratory pathogens as well as a negative Legionella pneumophilia Type 1 urine antigen test) detected no other respiratory pathogen except SARS-CoV-2.
Repeated echocardiograms did not display abnormalities of ventricular contractility or increased pulmonary artery resistance, despite an elevation of Pro-BNP to a maximum of 877 pg/ml on day 8 (A detailed course of laboratory values in this patient is available in the online supplemental data). Due to a gradual but significant decrease in his respiratory and overall condition (despite oxygen supplementation up to 6 L/min via a nasal oxygen nasal cannula), high-flow nasal cannula ventilation had to be started on day 7, and he was transferred to the pediatric intensive care unit on day 8.
His clinical condition was worsening based on increasing respiratory rate and effort, decreasing exercise capacity, and low SO2 despite high-flow nasal cannula ventilation. Given the complete lack of both B cells and the absence of detectable anti-SARS-CoV-2 antibodies, we assumed that this patient would not be able to mount a humoral immune response to his infection. Considering previous reports from other groups [4,5] we decided to transfuse two units of each 282 ml convalescence plasma from a healthy 52-year old blood donor with a recent history of SARS-CoV-2 infection. These plasma products were donated following the inclusion criteria of the CAPSID Trial (EudraCT 2020-001310-38), but were not delivered within this trial. In a neutralization assay, anti-SARS-CoV-2 antibodies with a titre of 80 were detectable (Dr. Corman, Institut für Virologie, Charité – Universitätsmedizin Berlin).
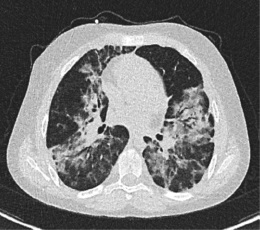
Prior to plasma transfusion on day 8 and 9, both parents and the patient gave written informed consent and the Clinical Ethics Committee of the Saarland University hospital provided an emergency approval for this individual treatment attempt (“Rescue Therapy”). Treatment with convalescence plasma was well tolerated. After the transfusion of the first unit, serum anti-SARS-CoV-2 antibodies increased to 428 AU/ml on day 9, to a maximum of 725 IU/L on day 10 after 2 units, and decreased again to 653 AU/ml on day 11. Clinically, this was accompanied by a gradual decrease in oxygen requirement and overall clinical improvement. High-flow nasal cannula ventilation could be stopped on day 11, and the patient was transferred from the pediatric ICU after 5 days to our regular non-intensive care unit. On the day of discharge from the hospital (day 16), a computer tomography of his chest still revealed extensive milk glass-appearing interstitial consolidations (Figure 3). PCR-based nasopharyngeal testing for SARS-CoV-2 revealed a positive result with a ct of 29 on day 8 and turned negative on day 14.
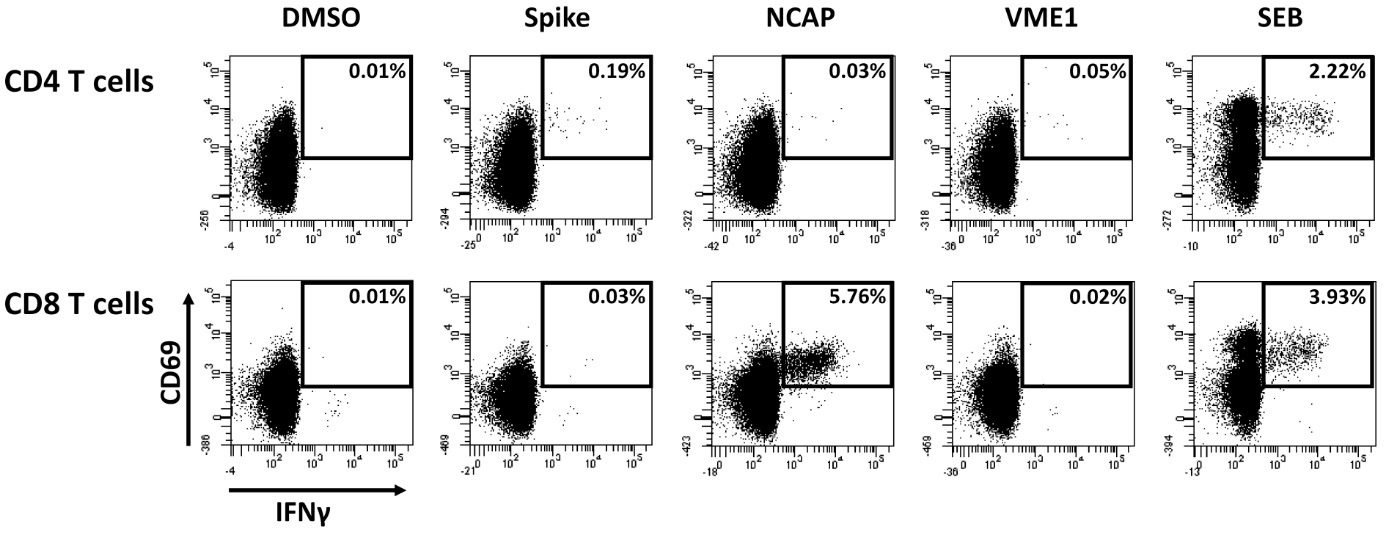
During the first week at home, he still had temperature peaks of up to 38.5°C in the evening and a SO2 of 90% at room air. His general condition (he lost 10% of his body weight) and his SO2 gradually improved and he could start walking outside and cycling again. The mother reported that he had some difficulties with concentration and short-term memory loss during home schooling, but these problems subsided gradually. Oral maintenance treatment of his T-lymphoblastic lymphoma was restarted on day 30. Of note, the patient developed a humoral immune response with continuously increasing anti-SARS-CoV-2 antibodies from 1360 AU/ml at day 14 up to 35.698 AU/ml on day 51. This is remarkable, as detailed lymphocyte analysis on day 57 from peripheral blood revealed only 0.31% circulating B cells, of which 92.9% had a IgD+/CD27- naïve phenotype. Among lymphocytes, 6.13% were NK cells and 85.0% were CD3 T cells comprising equal proportions of CD4 and CD8 T cells (48.6% each). Moreover, on day 57, SARS-CoV-2 specific CD4 and CD8 T cells were analyzed using intracellular staining of IFN-γ after stimulation with overlapping peptides encompassing the spike, Nucleocapsid (NCAP) and the membrane protein VME1 as previously described [6]. A total of 0.25% of SARS-CoV-2 specific CD4 T cells and 5.77% of specific CD8 T cells were detected with CD4 T cells predominantly reacting towards spike and specific CD8 T cells towards NCAP (Figure 4). Apart from IFN-γ, CD4 and CD8 T cells also had the ability to produce IL-2 and TNF-α (data not shown).
References
- André N, Rouger-Gaudichon J, Brethon B, Phulpin A, Thébault É, Pertuisel S, et al. COVID-19 in pediatric oncology from French pediatric oncology and hematology centers: High risk of severe forms? Pediatr Blood Cancer. 2020; 67: e28392.
- de Rojas T, Pérez-Martínez A, Cela E, Baragaño M, Galán V, Mata C, et al. COVID-19 infection in children and adolescents with cancer in Madrid. Pediatr Blood Cancer. 2020; 67: e28397.
- Stokes CL, Patel PA, Sabnis HS, Mitchell SG, Yildirim IB, Pauly MG. Severe COVID-19 disease in two pediatric oncology patients. Pediatr Blood Cancer. 2020; 67: e28432.
- Diorio C, Anderson EM, McNerney KO, Goodwin EC, Chase JC, Bolton MJ, et al. Convalescent plasma for pediatric patients with SARS-CoV-2-associated acute respiratory distress syndrome. Pediatr Blood Cancer. 2020; 67: e28693.
- Zaffanello M, Piacentini G, Nosetti L, Franchini M. The use of convalescent plasma for pediatric patients with SARS-CoV-2: A systematic literature review. Transfus Apher Sci. 2020.
- Schub D, Klemis V, Schneitler S, Mihm J, Lepper PM, Wilkens H, et al. High levels of SARS-CoV-2-specific T cells with restricted functionality in severe courses of COVID-19. JCI Insight. 2020.