Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Efficacy and safety of Chinese herbal medicine longdanxiegan decoction combined with val-acyclovir in herpes zoster: A systematic review and mate-analysis
Xiaoxu Wang1,3; Lingjun Wu2,3; Yihan Hou1,4; Shanshan Ding1,4; Siqing Wang1,4; Yinkai Wang1,4; Guangzhong Zhang1*
1Dermatological Department, Beijing Hospital of Traditional Chinese Medicine, Capital Medical University, Beijing 100010, China.
2Pediatric Department, Beijing Hospital of Traditional Chinese Medicine, Capital Medical University, Beijing 100010, China.
3Graduate School, Capital Medical University, Beijing 100069, China.
4Beijing University of Chinese Medicine, Beijing 100029, China.
*Corresponding Author : Guangzhong Zhang
Dermatological Department, Beijing Hospital of Traditional Chinese Medicine, Capital Medical University, Beijing 100010, China.
Email: zhgzh62000@126.com
Received : Apr 19, 2021
Accepted : May 19, 2021
Published : May 21, 2021
Archived : www.jcimcr.org
Copyright : © Zhang G (2021).
Abstract
Background: Herpes zoster is a global public problem that is characterised by a painful blistering rash and unilateral lesions and has a significant negative impact on the quality of patients’ lives. Longdanxiegan Decoction (LDXGD) is a classic herbal formula, and LDXGD combined with valacyclovir is widely used for herpes zoster patients in China. However, evidence-based medicine is not available. Therefore, it is necessary to systematically evaluate the effectiveness of LDXGD combined with valacyclovir in the treatment of herpes zoster, which will provide theoretical support for the treatment of herpes zoster.
Objective: This systematic review aimed to explore the effectiveness of LDXGD combined with val-acyclovir for herpes zoster and to compare LDXGD combined with val-acyclovir and val-acyclovir alone.
Methods: PubMed, Chinese Scientific Journals Database (VIP), China National Knowledge Infrastructure (CNKI), and Wan-fang Database were searched up to 6 April 2021. Randomised Controlled Trials (RCTs) were identified for herpes zoster treatment involving LDXGD combined with val-acyclovir. The quality of the literature was evaluated using the Cochrane assessment tool to reduce the risk of bias. RevMan 5.4.1 software was used to perform the meta-analysis.
Results: Five studies involving 414 participants were identified in this systematic review. Studies were of low to moderate methodological quality based on the risk of bias assessment. The results of our meta-analysis showed the relative benefits of LDXGD combined with valacyclovir in effective rates compared with valacyclovir alone (four studies; P=0.97, I2=0%, OR=4.48, 95%CI [1.89,10.67]), pain intensity (visual analogue scale) (MD: -1.40 mm; 95% CI [-1.89 to -0.92]), The incidence of PHN (RR 0.24; 95% CI [0.10, 0.55]; I2 =0%) and time to crust formation (MD= -1.66 days; 95% CI [2.01,1.30]). The safety of LDXGD combined with valacyclovir remains unclear.
Conclusion: Our systematic evaluation provides evidence for the clinical effectiveness and safety of LDXGD combined with valacyclovir for the treatment of herpes zoster. Further trials are necessary to collect evidence for the use of LDXGD.
Keywords: Herpes zoster; Longdanxiegan decoction; Valacyclovir; Systematic review; Randomized control trial.
Citation: Zhang G, Wang X, Wu L, Hou Y, Ding S, et al. Efficacy and safety of Chinese herbal medicine longdanxiegan decoction combined with val-acyclovir in herpes zoster: A systematic review and mate-analysis. J Clin Images Med Case Rep. 2021; 2(3): 1156.
Introduction
Herpes zoster, also called shinglesis, is the result of the reactivation of the varicella zoster virus [1]. After a primary infection with Varicella-Zoster Virus (VZV) in childhood, VZV establishes a dormant period in the sensory ganglia [1,2]. When VZV reactivates, it can travel along sensory nerve endings and cause herpes zoste [1,2]. Herpes zoster is characterised by a painful blistering rash and unilateral lesions that can usually lead to Postherpetic Neuralgia (PHN), which has a significant impact on the quality of patients’ lives [3,4]. The key risk factor for developing HZ is age [5,6]. Increasing studies have indicated that over 95% of adults have been infected with VZV worldwide, and about 30% of them will develop HZ later in life, and in those aged over 85 years, this proportion increases to 50% [7,8]. The most current treatment guidelines for herpes zoster mainly include antiviral medication [9]. Acyclovir and valacyclovir are the most commonly used antiviral drugs and represent the main antiviral agents used to treat herpes zoster [10]. Valacyclovir is a later-generation antiviral agent of acyclovir, which has improved pharmacokinetics and is considered a promising alternative to conventional acyclovir regimens [10]. However, a Cochrane review indicated that this type of medicine is not effective in reducing the incidence of Postherpetic Neuralgia (PHN), a common sequela of herpes zoster [11]. Therefore, it is necessary to develop a new treatment strategy for herpes zoster infection.
Although no evidence-based guidelines exist, Traditional Chinese Medicine (TCM) has been safely and effectively used in patients with herpes zoster for a long time. Longdanxiegan decoction (LDXGD) is a formula that can remove organ heat by TCM [12]. In traditional Chinese medicine, herpes zoster is caused by the heat of the organs, and LDXGD can remove heat from the organs and reduce toxins in many plaque herpes zoster patients. The LDXGD consists of 10 Chinese herbal medicines, including Thorowax root (Chai-hu in Chinese), Scutellaria baicalensis (Huang-qin in Chinese), Radix Gentianae (Long-dan-cao in Chinese), Gardenia jasminoides Ellis (Zhi-zi in Chinese), Rhizoma Alismatis (Ze-Xie in Chinese), Caulis Akebiae (Mutong in Chinese), Semen Plantaginis (Che-qian-zi in Chinese), Radix Angelicae Sinensis (Dang-gui in Chinese), Radix Rehmanniae Recen (Sheng-di in Chinese), and Radix Glycyrrhizae (Gan-cao in Chinese). Because of its considerable effect, LDXGD is commonly used for the treatment of herpes zoster in China. Thus, this review examined the efficacy and safety of the combination of LDXGD with valacyclovir for herpes zoster treatment. This will provide a new strategy for the treatment of herpes zoster.
Methods
Search strategies for identification of studies
We searched PubMed, Wan-fang Database, CNKI, and VIP from their inception to 6 April 2021 to identify RCTs that examined the efficacy and safety of the combination of LDXGD with valacyclovir in the treatment of herpes zoster. No restrictions on language We used the different search strategies for databases searching: For PubMed the keywords included: ((“Longdanxiegan Tang” [Title/Abstract] OR “Longdanxiegan Detection”[Title/Abstract]) OR (“valacyclovir”[Title/Abstract])) AND (“herpes zoster”[Mesh terms]) AND AND (“randomized controlled trial” OR “clinical trial”); for the 3 Chinese database, the keywords included: (“Dai Zhuang Pao Zhen” OR “She Chuan Chuang”) AND (“Fa Xi Luo Wei” OR “Longdanxiegan Tang”).
Inclusion criteria
Only RCTs that met the following criteria were included in this review: (a) all the included studies evaluated the efficacy and safety of LDXGD combined with valacyclovir for herpes zoster; (b) patients met the diagnostic criteria for herpes zoster; (c) RCTs comparing LDXGD combined with valacyclovir with valacyclovir alone; and (d)the data of the included literature was sufficient to support the evaluation.
Exclusion criteria
If the included literature meets the following conditions, it should be excluded: (a) studies were fundamental research or non-clinical RCT trials; (b) patients were diagnosed with complications of herpes zoster (zoster ophthalmicus, visceral or disseminated zoster, Ramsay Hunt syndrome, or bacterial infections), PHN, or patients combined with other serious diseases; and (c) use of other interventions to treat herpes zoster for a long time.
Types of outcome measures
The outcomes included: (a) therapeutic effective rate, defined as a significant reduction in pain and lesion improvement of at least 30%, according to a Chinese guideline (State Administration of Traditional Chinese Medicine, 1994), (b) the incidence of PHN, (c) pain intensity, (d) time to resolution of pain, and (e) time to crust formation.
Data extraction
Based on the above inclusion and exclusion criteria, two reviewers read the included literature independently, screened the study, and extracted the data. Quality assessment of the included studies was performed using the Cochrane Collaboration’s risk of bias tool. If there is any disagreement in the progress of assessments, a third researcher will assist in discussing and solving the problem.
Data synthesis and analysis
RevMan 5.4.1 software (Cochrane collaboration network) was used for data analysis. Continuous data are presented as Mean Difference (MD) with 95% confidence interval (CI) and I2. For dichotomous data, they were presented as odds ratio (OR) and Risk Ratio (RR), with 95% Confidence Interval (CI), and I2. In this meta-analysis, pain intensity, time to resolution of pain, and time to crust formation were presented as MD. The therapeutic effective rate was presented as OR, while the incidence of PHN and adverse effects were presented as RR. The random-effects model was used because of the heterogeneity among multiple studies. The result was considered statistically significant when P < 0.05. Because the number of included studies in this review was less than 10, it was not possible to explore publication bias. In addition, the protocol for this review was published in PROSPERO (Registration No. CRD42015029303).
Results
Study identification
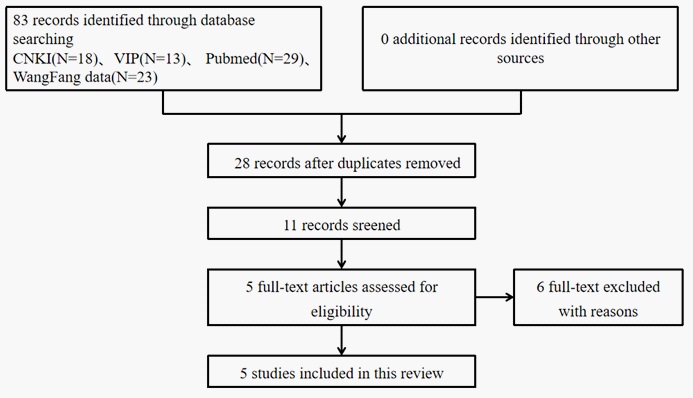
Database searches identified 83 articles, all of which were published studies, according to our search strategy. We excluded expert experience, reviews, theories, animal experiments, expert experience, case reports, and non-randomized controlled trials. After excluding duplicate records, we obtained 28 studies, and another 17 articles were excluded. Thus, 11 full-text articles were assessed for eligibility. Finally, five eligible RCTs (414 patients) met the inclusion criteria (Figure 1).
Study characteristics
Fourteen patients with herpes zoster were included in this review, and the sample size ranged from 60 [17] to 118 [15], and the median sample size was 80. All five studies were treated with LDXGD combined with valacycloviras versus valacyclovir alone for herpes zoster. All articles considered the therapeutic effective rate, PHN, pain intensity, time to resolution of pain, and time to crust formation as evaluation indexes. Patients in the treatment groups used LDXGD combined with valacycloviras as the related treatment. Patients in the control group received valacycloviras alone. The treatment duration of the included studies ranged from 7 days [14,17] to 4 weeks [16].
Methodological quality and risk of bias
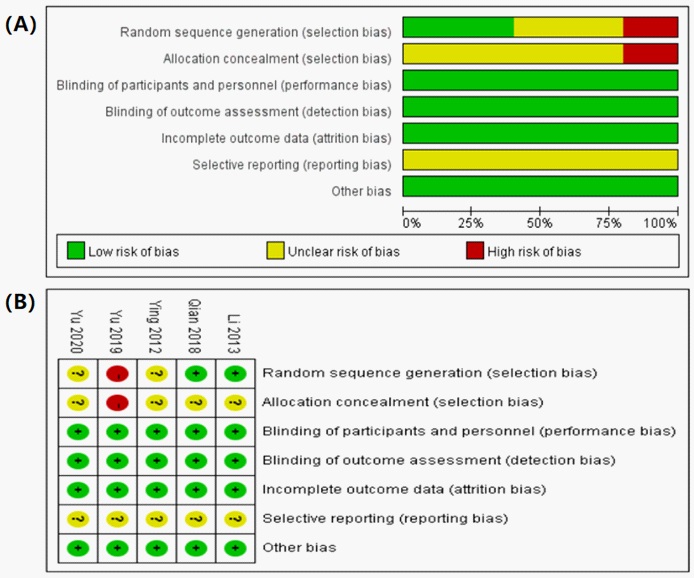
The methodological quality and risk of bias of these five RCTs were assessed according to the evaluation criteria in the Cochrane handbook and are summarised in Figure 2. Generally, the methodological quality of the included articles was low. Two studies used a random number table [15,17], which was assessed as having a low risk of bias for sequence generation. One study was assessed as having a high risk of bias [16] because they did not use any random classification and participants were divided into a research group and a control group according to the treatment method. The remaining studies only mentioned the use of random classification [14,18]; they did not provide sufficient information about the random numerical table method and were judged to be at unclear risk of bias. Others: There was no other bias in the included studies, which is unclear about the risk of bias. In addition, none of the trials provided sufficient information on the blinding method, selectivity in reporting data results, or completeness of outcome data, which could contribute to the improvement of therapeutic effectiveness.
Outcomes
Therapeutic effective rate
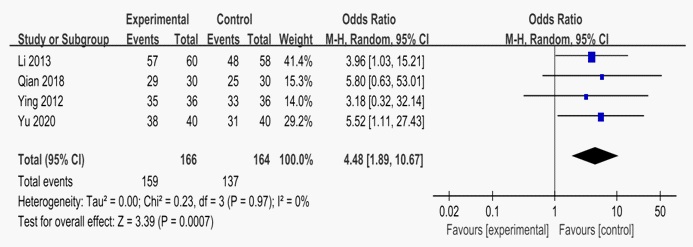
Four RCTs, including 330 patients, provided data for this system review. Four studies reported the clinical efficacy rate of LDXGD combined with valacyclovir compared with valacyclovir [14-15,17-18]. Heterogeneity test shows that P=0.97, I2=0%, OR=4.48, 95%CI [1.89,10.67]. The global effect test showed Z=3.39 (P=0.0007), and the results showed that the therapeutic efficacy rate of LDXGD combined with valacyclovir in the treatment of herpes zoster was higher than that in the control group.
The incidence of PHN
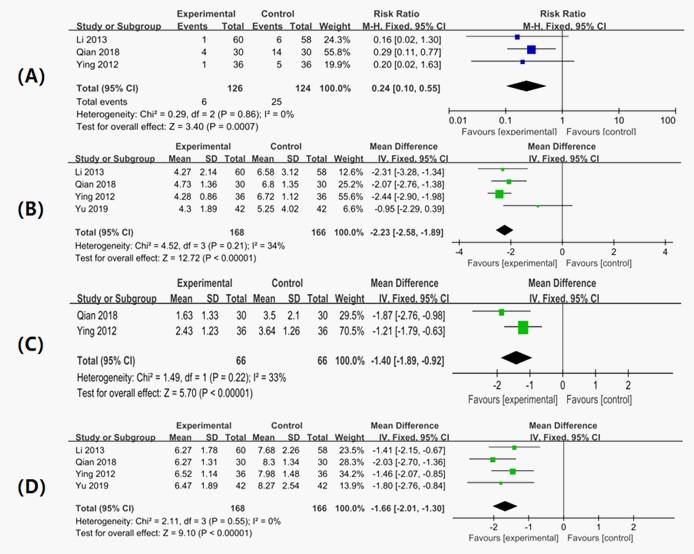
The incidence of PHN has been reported in three studies [14,15,17]. The definition of PHN is three months after resolution of the rash. Our analysis was performed based on this definition. The results of the meta-analysis demonstrated a significant reduction in the incidence of PHN in those who received LDXGD combined with valacyclovir compared with valacyclovir alone (RR, 0.24; 95% CI [0.10, 0.55]; I2=0%).
Pain intensity
Two studies reported pain intensity at the end of treatment [14,17]. The pain intensity in these studies was measured using the VAS pain scores. Meta-analysis showed that the pain score was 1.40 mm lower in the treatment groups than in the control groups (MD: -1.40 mm; 95% CI [-1.89, -0.92]). However, the difference between the two groups was less than 1 cm. Thus although the result was statistically significant, this may not be clinically meaningful.
Time to resolution of pain
Time to resolution of pain has been reported in four studies [14-17]. All studies evaluated the time from the start of treatment to the resolution of pain. A meta-analysis demonstrated that the time to resolution of pain was achieved 2.23 days earlier in the participants who received LDXGD combined with valacyclovir compared to valacyclovir alone (MD, -2.23 days; 95% CI,-2.58 -1.89.
Time to crust formation
The time to crust formation has been reported in four studies [14-17]. Crust formation measured from the start of treatment occurred 1.66 days earlier in those who received LDXGD combined with valacyclovir compared with those who received valacyclovir alone (MD= -1.66 days; 95% CI [2.01,1.30]).
Experimental: LDXGD combined with valacyclovir; Control, valacyclovir. LDXGD combined with valacyclovir versus valacyclovir alone.
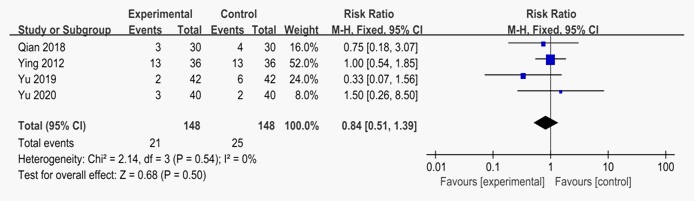
Adverse events
Four studies reported adverse effects [14,16-18]. All studies reported adverse events in the treatment and control groups, including drowsiness, diarrhoea, fatigue, and headache. Adverse events in these studies were not severe. Meta-analysis suggested that there was no significant difference between LDXGD combined with valacyclovir and valacyclovir alone (n = 296; RR: 0.84; 95% CI: 0.51 to 1.39; P=0.5). Thus, it is still unclear whether LDXGD combined with valacyclovir is more likely to have fewer adverse events than valacyclovir alone.
Discussion
LDXGD is a classic prescription in TCM, and is commonly used in the treatment of herpes zoster. In addition, it seems to be relatively safe due to the long-term use of LDXGD in TCM. However, the effectiveness and safety of LDXGD combined with valacyclovir for herpes zoster remains unknown. The findings from this review show the potential benefit of LDXGD combined with valacyclovir in reducing the incidence of PHN, promoting crust formation, and reducing pain intensity. A meta-analysis of five studies indicated that the therapeutic efficacy rate of LDXGD combined with valacyclovir in the treatment of herpes zoster was higher than that of valacyclovir alone, and the results were statistically significant. The results also demonstrated an improvement in lesions and a significant reduction in the incidence of PHN and pain intensity based on the use of LDXGD. At the same time, LDXGD treatment of herpes zoster is significantly beneficial in relaxing physical symptoms (such as dry mouth, bitter taste, yellow urine, and dry stool) caused by organ heat. This is worth promoting in clinical practice.
While many studies have reported some positive results, the overall studies included in this review had methodological flaws and the quality of these studies was not very high. This study has some limitations Listed as follows. First, the selective bias could have been caused by the lack of relevant foreign literature. Second, there is no description of the basis of the sample size estimation in the literature. Third, none of the studies described a clear method of random distribution. Fourth, the blind method was not mentioned in any research. Finally, not all the studies had long-term follow-up data which will help us improve the evaluation of LDXGD combined with valacyclovir for the treatment of herpes zoster. Thus, to a certain extent, the credibility of the research results is reduced. Although the research does not have a high quality, the conclusions demonstrate a certain reference value for clinical application. In the future, more high-quality research should be conducted to validate the effectiveness and safety of LDXGD combined with valacyclovir in the treatment of herpes zoster.
Acknowledgements: This work was supported by the National Natural Science Foundation of China (grant number 81974572).
Author contributions: Xiaoxu Wang conceived the idea of this study and wrote the paper. Wu, Hou, Wang, and Ding conducted a literature search and assessed their methodological quality. Disagreement was resolved by discussion between Yinkai Wang and Guangzhong Zhang. All authors approved the final version of the manuscript.
References
- Johnson RW, Levin MJ. Herpes Zoster and Its Prevention by Vaccination. Interdiscip Top Gerontol Geriatr. 2020; 43: 131-145.
- Kawai K, Yawn BP. Risk Factors for Herpes Zoster: A Systematic Review and Meta-analysis. Mayo Clin Proc. 2017; 92: 1806-1821.
- Tung YC, Tu HP, Wu MK, Kuo KL, Su YF, et al. Higher risk of herpes zoster in stroke patients. PLoS One. 2020; 15: e0228409.
- Bulilete O, Leiva A, Rullán M, Roca A, Llobera J. PHN Group. Efficacy of gabapentin for the prevention of postherpetic neuralgia in patients with acute herpes zoster: A double blind, randomized controlled trial. PLoS One. 2019; 14: e0217335.
- Koshy E, Mengting L, Kumar H, Jianbo W. Epidemiology, treatment and prevention of herpes zoster: A comprehensive review. Indian J Dermatol Venereol Leprol. 2018; 84: 251-262.
- Ehrenstein B. Diagnostik, Therapie und Prophylaxe des Herpes zoster Diagnosis, treatment and prophylaxis of herpes zoster. Z Rheumatol. 2020; 79: 1009-1017.
- Johnson RW, Alvarez-Pasquin MJ, Bijl M, Franco E, Gaillat J, et al. Herpes zoster epidemiology, management, and disease and economic burden in Europe: A multidisciplinary perspective. Ther Adv Vaccines. 2015; 3: 109–120.
- Marra F, Parhar K, Huang B, Vadlamudi N. Risk Factors for Herpes Zoster Infection: A Meta-Analysis. Open Forum Infect Dis. 2020; 7: ofaa005.
- Gross GE, Eisert L, Doerr HW, Fickenscher H, Knuf M, et al. S2k guidelines for the diagnosis and treatment of herpes zoster and postherpetic neuralgia. J Dtsch Dermatol Ges. 2020; 18: 55-78.
- Schuster AK, Harder BC, Schlichtenbrede FC, Jarczok MN, Tesarz J. Valacyclovir versus acyclovir for the treatment of herpes zoster ophthalmicus in immunocompetent patients. Cochrane Database Syst Rev. 2016; 11: CD011503.
- Chen N, Li Q, Yang J, Zhou M, Zhou D, He L. Antiviral treatment for preventing postherpetic neuralgia. Cochrane Database Syst Rev. 2014; (2): CD006866.
- Fan X, Su Z, Nie S, Yang J, Zhang X, et al. Efficacy and safety of Chinese herbal medicine Long Dan Xie Gan Tang in insomnia: A systematic review and meta-analysis. Medicine (Baltimore). 2020; 99: e19410.
- State Administration of Traditional Chinese Medicine, The Standard of Diagnosis and Assessment of Treatment Effects of Dermatological Conditions in Chinese Medicine, Nanjing University Press. 1994.
- Ying CHP, Ran NJ, Yang G. Clinical effect of valacyclovir combined with longdan xiegan decoction in herpes zoster. Chin J of Clinical Rational Drug Use. 2012; 5: 27-29.
- Li HX, Guo YM. Combination of “Longdan Xiegan Decoction” and western medicine for elderly herpes zoster: A report of 60 cases. SH. J. TCM. 2013; 47: 73-74.
- Yu J, Dong YL. Evaluation on the Curative Effect and Safety of Longdan Xiegan Decoction Combined with Valaciclovir in the Treatment of Herpes Zoster. Chinese Medicine Modern Distance Education Of China. 2019; 17: 106-107.
- Qian ZHZH. The clinical efficacy of Longdan Xiegan decotion combined with western medicine in the treatment of hepatobiliary damp-heated herpes zoster. Beijing. Beijing University Of Chinese Medicine. 2018.
- Yu N. Observation on clinical efficacy of valaciclovir hydrochloride tablets combined with Longdan Xiegan decoction on herpes zoster. Chin J Mod Drug Appl. 2020; 14: 192-193.