Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Myocarditis caused by salmonella enteritidis in a healthy 19-year-old male
Ina von Scheidt1*; Lena Maria Friedrich2; Amadeus Altenburger3 ; Thomas Pusl1
1 Department of Cardiology, Respiratory Medicine, Intensive Care, and Endocrinology, University Hospital Augsburg, University of Augsburg, Germany.
2 Department of Radiology, University Hospital Augsburg, University of Augsburg, Germany.
3 Radiology Augsburg Friedberg ÜBAG, Germany.
*Corresponding Author : Ina von Scheidt
Department of Cardiology, Respiratory Medicine,
Intensive Care, and Endocrinology. University
Hospital Augsburg, Stenglinstraße 2, 86156
Augsburg, Germany.
Email: ina.rupprecht@web.de
Received : Apr 20, 2021
Accepted : May 21, 2021
Published : May 25, 2021
Archived : www.jcimcr.org
Copyright : © von Scheidt I (2021).
Abstract
Myocarditis caused by nontyphoidal salmonellae is very rare in immunecompetent individuals. We present a case of a previous healthy 19-year-old male suffering from chest pain. Laboratory findings, ECG, echocardiogram and blood cultures led to the diagnosis of salmonella myocarditis. Cardiovascular magnetic resonance is a promising noninvasive diagnostic tool which is useful for follow-up and might help to confirm diagnosis. In patients with diarrhea and symptoms of angina pectoris, microbiological testing should include Salmonella enteritidis as possible cause for myocarditis.
Keywords: Cardiovascular magnetic resonance; endomyocardial biopsy; myocarditis; salmonella enteritidis.
Citation: von Scheidt I, Friedrich LM, Altenburger A, Pusl T. Myocarditis caused by salmonella enteritidis in a healthy 19-year-old male. J Clin Images Med Case Rep. 2021; 2(3): 1165.
Introduction
Myocarditis is defined histologically as an inflammation of the heart muscle with a large variety of infectious and noninfectious etiologies and an incidence of approximately 1.5 million cases in 2013 worldwide [1,2]. The diagnosis of myocarditis is usually based on clinical presentation, laboratory testing, electrocardiographic and echocardiographic findings and, in selected patients, other cardiac imaging tools like coronary angiography and Cardiovascular Magnetic Resonance (CMR). A definitive diagnosis can only be established by Endomyocardial Biopsy (EMB), although most of the patients do not undergo this invasive procedure [3,4].
Nontyphoidal salmonellae are a major cause for diarrhea worldwide with about 94 million cases and 155.000 deaths yearly [5]. The main reservoirs of these gram-negative bacteria represent animals and agricultural products [6]. Nontyphoidal salmonella gastroenteritis is usually self-limited and bacteremia or septic courses are very rare in immunocompetent individuals [5].
Case presentation
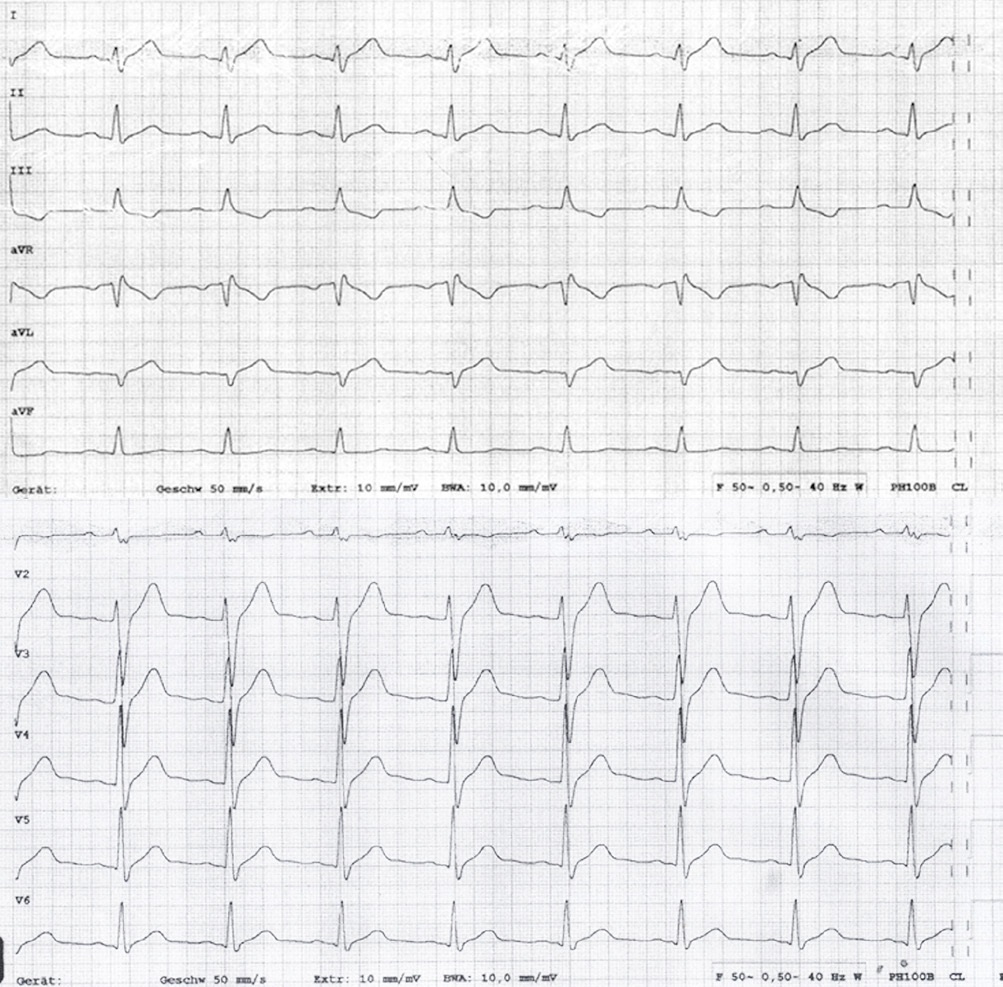
A 19-year-old man presented to our emergency department with an undulating, 1-week lasting fever, night sweats and abdominal pain. He complained about watery diarrhea and suffered from severe chest pain since the day before presentation. Two weeks earlier, he had returned from a travel to Cuba. His medical history was unremarkable. Physical examination of heart, lung and abdomen showed no abnormalities except for mild tachycardia of 103 bpm. Laboratory testing upon admission revealed an elevated WBC (11.2/nl, normal < 10.0/nl) and C-reactive protein (4.43 mg/dl, normal < 0.5 mg/dl). Heart enzymes were high (CK 487 U/l, normal < 190 U/l, CK-MB 57 U/l, normal < 25 U/l, high-sensitive troponin t 1017 pg/ml, normal < 14 pg/ml, NT-proBNP 1344 pg/ml, normal < 62,9 pg/ml). ECG showed sinus tachycardia and elevated ST-segments in I, aVL and V2-6 as well as negative T-wave in III (Figure 1). Left-ventricular function in echocardiogram was moderately reduced showing anterolateral hypokinesia. Suspecting myocarditis, the patient was transferred to our cardiology department. He was started on ibuprofen and acetaminophen as well as a heart failure treatment with ramipril, bisoprolol and ivabradine. Eventually, blood and stool cultures returned positive for Salmonella enteritidis. The initial empiric antibiotic therapy with piperacillin/tazobactam and azithromycin was changed to ciprofloxacin. The patient`s condition improved quickly. On day 3 of therapy the echocardiogram showed an improved left-ventricular function with mild inferior hypokinesia. Heart enzymes and inflammatory laboratory parameters normalized within 6 days. Immunoglobulin assay was normal, HIV test was negative. The patient was discharged on day 9 in good clinical condition. He received a 14-day course of ciprofloxacin and heart failure medication was continued until the last follow-up at 3 months. 3 months after hospitalization CMR revealed an almost normal left ventricular ejection fraction (53%), mild Dilated Cardiomyopathy (DCM) and no signs for an active or chronic myocarditis.
Discussion
Myocarditis can be a challenging diagnosis due to the wide spectrum of clinical manifestations, ranging from subclinical disease to acute coronary syndrome-like symptoms or overt heart failure that can in some individuals progress to life-threatening conditions, including arrhythmias and cardiogenic shock [1]. As an example, among 45 patients with suspected acute myocardial infarction and normal coronary angiograms, 35 patients had scintigraphic patterns of diffuse or focal myocarditis [7]. Similarly, in autopsy studies myocarditis was reported in up to 44 % of young persons after sudden cardiac death [8].
Most asymptomatic patients or patients with only mild symptoms and preserved left ventricular function have a good prognosis with low risk of complications [1]. In contrast, worse outcomes have been reported in patients with left ventricular ejection fraction < 50%, ventricular arrhythmias or low cardiac output syndrome upon presentation [9]. Prognosis in myocarditis also varies among different etiologies [10].
Viral infections are presumably the most common cause; nonetheless, bacterial infections range from 0.2 to 1.5 % and should always be considered [11]. Moreover, infections due to parasites and noninfectious causes like cardiotoxins (e.g. alcohol, anthracyclines or cocaine), hypersensitivity reactions, systemic disorders (e.g. celiac disease, sarcoidosis or thyrotoxicosis) and radiation should be taken into account [1,4].
Electrocardiogram, echocardiogram, laboratory testing and in some cases coronary angiography and CMR can establish diagnosis [3,4]. CMR is considered the most accurate noninvasive imaging tool for assessment of suspected acute myocarditis based on visualization of pathophysiological changes in myocardial tissue, including edema, hyperemia and myocyte necrosis or fibrosis defined in the Lake Louise Criteria (LLC) [12]. Novel quantitative CMR techniques like T1 and T2 mapping are a promising add-on which allow a more objective tissue characterization and might be able to overcome some of the previous limitations of the LLC [13]. The combination of clinical presentation and noninvasive cardiac imaging affirms diagnosis in most myocarditis cases. Nevertheless, EMB remains the goldstandard in myocarditis applying Dallas Criteria and immunohistochemical criteria. Considering the risks of this invasive technique and concerns about the diagnostic accuracy only selected patients receive EMB [14]. EMB is recommended for patients with unexplained fulminant heart failure, heart failure within 2 weeks to 3 months combined with dilated LV and/or higher grade arrhythmia or persistent heart failure despite usual care over 2 weeks [15]. Indication for EMB should consider likelihood that histological results would change therapy [3]. Myocarditis treatment comprises heart failure therapy in accordance to clinical presentation, mechanical circulatory support or heart transplantation in cases of severe and persistent heart failure, therapy for arrhythmia if needed and anticoagulation in case of atrial fibrillation, embolism or thrombus [1]. Nonsteroidal antiinflammatory drugs should not generally be used in patients with heart failure [16]. Moreover, alcohol restriction is recommended [17]. Physical exercise might not be performed during acute phase of myocarditis and heart failure. A return to competitive sports is possible after 3 to 6 months in case of normalized holter monitor, stress testing and electrocardiogram [18].
Nontyphoidal salmonellae are a major reason for diarrhea worldwide and cause about 94 million cases and 155.000 deaths per year. This food associated infection is usually self-limited [5]. Less than 5% of individuals with nontyphoidal salmonella gastroenteritis develop bacteremia what may be owed to an unappreciated immunological dysfunction like a HIV infection. Bacteremia can cause various complications like endocarditis, pericarditis, vascular infections or osteomyelitis [15,19]. The primary cardiovascular involvement is endocarditis, especially in patients with pre-existing atherosclerosis [20]. For salmonella bacteremia, a 14- day-course of fluoroquinolones or third-generation cephalosporin is recommended.
Ingested infectious dose determines frequency and rapidity of salmonella infection. Infectious threshold might be lower during antibiotic use or with impaired acidic barrier. Acidic environment of the stomach and normal intestinal microbial flora are protective host factors [21-25]. Adherence to and subsequent invasion through GI tract is complex and regulated by multiple genes. Salmonellae can survive within macrophages and spread into circulation [26]. Innate, cell mediated as well as humoral immune response are important to overcome infection. Impaired immunocompetence of different etiology like HIV, sickle cell disease, malaria, glucocorticoid use or B-cell disorders are associated with an aggravated course of disease [27-31].
There are only a few case reports about patients suffering from myocarditis due to nontyphoidal salmonella infection [32- 34].
In our case report, the patient was a young man with no risk factors for coronary heart disease and no identifiable immunodeficiency. Elevated heart enzymes and inflammatory parameters in combination with the ECG findings and the echocardiogram led to the diagnosis which was reassured after finding S. enteritidis in the blood culture. Anti-infective, anti-inflammatory and heart-failure therapy worked well and the patient recovered quickly. Therefore, we did not perform CMR or EMB to prove diagnosis during initial hospital stay. Outpatient CMR 3 months later confirmed a mild DCM.
In patients with diarrhea and symptoms of angina pectoris, S. enteritidis should be considered as possible germ causing myocarditis. As S. enteritidis bacteremia is more likely in patients with immunodeficiency, a screening for immunological dysfunction seems to be sensible. CMR as a non-invasive imaging modality is an important part of the diagnostic approach to myocarditis which is not only useful for follow-up examination but can also confirm diagnosis. Further research is needed to evaluate the clinical utility of novel quantitative mapping techniques in CMR and to integrate them into clinical workflows. Apart from that, there is no case of S. enteritidis myocarditis undergoing EMB procedure to our knowledge.
References
- Caforio AL, Pankuweit S, Arbustini E, Basso C, Gimeno-Blanes J, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013; 34: 2636-2648.
- Global Burden of Disease Study C. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990- 2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2015; 386: 743-800.
- Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, et al. 2013 ACCF/AHA guideline for the management of heart failure: executive summary: A report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation. 2013; 128: 1810-1852.
- Cooper LT, Jr. Myocarditis. N Engl J Med. 2009; 360: 1526-1538.
- Majowicz SE, Musto J, Scallan E, Angulo FJ, Kirk M, O’Brien SJ, et al. The global burden of nontyphoidal Salmonella gastroenteritis. Clin Infect Dis. 2010; 50: 882-889.
- Braden CR. Salmonella enterica serotype Enteritidis and eggs: A national epidemic in the United States. Clin Infect Dis. 2006; 43: 512-517.
- Sarda L, Colin P, Boccara F, Daou D, Lebtahi R, et al. Myocarditis in patients with clinical presentation of myocardial infarction and normal coronary angiograms. J Am Coll Cardiol. 2001; 37: 786-792.
- Basso C, Calabrese F, Corrado D, Thiene G. Postmortem diagnosis in sudden cardiac death victims: macroscopic, microscopic and molecular findings. Cardiovasc Res. 2001; 50: 290-300.
- Ammirati E, Cipriani M, Moro C, Raineri C, Pini D, Sormani P, et al. Clinical Presentation and Outcome in a Contemporary Cohort of Patients With Acute Myocarditis. Circulation. 2018; 138: 1088-1099.
- Caforio AL, Calabrese F, Angelini A, Tona F, Vinci A, et al. A prospective study of biopsy-proven myocarditis: prognostic relevance of clinical and aetiopathogenetic features at diagnosis. Eur Heart J. 2007; 28: 1326-1333.
- Wasi F, Shuter J. Primary bacterial infection of the myocardium. Front Biosci. 2003; 8: s228- 231.
- Kotanidis CP, Bazmpani MA, Haidich AB, Karvounis C, Antoniades C, et al. Diagnostic Accuracy of Cardiovascular Magnetic Resonance in Acute Myocarditis: A Systematic Review and Meta-Analysis. JACC Cardiovasc Imaging. 2018; 11: 1583-1590.
- . Ferreira VM. CMR Mapping for Myocarditis: Coming Soon to a Center Near You. JACC Cardiovasc Imaging. 2018; 11: 1591- 1593.
- Baughman KL. Diagnosis of myocarditis: death of Dallas criteria. Circulation. 2006; 113: 593- 595.
- Cooper LT, Baughman KL, Feldman AM, Frustaci A, Jessup M, et al. The role of endomyocardial biopsy in the management of cardiovascular disease: a scientific statement from the American Heart Association, the American College of Cardiology, and the European Society of Cardiology. Circulation. 2007; 116: 2216- 2233.
- Costanzo-Nordin MR, Reap EA, O’Connell JB, Robinson JA, Scanlon PJ. A nonsteroid anti- inflammatory drug exacerbates Coxsackie B3 murine myocarditis. J Am Coll Cardiol. 1985; 6: 1078-1082.
- Miller H, Abelmann, WH. Effects of dietary ethanol upon experimental Trypanosomal (T cruzi) myocarditis. Proc Soc Exp Biol Med. 1967; 126: 193.
- Maron BJ, Udelson JE, Bonow RO, Nishimura RA, Ackerman MJ, et al. Eligibility and Disqualification Recommendations for Competitive Athletes With Cardiovascular Abnormalities: Task Force 3: Hypertrophic Cardiomyopathy, Arrhythmogenic Right Ventricular Cardiomyopathy and Other Cardiomyopathies, and Myocarditis: A Scientific Statement From the American Heart Association and American College of Cardiology. Circulation. 2015; 132: e273-280.
- Cohen JI, Bartlett JA, Corey GR. Extra-intestinal manifestations of salmonella infections. Medicine (Baltimore). 1987; 66: 349-388
- Hsu RB, Lin FY. Risk factors for bacteraemia and endovascular infection due to non-typhoid salmonella: a reappraisal. QJM. 2005; 98: 821-827.
- Blaser MJ, Newman LS. A review of human salmonellosis: I. Infective dose. Rev Infect Dis. 1982; 4: 1096-1106.
- Ryan CA, Nickels MK, Hargrett-Bean NT, Potter ME, Endo T, et al. Massive outbreak of antimicrobial-resistant salmonellosis traced to pasteurized milk. JAMA. 1987; 258: 3269-3274.
- Neal KR, Briji SO, Slack RC, Hawkey CJ, Logan RF. Recent treatment with H2 antagonists and antibiotics and gastric surgery as risk factors for Salmonella infection. BMJ. 1994; 308: 176.
- Giannella RA, Broitman SA, Zamcheck N. Gastric acid barrier to ingested microorganisms in man: studies in vivo and in vitro. Gut. 1972; 13: 251-256.
- Endt K, Stecher B, Chaffron S, Slack E, Tchitchek N, Benecke A, et al. The microbiota mediates pathogen clearance from the gut lumen after non-typhoidal Salmonella diarrhea. PLoS Pathog. 2010; 6: e1001097.
- Hensel M, Shea JE, Waterman SR, Mundy R, Nikolaus T, Banks G, et al. Genes encoding putative effector proteins of the type III secretion system of Salmonella pathogenicity island 2 are required for bacterial virulence and proliferation in macrophages. Mol Microbiol. 1998; 30: 163-174.
- Ramos JM, Garcia-Corbeira P, Aguado JM, Arjona R, Ales JM, Soriano F. Clinical significance of primary vs. secondary bacteremia due to nontyphoid Salmonella in patients without AIDS. Clin Infect Dis. 1994; 19: 777-780.
- . Gruenewald R, Blum S, Chan J. Relationship between human immunodeficiency virus infection and salmonellosis in 20- to 59-year-old residents of New York City. Clin Infect Dis. 1994; 18: 358-363.
- Levine WC, Buehler JW, Bean NH, Tauxe RV. Epidemiology of nontyphoidal Salmonella bacteremia during the human immunodeficiency virus epidemic. J Infect Dis. 1991; 164: 81-87.
- Thamlikitkul V, Dhiraputra C, Paisarnsinsup T, Chareandee C. Non-typhoidal Salmonella bacteraemia: clinical features and risk factors. Trop Med Int Health. 1996; 1: 443-448.
- Mastroeni P, Simmons C, Fowler R, Hormaeche CE, Dougan G. Igh-6(-/-) (B-cell-deficient) mice fail to mount solid acquired resistance to oral challenge with virulent Salmonella enterica serovar typhimurium and show impaired Th1 T-cell responses to Salmonella antigens. Infect Immun. 2000; 68: 46-53.
- Sundbom P, Suutari AM, Abdulhadi K, Broda W, Csegedi M. Salmonella enteritidis causing myocarditis in a previously healthy 22-year-old male. Oxf Med Case Reports. 2018; 2018: omy106.
- Al Shamkhani W, Ajaz Y, Saeed Jafar N, Roy Narayanan S. Myocarditis and Rhabdomyolysis in a Healthy Young Man Caused by Salmonella Gastroenteritis. Case Rep Infect Dis. 2015; 2015: 954905.
- Villablanca P, Mohananey D, Meier G, Yap JE, Chouksey S, Abegunde AT. Salmonella Berta myocarditis: Case report and systematic review of non-typhoid Salmonella myocarditis. World J Cardiol. 2015; 7: 931-937.