Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Occult strongyloides stercoralis infection masquerading as a drug reaction with eosinophilic pneumonia
Heather Kirkham1; Stephanie Carnes1; Joshua Lieberman1; Seth Cohen2; Deepti Reddi1*
1 Department of Laboratory Medicine and Pathology, University of Washington Medical Center, USA.
2 Division of Allergy and Infectious Diseases, University of Washington Medical Center, USA.
*Corresponding Author : Deepti Reddi
Department of Laboratory Medicine and Pathology,
University of Washington Medical Center, USA.
Email: dreddi@uw.edu
Received : May 05, 2021
Accepted : May 24, 2021
Published : May 27, 2021
Archived : www.jcimcr.org
Copyright : © Reddi D (2021).
Abstract
Background: Pseudomelanosis or melanosis duodeni is seen in association with drugs, microorganisms or occasional bleeding, usually from peptic ulceration. We present a case of Strongyloides stercoralis presenting as pseudomelanosis duodeni during anemia workup after patient’s initial presentation as eosinophilic pneumonia.
Case presentation: The patient is a 77 year-old female with a history of diastolic congestive heart failure, chronic kidney disease, diabetes mellitus, and hypertension who presented for evaluation of black stool. Two months earlier, in her prior admission for acute eosinophilic pneumonitis, the patient’s hematocrit was 26%. In her current admission with heme-positive dark stool on exam, the hematocrit dropped to 19%. The patient underwent esophagogastroduodenoscopy to identify the source of bleeding. The upper endoscopy revealed three non-bleeding ulcers in the gastric antrum and discoloration of the duodenal bulb and second portion of the duodenum. By histology, the duodenal biopsies showed areas of hemosiderin-laden macrophages in the lamina propria, focal helminth eggs and larvae in the crypt lumen with sizes ranging from 35-65 microns x ~20 microns. Polymerase chain reaction of the paraffin embedded tissue with 28S primer set detected Strongyloides stercoralis DNA, confirming the histologic findings. Given the confirmation of Strongyloides stercoralis, the patient’s initial presentation of eosinophilic pneumonia (Loeffler syndrome) may be a result of the parasitic infection or the successful response to steroid treatment for acute eosinophilic pneumonia caused Strongyloidiasis hyperinfection. The patient’s symptoms were improved with Ivermectin and hematocrit level increased to 28%.
Conclusions: This case highlights the importance in recognition of helminth infection in the evaluation of eosinophilic pneumonia and pseudomelanosis duodeni.
Keywords: Pseudomelanosis duodeni; Strongyloides stercoralis; Eosinophilic pneumonia; polymerase chain reaction.
Citation: Kirkham H, Carnes S, Lieberman J, Cohen S, Reddi D. Occult strongyloides stercoralis infection masquerading as a drug reaction with eosinophilic pneumonia. J Clin Images Med Case Rep. 2021; 2(3): 1166.
Background
Strongyloides stercoralis is found in tropical and subtropical areas across the globe and is more prevalent in rural and remote areas or those with poor sanitation. The parasite’s life cycle alternates between free-living and parasitic cycles which include autoinfection. Autoinfection is of importance because this can result in persistent infection even after residing in a non-endemic area and may contribute to hyperinfection syndrome [1]. Here we present a case of Strongyloides stercoralis presenting as pseudomelanosis duodeni during anemia workup after patient’s initial presentation as eosinophilic pneumonia.
Case presentation
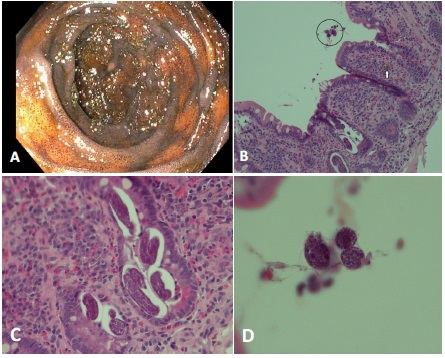
A 77-year-old female with a past medical history of chronic kidney disease, diabetes mellitus, labile hypertension and anemia presented to the hospital with one month duration of shortness of breath, recent fever, chills, nausea, vomiting, and body aches. Although the patient is originally from Korea and has extensively traveled throughout Asia in the past, she did not report any recent travel history within past three years. She also had no history of gastrointestinal disease or colon cancer. On examination she was found to have diastolic heart failure with pulmonary hypertension and acute hypoxemic respiratory failure with patchy consolidations in the right lung and diffuse bilateral ground glass opacities. Antibiotic and diuretic therapies were initiated however the patient had persistent pulmonary infiltrates and peripheral eosinophilia. A Bronchoalveolar Lavage (BAL) was performed and was found to have an eosinophilic predominance of 32% eosinophils (reference range 0-1%). Although patient reported to have one month duration of shortness of breath, she was thought to have eosinophilic pneumonia as a result of starting a new medication, allopurinol, two weeks prior to admission. She was discharged home on a prednisone taper and instructed to follow up with a pulmonologist. In the interim the patient’s chest X-ray improved, although the patient remained short of breath and had an acute worsening of anemia. One month later the patient presented with diffuse weakness, dark, loose stools, increased dyspnea, abdominal distention, nausea, puffiness of her face and persistent anemia. Endoscopy revealed three non-bleeding ulcers in the gastric antrum, diffuse discoloration of the duodenal bulb and second portion of the duodenum (Figure 1a). By histology, the duodenal biopsies showed areas of hemosiderin-laden macrophages in the lamina propria, helminth larvae in the crypt lumen with sizes ranging from 35-65 microns x ~20 microns (Figure 1 b,c) and focal helminth eggs (Figure 1d). The histopathologic differential included Strongyloides stercoralis, hookworm, or Ascaris spp. Polymerase Chain Reaction (PCR) of the paraffin embedded tissue with 28S primer set detected Strongyloides stercoralis DNA, confirming the histologic finding (Figures 2a,b). Steroids were discontinued and the patient was started on Ivermectin with subsequent improvement in her acute symptoms and hematocrit increased to 28%. In the most recent visit, the patient’s dyspnea on exertion has improved with diuretics, 40 mg torsemide daily. In addition, hematocrit continued to increase to 31% and hemoglobin increased to 10.4 g/dL (Table 1).
Table 1: Chronological summary of inpatient and outpatient laboratory values in 10 month period.
Dates |
Hemoglobin (g/dL) |
Hematocrit (%) |
Serum Iron (mcg/dL) |
Disease progression |
08/2019 |
7.7 |
29 |
17 |
Inpatient admission |
09/2019 |
8.2 |
26 |
|
Discharged with low-dose steroids |
10/2019 |
7.3 |
|
|
|
11/2019 |
5.8 |
19 |
27 |
Upper endoscopy |
12/2019 |
7.7 |
|
|
Ivermectin therapy |
01/2020 |
9.0 |
28 |
71 |
|
02/2020 |
10.2 |
|
|
|
03/2020 |
9.4 |
|
|
|
04/2020 |
9.8 |
31 |
78 |
|
05/2020 |
9.4 |
|
|
|
06/2020 |
10.4 |
|
|
|
Discussion
This case highlights an important differential of strongyloidiasis in the evaluation of eosinophilic pneumonia and pseudomelanosis duodeni. Eosinophilic pneumonia can be idiopathic or secondary to drugs, toxins, infections, asthma and systemic syndromes. Other entities to consider in this differential include drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome and Loeffler syndrome. DRESS syndrome is a cutaneous and systemic drug reaction which can present with fever, skin eruption and peripheral eosinophilia. A challenging diagnostic dilemma associated with DRESS syndrome is the progressive worsening of symptoms after the withdrawal of the offending agent [2]. Loeffler syndrome is a specific eosinophilic pneumonia which is transient and associated with the transpulmonary passage of parasitic larvae resulting in peripheral eosinophilia and radiographic infiltrates. It is most commonly associated with Ascaris lumbricoides, Strongyloides stercoralis and hookworms [3].
In our patient’s case, the cause of her initial clinical presentation with eosinophilic pneumonia is not entirely clear, particularly since symptom severity may be subjective or complicated by underlying disease. Our patient complained of shortness of breath prior to starting allopurinol and was found to have eosinophilia on BAL. This sequence of symptoms before drug exposure argues against allopurinol as the cause of her eosinophilic pneumonia. If Strongyloides stercoralis was on the initial differential, analysis of BAL for organisms may have proved valuable. After discontinuation of allopurinol and initiation of corticosteroid treatment, the patient experienced persistent shortness of breath with radiographic improvement of her pneumonia and worsening gastrointestinal symptoms. These observations indicate the exacerbation of the gastrointestinal manifestations of her helminth infection, but do not definitively resolve the cause of her pulmonary symptoms.
About one month after treatment with corticosteroids, our patient presented with symptoms of anemia, black stools, abdominal pain, puffiness of the face and endoscopic findings of pseudomelanosis duodeni. Pseudomelanosis duodeni is a clinically rare condition with pigmented macrophages in the lamina propria of the duodenum secondary to hemosiderin, lipomelanin or lipofuscin within macrophage lysosomes [4]. On endoscopy, the findings are often incidental, and the appearance can be a striking speckled black pigmentation of the duodenal mucosa. The pathogenesis of pseudomelanosis duodeni is not fully understood but associated with a variety of medical conditions such as hypertension, chronic kidney disease, diabetes mellitus, gastrointestinal bleeding, oral iron intake and medications. It is more common in the sixth and seventh decade of life with a female predominance [5]. Other conditions that may be considered in the context of these endoscopic or histologic findings include brown bowel syndrome, hemosiderosis/hemochromatosis or malignant melanoma [4].
In our patient’s case, biopsy of the duodenum revealed organisms within the crypts, which were later confirmed to be Strongyloides stercoralis. The previous treatment with steroids for eosinophilic pneumonia likely resulted in suppression of the patient’s immune system leading to hyperinfection by a chronic infection of Strongyloides stercoralis which then presented with gastrointestinal bleeding and anemia. In our patient the iron deficiency anemia is secondary to the inflammation of the intestinal mucosa exacerbated by hematophagism and bleeding ulcers as result of S. stercoralis infection itself [6]. Treatment with corticoids is the most important risk factor to develop S. stercoralis hyperinfection. The metabolites from corticosteroids resemble hydroxyecdysone, an ecdysteroid hormone that induces the transformation of rhabditiform larvae to infective filariform larvae, subsequently increasing the rate of autoinfection [7].
Diagnosis of strongyloidiasis can be made by the microscopic identification of rhabditiform or filariform larvae in the stool, duodenal fluid, biopsy specimen and sputum. Serial samples are typically required due to low parasite burden and low larval output [1]. In stool samples, Baermann’s method and agar plate culture methods are used to isolate the nematode larvae from stools [8]. Koga agar plate culture can be diagnostic, whereby fresh stool is incubated on an agar plate, and the larvae of helminths move across the plate, creating characteristic tracks in the subsequent bacterial growth. In our patient we performed only once both microscopic examination by Trichrome permanent stain on concentrated stool sample and Koga agar plate culture method. Both results were negative, highlighting the need for serial sampling. Unfortunately, additional samples could not be obtained in our case. Antibody detection by Enzyme Immunoassay (EIA) has the highest sensitivity (≥90%) of antibody based tests for Strongyloides infection diagnosis making it the recommended test by the Center of Disease Control, if serology is to be pursued. However, serology assays have significant cross-reactivity with a variety of helminths, hampering their use in endemic areas and do not differentiate between past and current infection [9-11]. PCR methods can be employed to detect Strongyloides spp. in fresh, frozen or nonformalin fixed stool specimens, but these assays are not widely available at this time [1]. The use of broad-range fungal PCR on paraffin embedded tissue is not validated, but can detect Strongyloides stercoralis because there is enough overlap in primer binding sites for the 28S rDNA locus [12].
A parasitic infection such as Strongyloides stercoralis should be included in the differential for eosinophilic pneumonia among those patients with a travel history, however remote. This case highlights the overlap in the clinical presentations of DRESS and Loeffler syndromes, and how these diagnoses can be intertwined. Chronic strongyloidiasis is generally asymptomatic, and treatment with steroids may lead to hyperinfection, as is suspected in our clinical case.
References
- Global Health, Division of Parasitic Disease and Malaria. Strongyloidiasis. Center for Disease Control and Prevention. 2019.
- Corneli HM. DRESS Syndrome: Drug Reaction With Eosinophilia and Systemic Symptoms. Pediatr Emerg Care. 2017; 33: 499- 502.
- Allen J, Wert M. Eosinophilic Pneumonias. J Allergy Clin Immunol Pract. 2018; 6: 1455-1461.
- Abumoawad A. Pseudomelanosis duodeni: A short review. American Journal of Digestive Disease. 2015.
- de Magalhães Costa MH, Fernandes Pegado Mda G, Vargas C, et al. Pseudomelanosis duodeni associated with chronic renal failure. World J Gastroenterol. 2012; 18: 1414-1416.
- Teixeira MC, Inês EJ, Pacheco FT, et al. Asymptomatic Strongyloides stercoralis hyperinfection in an alcoholic patient with intense anemia. J Parasitol. 2010; 96: 833-835.
- Teixeira MC, Pacheco FT, Souza JN, Silva ML, Inês EJ, et al. Strongyloides stercoralis Infection in Alcoholic Patients. Biomed Res Int. 2016; 2016: 4872473.
- Inês Ede J, Souza JN, Santos RC, et al. Efficacy of parasitological methods for the diagnosis of Strongyloides stercoralis and hookworm in faecal specimens. Acta Trop. 2011; 120: 206-210.
- Arifin N, Hanafiah KM, Ahmad H, Noordin R. Serodiagnosis and early detection of Strongyloides stercoralis infection. J Microbiol Immunol Infect. 2019; 52: 371-378.
- Inês Ede J, Silva ML, Souza JN, Teixeira MC, Soares NM. The role of glycosylated epitopes in the serodiagnosis of Strongyloides stercoralis infection. Diagn Microbiol Infect Dis. 2013; 76: 31-35.
- Norsyahida A, Riazi M, Sadjjadi SM, et al. Laboratory detection of strongyloidiasis: IgG-, IgG4 - and IgE-ELISAs and cross-reactivity with lymphatic filariasis. Parasite Immunol. 2013; 35: 174- 179.
- Konnick EQ, Chow SK, Reder NP, et al. Incidental identification of Strongyloides stercoralis infection by broad-range 28S rDNA gene sequencing in a patient with a hematolymphoid malignancy. Diagn Microbiol Infect Dis. 2016; 86: 362-364.