Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Bilateral cochlear implantation in a patient with osteopathia striata voorhoeve and cranial sclerosis
Kruthika Thangavelu1; Jochen Müller-Mazzotta1; Maximilian Schulze2; Rainer Weiß1; Boris A Stuck1; Katrin Reimann1*
1 Department of Otorhinolaryngology, Head and Neck Surgery, University Hospital Marburg, Philipps-Universität Marburg, Germany.
2 Department of Neuroradiology, University Hospital Marburg, Philipps-Universität Marburg, Germany.
*Corresponding Author: Kruthika Thangavelu
Department of Otolaryngology, Head and Neck Surgery,
University Hospital Marburg, Baldingerstr 1, 35043
Marburg, Germany.
Email: Kruthika.thangavelu@gmail.com
Received : Apr 22, 2021
Accepted : May 25, 2021
Published : May 28, 2021
Archived : www.jcimcr.org
Copyright : © Thangavelu K (2021).
Abstract
Objective: Osteopathia Striata with Cranial Sclerosis (OS-CS) is a rare form of X-linked dominant sclerosing bone dysplasia. The estimated prevalence of OS-CS is less than 1 in 1,000,000 in the general population with a little over 100 reported cases so far. Up to half of the patients suffer from hearing loss. Hearing loss is usually conductive rather than mixed or sensorineural in patients with OS-CS sclerosis. Herein, we report our experience with a patient who although initially had a conductive hearing loss, later developed sensorineural hearing loss on both sides and was treated with bilateral cochlear implantation.
Patient: Thirteen-year-old female patient suffering from Osteopathia striata with cranial sclerosis and functional deafness in the left ear and a high-grade sensorineural hearing loss in the right ear.
Intervention: Bilateral cochlear implantation
Main outcome measures: Postoperative hearing gain, clinical outcome, audiological examination.
Results: The patient showed good hearing gain of 30 dB on the left side and between 30 dB and 40 dB on the right side.
Conclusions: Despite the cranial sclerosis, cochlear implantation is a viable option in improving life quality of patients with Osteopathia striata. Nevertheless, it should be noted that in such cases, there is a possibility of progressive sclerosis leading to further narrowing of the internal auditory canal or even complete closure thereby damaging the auditory nerve irreversibly.
Keywords: Purple urine bag syndrome (PUBS); purple discolouration; urinary tract infection (UTI); long-term catheterization.
Citation: Thangavelu K, Müller-Mazzotta J, Schulze M, Weiß R, Stuck BA, et al. Bilateral cochlear implantation in a patient with osteopathia striata voorhoeve and cranial sclerosis. J Clin Images Med Case Rep. 2021; 2(3): 1171.
Introduction
Osteopathia striata or Voorhoeve Disease is a rare type of sclerosing bone dysplasia, first described by Voorhoeve in 1924 [1]. It is an autosomal dominant or sporadic inherited disorder that usually affects all bones except the skull and the clavicle, and has a particular predilection for long bones [2]. It can present either in isolation in pure form or as part of a variety of syndromes [3].
The pure form of the disorder does not exhibit any associated physical abnormalities or characteristic laboratory findings, and diagnosis is based exclusively on radiographic findings. The hallmark of the disorder is the delicate or sometimes coarse, dense linear striations as a result of a failure of remodeling of the persistent mature lamellar bone. This condition appears to reflect old bone remodeling and the lesions are not associated with increased scintigraphy [3].
Hurt reported a rare combination of Osteopathia Striata with Cranial Sclerosis (OS-CS) [4]. He recognized the association between dense striations in the long bones and pelvis typical of osteopathia striata, and thickening and sclerosis of the skull, particularly at the base but also minimally affecting the vault, as an X-linked dominant entity [5]. Cranial sclerosis is particularly disabling leading to hearing loss and paralysis of cranial nerves. Affected female patients usually present features related to cranial sclerosis such as macrocephaly, wide nasal bridge, ocular hypertelorism, abnormalities of the palate and mild learning disabilities. Occasional surviving males have cardiac, intestinal, and genitourinary malformations, in addition to hyperostosis. Osteopathia striata is also a frequent feature of focal dermal hypoplasia [6].
OS-CS has recently been identified to be associated with mutations in the Wilms tumor gene on the X chromosome (WTX), a repressor of WNT signaling (beta-catenin pathway implicated in control of target genes in the nucleus) [6]. The estimated prevalence of OS-CS is less than 1 in 1,000,000 in the general population with a little over 100 reported cases so far. Hearing loss has been reported in up to half of the patients [7].
This report discusses a patient who displayed phenotypic characteristics associated with OS-CS and received cochlear implantation on both sides because of sensorineural hearing loss
Case description
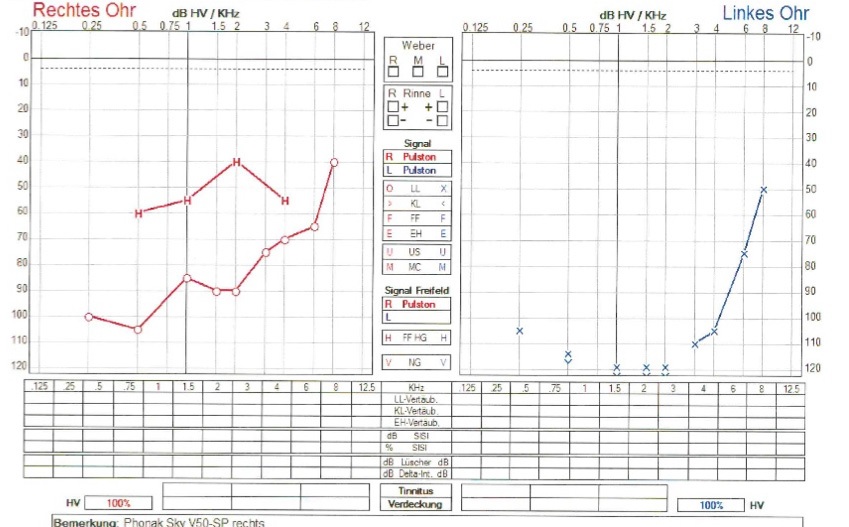
The patient, a 13-year-old female adolescent, was the product of a gravida 1, para 1, unrelated parents. The pregnancy was uncomplicated, and there was no exposure to alcohol, tobacco, or illicit drugs during the pregnancy. The mother, however, suffers from osteopathia striata and sensorineural hearing loss although with comparatively milder symptoms. At birth, the patient was noted to have microcephaly. She later progressively developed mental retardation, hyperopia, astigmatisms and strabismus. She presented in our hospital in the department of phoniatrics and pediatric audiology at the age of 2 years with known hearing loss and no other clear history. A low-grade conductive hearing loss was diagnosed using Click-BERA. A second BERA after the cleaning of middle ear through myringotomy, showed no relevant hearing loss. After 3 years, the patient presented again with conductive hearing loss because of middle ear effusions, which was treated with myringotomy and middle ear ventilation tubes. At the age of 12, she presented to us again with progressive hearing loss and her audiological tests showed a functional deafness in the left ear and a high-grade sensorineural hearing loss in the right ear (Figure 1). Due to her mental retardation, the girl’s vocabulary was already limited. But in the last months, both her perceptive and expressive language skills had notably deteriorated. Her pronunciation had grown sloppy and her vocabulary was found to be more restricted than usual. Cochlear implantation was considered initially for the left side and respective preoperative diagnostics were performed.
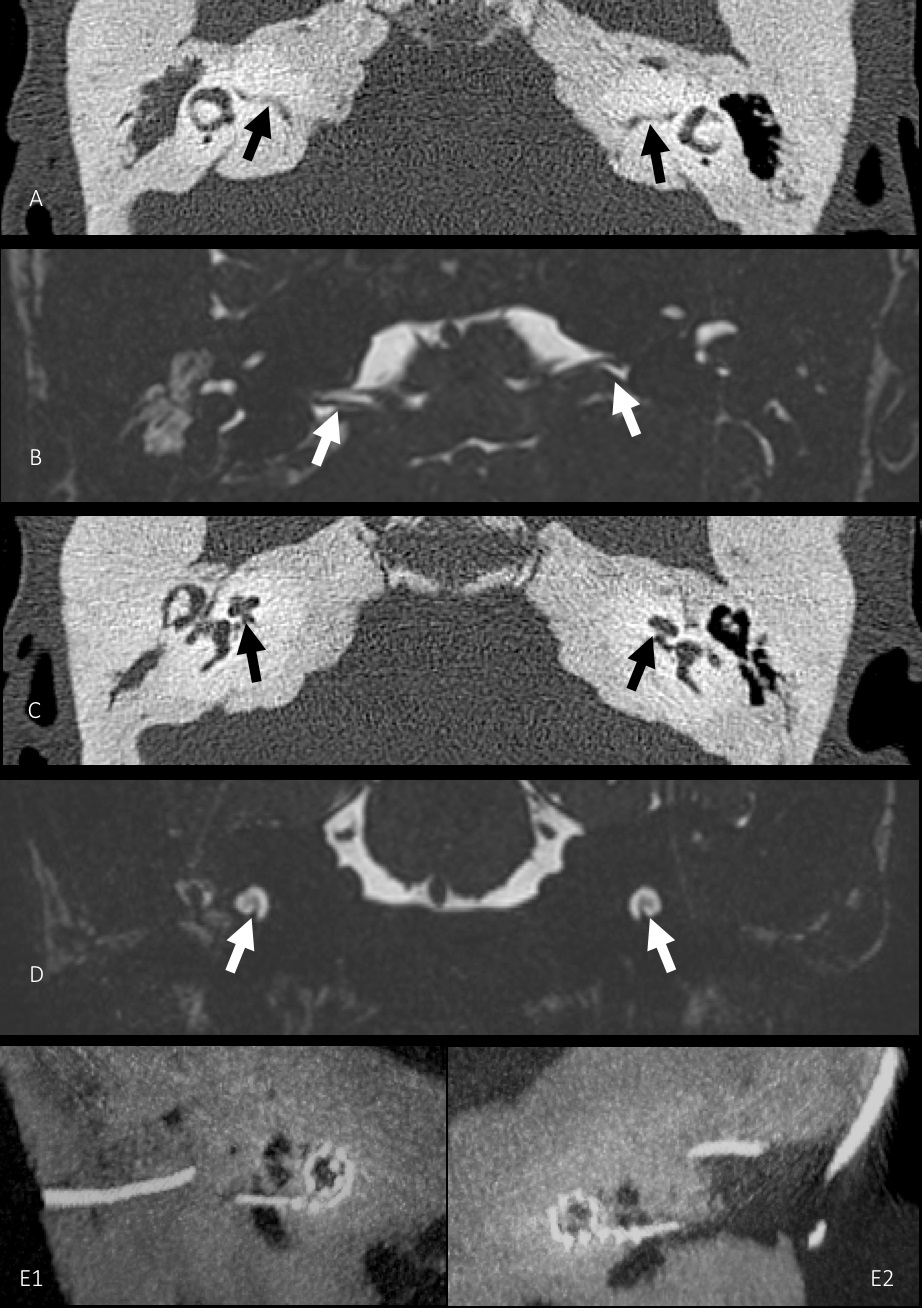
Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) scans of the temporal bones showed sclerosis of the temporal and petrous bone on both sides with narrowing of the inner ear canal. Despite this, the labyrinth showed normal configuration with normal fluid signal on both sides. The auditory nerve was intact and showed no signs of degeneration (Figure 3).
Cochlear implantation was performed as planned on the left side. The surgery was done with the help of facial nerve monitoring. No specific intraoperative complications were noted. The patient did not develop any post-operative complications (Figure 3).
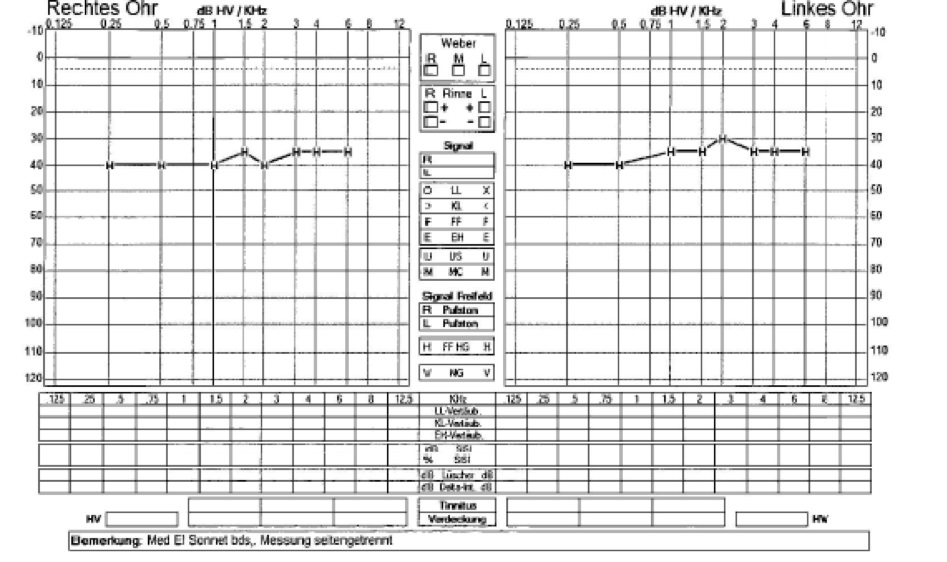
Post operatively the patient showed good improvement of her hearing. The audiological test after switching on the implant showed a hearing gain of around 30 dB across all frequencies (Figure 2). In the meantime, she showed progressive hearing loss on the right side despite having a hearing aid. Eight months after the left side, cochlear implantation was also done on the right side. Audiometry performed 7 weeks after switch-on on the right side showed a hearing gain between 30 and 40 dB across all frequencies (Figure 2). The Goettingen child hearing test showed that the patient was able to recognize and repeat 50% of the words correctly at 80 dB on the right side with cochlear implantation only on the right side. On the left side, the patient recognized and repeated 70% of the words at 80 dB with cochlear implantation on both sides. Since then, the patient has showed good acceptance of and compliance with cochlear implantation and achieved progressively better hearing.
Discussion
Bilateral sensorineural hearing loss of 40 dB or more has been found in about 1 to 2 in every 1000 newborns in developed countries [8]. Hearing loss is found to be usually conductive rather than mixed or sensorineural in patients with OS-CS sclerosis [9-14]. Mixed or sensorineural hearing loss [15]. and deafness [16,17] have been described in very few cases in the literature.
The otologic manifestations causing conductive hearing loss in OS-CS are recurrent serous otitis media, sclerotic mastoids, narrowing of the middle ear cavity and Eustachian tubes and rarely ossicular fixation. Temporal bone CT is usually used to evaluate the various etiologies of conductive hearing loss [6]. On the other hand, etiology of sensorineural hearing loss has been difficult to pinpoint given the limited number of available case studies. In our case, although the patient showed a conductive hearing loss in her initial years, she later developed a sensorineural hearing loss. The sensorineural hearing loss in our patient may be attributed to the massive sclerosis of the cochlear capsule or due to the compression of the artery supplying blood to the narrow inner auditory canal.
Although conductive hearing loss has been variously treated with middle ear surgery or with ventilation tubes depending on its etiology, hearing aid seems to be the standard treatment of choice in case of sensorineural hearing loss in the few reported cases so far [16,18,19].
Cochlear implantation has become a very viable treatment option in patients with bilateral deafness and profound sensorineural hearing loss. Over the years, it has gradually begun to be used in patients with severe sensorineural hearing loss who have not benefited from hearing aids. At present, an increasing proportion of implanted patients are adults with postlingual deafness, where the only limitations are their general condition and biological age [20]. To our knowledge, ours is the first case describing bilateral cochlear implantation in a case of osteopathia striata Voorhoeve with cranial sclerosis, and other sclerosing bone dysplasias in the cranium. Although cochlear implantation was indicated and considered in one other case of a child with Saethre-Chotzen syndrome and sensorineural hearing loss, it was not done before the report was published [21]. Despite mental retardation in our patient, post-operative benefit from the cochlear implantation was considerable. Although difficult anatomical conditions might constitute a challenge for surgeons in cases of cranial sclerosis, better imaging possibilities can help surgeons to evaluate tricky anatomical variations. Despite the cranial sclerosis, cochlear implantation is a viable option in improving life quality of such patients. Nevertheless, it should be noted that in such cases, there is a possibility of progressive sclerosis leading to further narrowing of the internal canal or even complete closure thereby damaging the auditory nerve irreversibly.
The incidence of bone dysplasias in general is low and Voorhoeve is a rare form of bone dysplasia. Voorhoeve disease presenting with cranial sclerosis is even rarer. A limited patient group makes it difficult to study the benefits of cochlear implantation in these cases. Therefore, in spite of this being a case report about a single patient, we believe that it is still a valuable addition to the sparse literature on the topic.
References
- Voorhoeve N. L’image radiologique non encore decrite d’une anomalie du squelette: Ses rapports avec la dyschondroplasie et l’osteopathia condensais disseminata. Acta radiologica. 1924: 407-427.
- Fairbank HT. 14. Osteopathia Striata. The Journal of Bone and Joint Surgery. British 1950; 32: 117-125.
- Greenspan A. Sclerosing bone dysplasias- A target-site approach. Skeletal Radiol 1991; 20: 561-583.
- Hurt RL. Osteopathia striata-Voorhoeve’s disease: Report of a case presenting the features of osteopathia striata and osteopetrosis. J Bone Joint Surg Br. 1953; 35-B: 89-96.
- Holman SK, Morgan T, Baujat Get al. Osteopathia striata congenita with cranial sclerosis and intellectual disability due to contiguous gene deletions involving the WTX locus. Clin Genet. 2013; 83: 251-256.
- Zicari AM, Tarani L, Perotti Det al. WTX R353X mutation in a family with osteopathia striata and cranial sclerosis (OS-CS): Case report and literature review of the disease clinical, genetic and radiological features. Ital J Pediatr. 2012; 38: 27.
- Magliulo G, Parrotto D, Zicari AMet al. Osteopathia striata-cranial sclerosis: otorhinolaryngologic clinical presentation and radiologic findings. Am J Otolaryngol 2007; 28: 59-63.
- Brookhouser PE. Incidence and prevalence. TITLE Consensus Development Conferenceon Early Identification of Hearing Impairment in Infants and Young Children (Bethesda, Maryland. 1993; 1-3: 29.
- Rott HD, Krieg P, Rutschle Het al. Multiple malformations in a male and maternal osteopathia strata with cranial sclerosis (OSCS). Genet Couns. 2003; 14: 281-288.
- Viot G, Lacombe D, David A, et al. Osteopathia striata cranial sclerosis: non-random X-inactivation suggestive of X-linked dominant inheritance. Am J Med Genet. 2002; 107: 1-4.
- Pellegrino JE, McDonald-McGinn DM, Schneider A, et al. Further clinical delineation and increased morbidity in males with osteopathia striata with cranial sclerosis: an X-linked disorder? Am J Med Genet. 1997; 70: 159-165.
- Piechowiak H, Goebel FD, Hirche Uet al. Cranial sclerosis with striated bone disease (osteopathia striata). Klin Padiatr 1986; 198: 418-424.
- Horan FT, Beighton PH. Osteopathia striata with cranial sclerosis. An autosomal dominant entity. Clin Genet. 1978; 13: 201-206.
- Jones MD, Mulcahy ND. Osteopathia striata, osteopetrosis, and impaired hearing. A case report. Arch Otolaryngol. 1968; 87: 116-118.
- Bass HN, Weiner JR, Goldman Aet al. Osteopathia striata syndrome. Clinical, genetic and radiologic considerations. Clin Pediatr (Phila). 1980; 19: 369-373.
- De Keyser J, Bruyland M, De Greve Jet al. Osteopathia striata with cranial sclerosis. Report of a case and review of the literature. Clin Neurol Neurosurg. 1983; 85: 41-48.
- Franklyn PP, Wilkinson D. Two cases of osteopathia striata, deafness and cranial osteopetrosis. Ann Radiol (Paris). 1978; 21: 91- 93.
- Berenholz L, Lippy W, Harrell M. Conductive hearing loss in osteopathia striata-cranial sclerosis. Otolaryngol Head Neck Surg. 2002; 127: 124-126.
- Luerssen K, Ptok M. Osteopathia striata with cranial sclerosis and hearing loss. Eur Arch Otorhinolaryngol. 2006; 263: 123- 126.
- Szyfter W, Karlik M, Sekula Aet al. Current indications for cochlear implantation in adults and children. Otolaryngol Pol. 2019; 73: 1-5.
- Lee S, Seto M, Sie K, et al. A child with Saethre-Chotzen syndrome, sensorineural hearing loss, and a TWIST mutation. Cleft Palate Craniofac J. 2002; 39: 110-114.