Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Ultrasound-guided diagnosis of an uncommon stroke: A case of giant cell arteritis
Mariella Baldini1 ; Sara Giannoni1 *; Elisa Cioffi2 ; Elisa Grifoni2 ; Elisabetta Bertini1 ; Eleonora Cosentino2 ; Ira Signorini2 ; Cinzia Grisanti1 ; Serena Colon1 ; Chiara Stagnaro3 ; Antonio Segneri4 ; Stelvio Sestini5 ; Leonello Guidi1 ; Luca Masotti2
1 Neurology, San Giuseppe Hospital, Empoli, Italy.
2 Internal Medicine II, San Giuseppe Hospital, Empoli, Italy.
3Rheumatology Unit, Department of Clinical and Experimental Medicine, University of Pisa, Pisa, Italy.
4Radiology, San Giuseppe Hospital, Empoli, Italy.
5Unit of Nuclear Medicine, Department of Diagnostic Imaging, S. Stefano Hospital, Prato, Italy.
*Corresponding Author: Sara Giannoni
Neurology, San Giuseppe Hospital, Empoli, Italy.
Email: giannonisara@gmail.com
Received : Apr 22, 2021
Accepted : May 27, 2021
Published : May 31, 2021
Archived : www.jcimcr.org
Copyright : © Giannoni S (2021).
Abstract
Giant Cell Arteritis (GCA) is an immune-mediated vasculitis of large- and medium-sized vessels. Stroke happens in 3% to 7% of the cases, and identifying GCA properly is important because potentially curative treatment exists. We present a case of an 83-year-old woman, in whom suspected GCA was revealed by stroke caused by Vertebral Artery (VA) inflammation. The uncommon stroke localization and the ultrasound findings suggested vasculitic etiology of lesions. 18-Fluorodeoxyglucose-Positron Emission Tomography (18-FDG-PET) confirmed inflamed VA walls. Patient was treated with oral steroids and antiplatelet therapy, with improvement of clinical and ultrasonographic status. In our patient clinical diagnosis of GCA was challenging due to limited classical symptoms. Arguments in favor of arteritis were the symmetric and bilateral involvement of the VA, the sonographic halo sign and atypical stroke localization.
Abbreviations: VA: Vertebral Artery; MRI: Magnetic Resonance Imaging; 18-FDG-PET: 18-Fluorodeoxyglucose-Positron Emission Tomography; AICA: Anterior Inferior Cerebellar Artery; PICA: Posterior Inferior Cerebellar Artery; GCA: Giant Cell Arteritis.
Citation: Baldini M, Giannoni S, Cioffi E, Grifoni E, Bertini E, et al. Ultrasound-guided diagnosis of an uncommon stroke: A case of giant cell arteritis. J Clin Images Med Case Rep. 2021; 2(3): 1173.
Introduction
Isolated bilateral cerebellar stroke is extremely uncommon and only a few cases have been described in the literature [1]. We present the case of an 83-year-old woman with bilateral cerebellar dentate nuclei stroke, caused by multifocal stenosis in bilateral Vertebral Artery (VA) due to arteritis. The diagnosis was achieved by integrating Magnetic Resonance Imaging (MRI), Computed Tomography (TC) and 18-FluorodeoxyglucosePositron Emission Tomography (18-FDG-PET) analysis and follow up after steroid treatment, but most of all by neurosonologic findings.
Case presentation
An 83-year-old woman had a sudden onset of postural instability and bilateral hypoacusia. The patient did not have any hypovolemic event right before the admission and past medical history wasn’t significant for headache or diffuse muscle pain. Patient was treated for hepatocarcinoma seven years before, in absence of cancer relapse.
Laboratory tests didn’t show significant pathological findings suggesting inflammation. During hospitalization the patient developed acute confusional state and delirium treated with antipsychotic drugs.
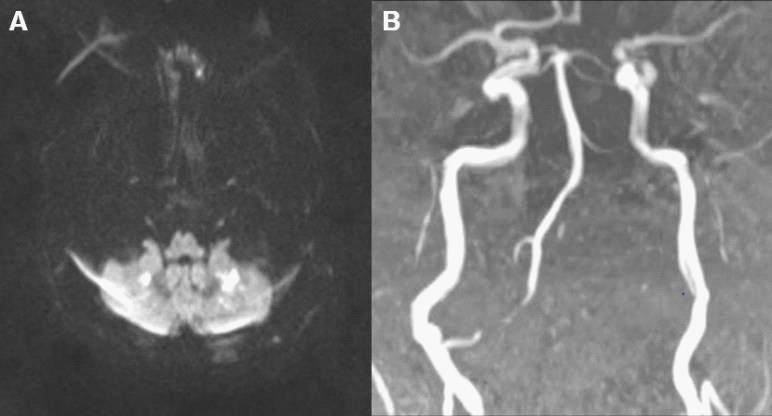
TC-Angiography showed a filiform lumen of right VA and occlusion of V3-V4 segment of left VA, right Anterior Inferior Cerebellar Artery (AICA) and left Posterior Inferior Cerebellar Artery (PICA). Brain MRI pointed out restricted diffusion lesions in bilateral cerebellar dentate nuclei, consistent with acute ischemic stroke. MR-angiography confirmed the previous intracranial vascular findings (Figure 1).
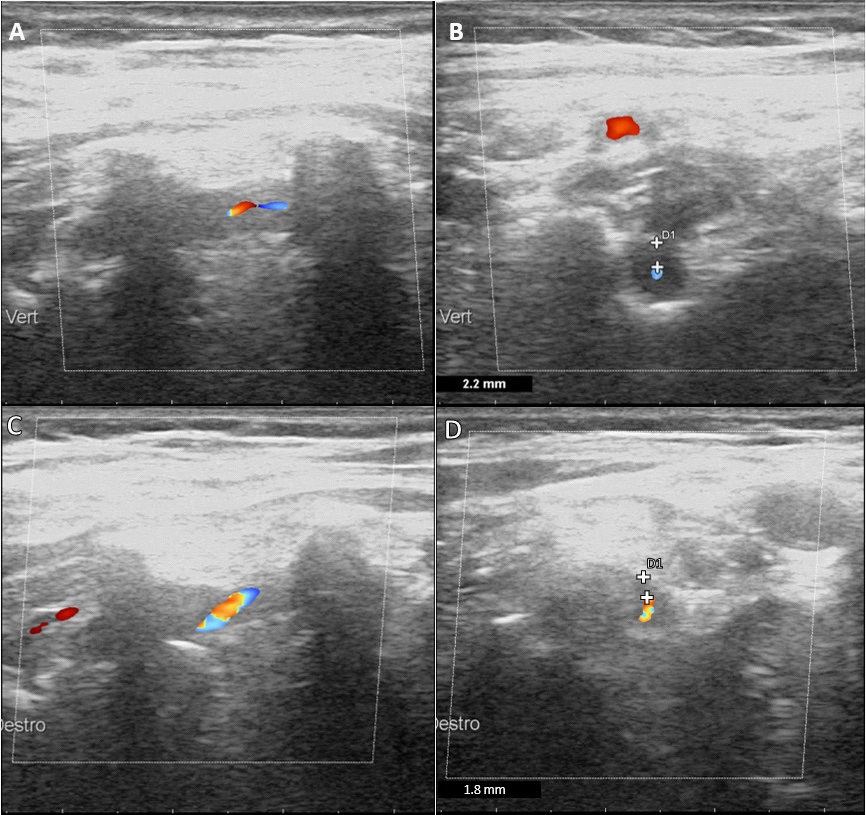
Initial extracranial duplex sonography showed mild atherosclerotic vascular changes with small hyperechogenic plaques in both carotid bifurcations. Instead, the assessment of bilateral V2-VA revealed a turbulent flow with increased flow velocities reaching 180 cm/sec in the right and 186 cm/sec in the left. Both VA showed reduced color filling and a hypoechogenic circumferential vessel wall thickening consistent with a dark halo sign [2], more evident in right V2-VA (Figure 2 A,B). Furthermore, multiple focal stenoses (“String of Beads”) were evident in the left V1-VA.
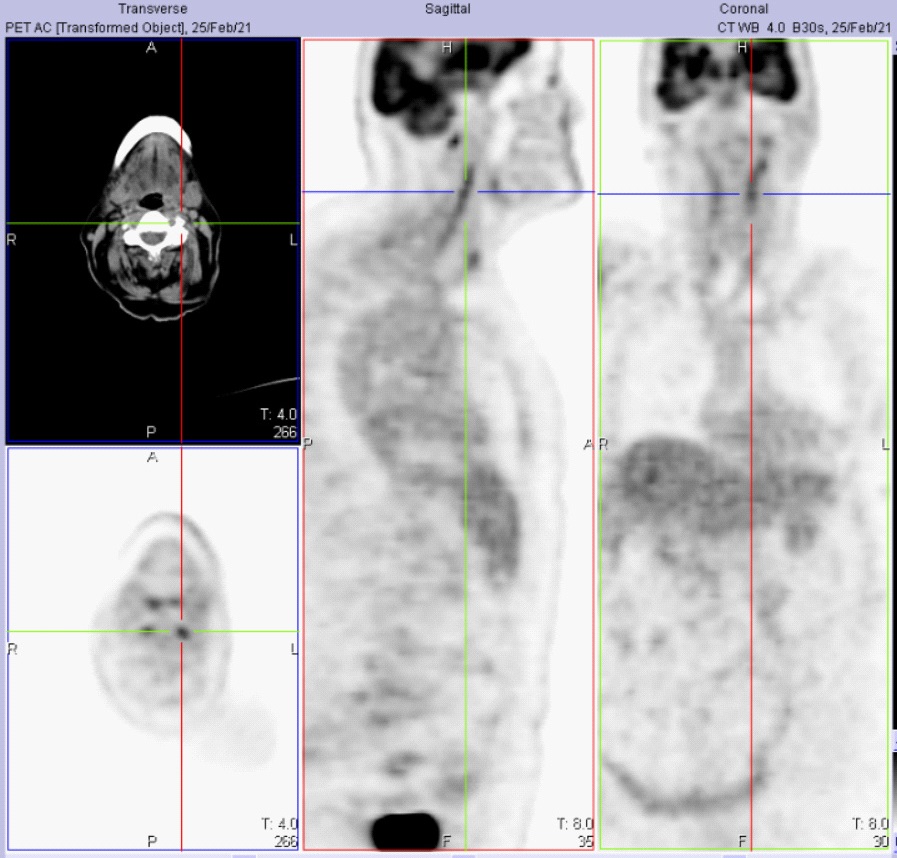
Digital subtraction angiography was not performed and all the other causes of bilateral posterior stroke, including hypoperfusion, were ruled out. Finally, the patient performed a 18FDG-PET, which highlighted increased FDG uptake of inflamed VA walls (Figure 3).
Consequently, a corticosteroid therapy was started (80 mg of 6-Methyl-Prednisolone), inducing an overall clinical improvement at the time of hospital discharge. Follow up ultrasound evaluation after treatment showed a reduction of echographic halo sign (Figure 2 C,D) and flow velocities. The patient’s clinical condition remained good.
Discussion
Isolated bilateral cerebellar stroke is extremely uncommon and only a few cases have been described in the literature [1]. The main clinical neurological manifestations may be cranial neuropathies, neuro-otologic and neuropsychiatric syndromes. The spectrum of symptoms in the event of VA involvement comprises headaches and neck pains but also transient ischemic attack (TIA)/stroke [3,4]. If present, a symmetric involvement of the VA is common in Giant Cell Arteritis (GCA) [5,6]. Indeed, among the intracranial arterial vessels, the VA are the most likely to be affected by GCA, especially in the first tract beyond the point of dural perforation, due to the presence of internal elastic membrane which represents a common target of inflammation.
The most frequent cause of steno/occlusive VA disease is atherosclerosis, as our patient showed in carotid arteries. Indeed, an atherosclerotic etiology had also to be considered, particularly in view of her age and vascular risk profile.
Duplex ultrasound relevantly contributed to the diagnosis of vasculitis, particularly by visualization of the inflammatory vessel wall, which appeared as hypogenic mural thickening.
Differential diagnosis is also with intramural hematoma and dissection, but in these cases the halo sign is in eccentric location, in contrast with arteritis, where it appears mostly concentric. Considering the clinical and radiological diagnosis, a positive dark halo sign had a specificity of 99.5%, and stenoses or occlusion had specificity of 96% [8].
Anyway, the uncommon stroke localization and the ultrasound findings suggested vasculitic etiology of lesions, like GCA. Furthermore, 18-FDG-PET uptake of VA suggested diagnosis of GCA instead of atherosclerosis.
The treatment was corticosteroids therapy (80 mg of prednisolone) until GCA symptoms and acute phase markers resolved [9].
Conclusion
In our case arguments in favor of arteritis, like GCA, were the uncommon site of stroke, the sonographic findings in both V2-VA, the 18FDG-PET pattern and the clinical-sonological improvement after steroid medication.
In conclusion, when isolated bilateral cerebellar stroke appears in clinical practice, VA must be carefully investigated, and it should raise suspicion of GCA.
Acknowledgements: Acknowledge anyone who provided intellectual assistance, technical help (including with writing and editing), or special equipment or materials. Provide information about funding by including specific grant numbers and titles.
References
- John S, Hegazy M, Cheng Ching E, Katzan I. Isolated bilateral middle cerebellar peduncle infarcts. J Stroke Cerebrovasc Dis. 2013; 228: e645-646.
- García-García J, Ayo-Martín Ó, Argandoña-Palacios L, Segura T. Vertebral artery halo sign in patients with stroke: A key clue for the prompt diagnosis of giant cell arteritis. Stroke. 2011; 42: 3287-3290.
- Caselli RJ, Hunder GG, Whisnant JP. Neurologic disease in biopsy-proven giant cell (temporal) arteritis. Neurology. 1988; 38: 352-359.
- Caselli RJ, Hunder GG, Whisnant JP. Neurologic disease in biopsy-proven giant cell (temporal) arteritis. Neurology. 1988; 38: 352-359.
- Wilkinson IM, Russell RW. Arteries of the head and neck in giant cell arteritis. A pathological study to show the pattern of arterial involvement. Arch Neurol. 1972; 27: 378-391.
- Crompton MR. The visual changes in temporal (giant-cell) arteritis. Report of a case with autopsy findings. Brain. 1959; 82: 377- 390.
- Valdueza JM, Schreiber SJ, Roehl JE, Klingebiel R. Neurosonology and Neuroimaging of Stroke. Book. 2008.
- Schmidt WA, Gromnica-Ihle E. Duplex ultrasonography in temporal arteritis. Ann Intern Med. 2003; 138: 609.
- Mackie SL, Dejaco C, Appenzeller S, Camellino D, Duftner C, et al. British Society for Rheumatology guideline on diagnosis and treatment of giant cell arteritis. Rheumatology (Oxford). 2020; 59: e1-e23.