Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Lung adenocarcinoma characterized by multiple thromboemboli was misdiagnosed as primary anti-phospholipid syndrome: A case report
Xiaoxi Shan1 ; Dongxing Liu2 ; Shouchen Lv3 ; Xiaoyan Lin4 ; Mingjie Liu5 ; Qingrui Yang5 *; Ling Zhu4 *
1 Department of Respiratory Medicine, Yantai Yuhuangding Hospital, Yantai 264010, China.
2 Department of Emergency Medicine, Provincial Hospital affiliated to Shandong University, Jinan 250021, China.
3Department of Radiology, Provincial Hospital affiliated to Shandong University, Jinan 250021, China..
4Department of Pathology, Provincial Hospital affiliated to Shandong University, Jinan 250021, China.
*Corresponding Author: Ling Zhu1
& Qingrui Yang2
1 Department of Respiratory Medicine, Yantai Yuhuangding Hospital, Yantai 264010, China.
2 Department of Emergency Medicine, Provincial Hospital affiliated to Shandong University, Jinan 250021, China.
Email: 8253176871@163.com; 105895sdjn@sina.com
Received : Apr 22, 2021
Accepted : May 27, 2021
Published : May 31, 2021
Archived : www.jcimcr.org
Copyright : © Zhu L & Yang Q (2021).
Abstract
Giant Cell Arteritis (GCA) is an immune-mediated vasculitis of large- and medium-sized vessels. Stroke happens in 3% to 7% of the cases, and identifying GCA properly is important because potentially curative treatment exists. We present a case of an 83-year-old woman, in whom suspected GCA was revealed by stroke caused by Vertebral Artery (VA) inflammation. The uncommon stroke localization and the ultrasound findings suggested vasculitic etiology of lesions. 18-Fluorodeoxyglucose-Positron Emission Tomography (18-FDG-PET) confirmed inflamed VA walls. Patient was treated with oral steroids and antiplatelet therapy, with improvement of clinical and ultrasonographic status. In our patient clinical diagnosis of GCA was challenging due to limited classical symptoms. Arguments in favor of arteritis were the symmetric and bilateral involvement of the VA, the sonographic halo sign and atypical stroke localization.
Keywords: Adenocarcinoma; Antibodies; Antiphospholipid; Antiphospholipid Syndrome; Lung neoplasms; Venous thrombosis
Citation: Yang Q, Zhu L, Shan X, Liu D, Lv S. Lung Adenocarcinoma characterized by multiple thromboemboli was misdiagnosed as primary anti-phospholipid syndrome: A case report. J Clin Images Med Case Rep. 2021; 2(3): 1174.
Introduction
It is well known that Anti-Phospholipid Syndrome (APS) is a rare autoimmune disorder characterized by the presence of vascular thrombosis and obstetric complications. It can be accompanied by persistent levels of antiphospholipid antibodies (aPLs). Thrombotic events associated with increased levels of aPLs can be a predictor of malignancy. In the current report, we described a case of lung adenocarcinoma, accompanied by presentation of multiple organ thromboses, who was misdiagnosed as Primary APS (PAPS). We analyzed the complicated process of the misdiagnosis, and recorded the clinical and biochemical data of this patient. It is noteworthy that increase of clinicians’ and patients’ awareness may improve early and correct diagnosis of lung adenocarcinoma and enhance overall survival rate of such patients.
Case report
A 41-year-old female patient was admitted to the Department of Respiratory Medicine, Shandong Qianfoshan Hospital Affiliated to Shandong University, (Jinan, China) on January 18, 2011, and she was suffering from chest pain with hemoptysis. Three months before her admission to our hospital, she frequently suffered from paroxysmal nocturnal dyspnea. The patient’s symptoms were severe since the last week before her admission. She was then admitted to our hospital for hemoptysis of 20 ml and a moderate level of dyspnea without fever. Upon admission, Computed Tomography (CT) of her chest performed before her admission displayed pulmonary infiltrates and a pericardial effusion. According to history of her medical records, there was no abnormality. Three months before her admission to our hospital, the case had an abortion when she was at 10th week of pregnancy.
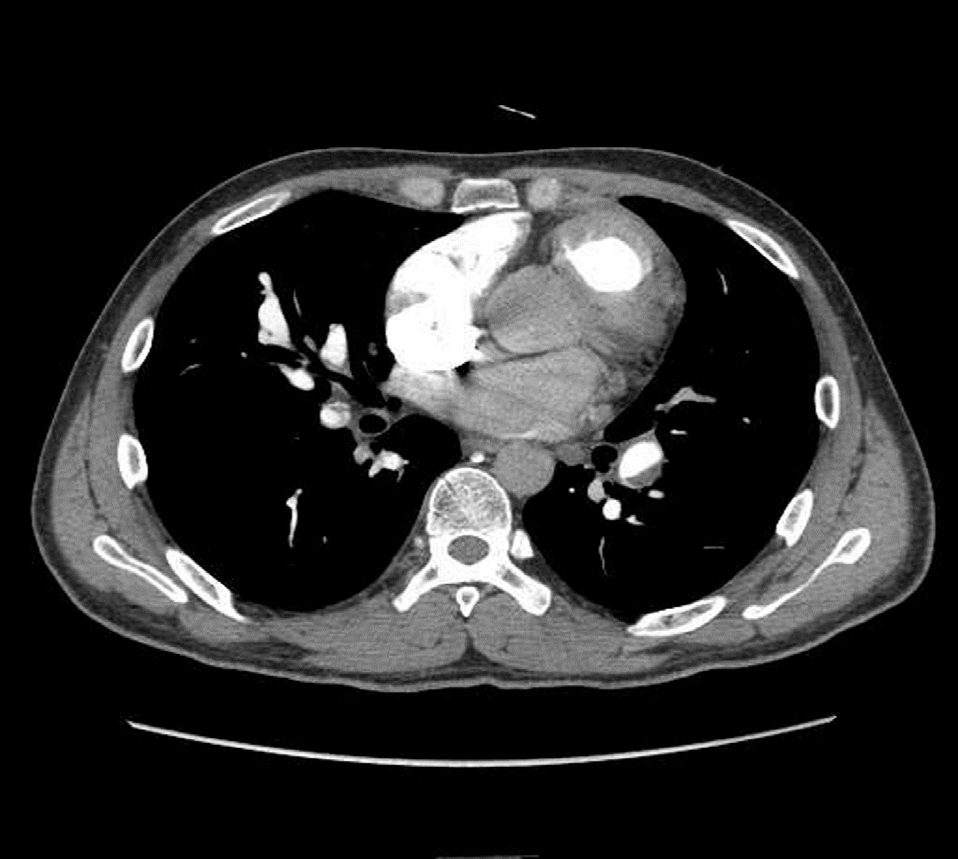
Physical examination upon admission showed no abnormality. The Electrocardiogram (ECG) revealed abnormal Q-wave and the limb leads had a lower voltage compared with her past ECG findings. The CT Pulmonary Angiography (CTPA) indicated “Pulmonary Embolism” (Figure 1). According to the data of ECG and CTPA, we diagnosed pulmonary embolism and prescribed Low-Molecular-Weight Heparin (LMWH) for anticoagulation.
Two hours after her admission, her chest pain was aggravated suddenly, with a severe pain in her left chest. The secondary ECG showed dynamic changes compared with the primary ECG. The patient was highly suspected of suffering from the acute Anterior Myocardial Infarction (AMI). Then, the laboratory examinations were conducted, and they revealed the elevated levels of high-sensitive cardiac troponin T (HS-TnT), Creatine Kinase-Myocardial Band (CK-MB), Myoglobin (MYO), and BType Natriuretic Peptide (BNP). We also conducted short-term dual-antiplatelet treatment with aspirin and clopidogrel. The next ECG revealed the course of acute AMI. Five days after her admission, the patient was found with the loss of a sudden attack of consciousness and focal seizures in her both eyes. The muscle strength of the left upper and bilateral lower extremities was grade 4, while that of the right upper one was grade 0. The muscle tone of the right extremities was lowered, whereas that of the left ones was normal. Babinski sign was positive. The emergency Magnetic Resonance Imaging (MRI) showed the multi-infarction nidi at the cerebral hemispheres and the right cerebellar hemisphere (acute period), and the marked lesion was located at the right cerebellar hemisphere (large hemispheric infarction). She was given dehydration and diuresis therapy.
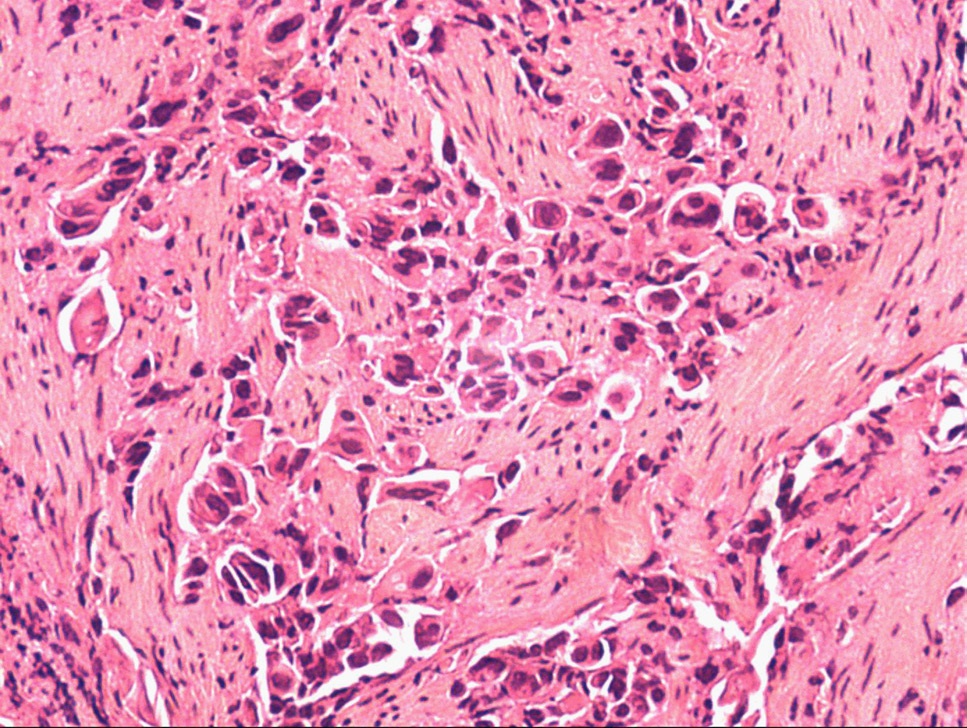
Laboratory examination on admission indicated the elevated levels of C-Reactive Protein (CRP) (25.4 mg/l), D-dimer (1226.4 ug/ml), anti-β(2)-glycoprotein I (anti-β(2)GPI) (43.91 RU/ml), anticardiolipin (aCL) antibody of immunoglobulin G (IgG) (48.76 U/ml), and aCL antibody of immunoglobulin M (IgM) (43.83 U/ml). Besides, antinuclear antibodies (anti-double stranded DNA (anti-dsDNA) and anti-SM), anti-nucleosome antibodies, anti-myeloperoxidase antibodies, Rheumatoid Factor (RF), AntiStreptolysin O (ASO), and platelet count were all within normal limits. According to the results of clinical and laboratory examinations, the patient was diagnosed with APS. We continued to prescribe anticoagulants and glucocorticoids. The clinical symptoms improved, while hemoptysis occurred occasionally. The patient was self-discharged from our hospital, and then, she was re-admitted for CT of her chest. The CT of chest showed hilar enlargement. After that, the fiberoptic bronchoscopy was carried out, which indicated lung adenocarcinoma (Figure 2).
Discussion
Increased incidence of aPLs antibodies has been reported in patients with malignancies. Patients diagnosed with lung adenocarcinoma have demonstrated a high prevalence of aPLs [1]. The presence of aPLs in patients with malignancies is associated with important implications in their treatment and prognosis [2]. LAC is associated with the increased risk of thrombosis in lung adenocarcinoma patients and is correlated with decreased survival rate [3]. In a larger cohort of patients with positive aCL, the risk of cancer-related mortality was elevated by 2.6-fold [4]. APLs are significantly prevalent in patients with infectious diseases, autoimmune disorders, malignant diseases, and even in healthy elderly individuals. The correct diagnosis of APS is therefore a main challenge. In case of under-diagnosis of APS, negative consequences may appear owning to the uncontrolled thrombosis. However, over-diagnosis of APS can lead to missed and delayed diagnosis. The core question is that, can a patient be diagnosed with APS based exclusively on the presence of positive aPLs? According to the criteria presented for diagnosing APS, aPLs must be detected on 2 occasions with interval of < 12 weeks to determine the presence of APS. The criteria for the laboratory diagnosis of APS have still remained controversial. At present, there are at least 4 different method reporting the results of the testing of aCL and anti-β2GPI [5]. Whether patients with similar clinical manifestations, while with different patterns and combinations of positive aPLs test results should be considered equivalent is another challenge. LA showed the highest level of association with thrombotic complications and IgG, whereas IgM aB2GPI was not correlated with unprovoked thrombotic events [6]. Additionally, clinical manifestations, such as deep vein thrombosis and early miscarriages are frequent in the general population. Hence, the presence of aPLs for diagnosing APS may be misleading [7].
A previous study suggested that pulmonary embolism and deep venous thrombosis can be taken as predictors of an occult malignancy into account [8]. The association between aPLs and thrombotic events was confirmed in several prospective studies and in animal models [9]. The pathogenic role of aPLs in the development of thrombosis remains elusive. It was reported that acute coronary syndrome in very young patients may represent a thrombotic event independent of underlying atherosclerotic disease. APLs are well-known risk factors for thrombosis, loss of pregnancy, and other clinical manifestations of APS [10]. Moreover, aPLs have been reported as the most important risk factor for arterial and venous thromboses, stroke, and myocardial infarction. In 2012, Otomo et al. pointed out that aPL-S is a useful quantitative index for diagnosing APS and may be a predictive marker for thrombosis in autoimmune diseases [11]. Any patients who are suspected of having APS should be visited by a multidisciplinary team of specialists for making a correct diagnosis [12]. When the underlying malignancy is in remission, the aPLs can be reduced or even disappeared [13].
In recent years, a great number of scholars have suggested that aPLs cannot merely cause thrombosis and become pathogenic when a second factor is present. The “two-hit hypothesis” has been proposed, in which aPL (the first hit) can only exert their prothrombotic influence in the presence of another thrombophilic condition (the second hit) [14]. According to the “two-hit hypothesis”, the lung cancer is the “second hit” that triggers thrombosis in the case mentioned above.
In the current case report, a 41-year-old woman with an abortion history was investigated. Although measurements of aPLs could not be repeated after 12 weeks, the presence of aPLs in association with thrombotic phenomena is highly suggestive of APS. In this patient, there was a challenge to determine whether the she was suffering from APS or whether the presence of aPLs could be a paraneoplastic phenomenon. However, this distinction may have a slight influence on the treatment plan. Long-term anticoagulation should be taken in case of the existence of thrombotic events and elevated levels of aPLs.
Conclusion
This case report highlighted the need for early diagnosis and effective treatment of cancer patients with positive aPLs. Diagnosis and differential diagnosis of cases who are suspected of having APS require the collaboration of a multidisciplinary team of specialists. Hence, the early diagnosis and treatment of hidden cancers can significantly reduce the rates of mortality and morbidity in cancer patients with positive aPLs.
References
- Islam M A. Antiphospholipid antibodies and antiphospholipid syndrome in cancer: Uninvited guests in troubled times[C]// Seminars in cancer biology. Academic Press. 2020; 64: 108-113.
- Gómez-Puerta, José A, et al. Antiphospholipid antibodies associated with malignancies: clinical and pathological characteristics of 120 patients. Seminars in arthritis and rheumatism. WB Saunders. 2006. 35; 5.
- Galli, Monica, et al. Lupus anticoagulants are stronger risk factors for thrombosis than anticardiolipin antibodies in the antiphospholipid syndrome: A systematic review of the literature. Blood. 2003; 101: 1827-1832.
- Endler, Georg et al. Anti-cardiolipin antibodies and overall survival in a large cohort: preliminary report. Clinical chemistry. 2006; 52: 1040-1044.
- Pierangeli SS, et al. Criteria’aPL tests: Report of a task force and preconference workshop at the 13th International Congress on Antiphospholipid Antibodies, Galveston, Texas, April 2010. Lupus. 2011; 20: 182-190.
- Gardiner C, et al. Diagnosis of antiphospholipid syndrome in routine clinical practice. Lupus. 2013; 22: 18-25.
- Andreoli, Laura, and Angela Tincani. Beyond the syndrome: antiphospholipid antibodies as risk factors. Arthritis & Rheumatism. 2012; 64: 342-345.
- Font C, Vidal L, Espinosa G, et al. Solid cancer, antiphospholipid antibodies, and venous thromboembolism[J]. Autoimmunity reviews. 2011; 10: 222-227.
- Mahaum N, Prete PE: Antiphospholipid antibodies in malignancy: Are these pathogenic or epiphenomena?. JCR: Journal of Clinical Rheumatology. 2011; 17: 429-431.
- Mahaum, Nayab, and Pamela E. Prete. Antiphospholipid antibodies in malignancy: Are these pathogenic or epiphenomena?. JCR: Journal of Clinical Rheumatology. 2011; 17: 429-431.
- Roubey RAS. Risky business: The interpretation, use, and abuse of antiphospholipid antibody tests in clinical practice. Lupus. 2010; 19: 440-445
- Cohen, Danielle, et al. Diagnosis and management of the antiphospholipid syndrome. Bmj. 2010; 340: 1125-1132.
- Tincani A, Taraborelli M, Cattaneo R. “Antiphospholipid antibodies and malignancies.” Autoimmunity reviews. 2010; 9: 200-202.
- Meroni, Pier Luigi, et al. Pathogenesis of antiphospholipid syndrome: understanding the antibodies. Nature Reviews Rheumatology. 2011; 7: 330-339.