Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Overexpression of SOX2 is associated with poor prognosis in human breast cancer
Raquel C Rodrigues1 ; Camila P Almeida2 ; Milena C M Oliveira2 ; Enio Ferreira2 ; Thais S Ribeiro2 ; Isabella T Borges2 ; Hipacia W Gomes3 ; Cleida A Oliveira3 ; Helen L Del Puerto2 *; Almir S Martins1
1Program in Health and Development in the Midwest Region of UFMS, Campo Grande, Belo Horizonte, Brazil.
2Department of General Pathology, Federal University of Minas Gerais, Belo Horizonte, Brazil.
3Department of Morphology, Federal University of Minas Gerais, Belo Horizonte, Brazil.
*Corresponding Author: Helen L Del Puerto
Department of General Pathology, Institute of Biological Sciences, Federal University of Minas Gerais. Av.
Antônio Carlos, 6627, Pampulha, 31270-901 Belo
Horizonte, MG, Brazil
Email: helendelpuerto@ufmg.br
Received : Apr 26, 2021
Accepted : Jun 01, 2021
Published : Jun 04, 2021
Archived : www.jcimcr.org
Copyright : © Puerto HLD (2021).
Abstract
SOX2 is a transcription factor that activates or suppresses genes involved in cellular differentiation, proliferation, and apoptosis. The deregulation of gene expression programs can lead to cancer initiation, promotion, and progression. The present study investigated SOX2 immunolocalization and expression in Invasive Ductal Carcinoma (IDC), and its correlation with clinical-pathological characteristics of the tumor. Immunohistochemical expression of SOX2 was evaluated in 19 cases of IDC and correlated with clinical-pathological tumor features. To investigate the correlation of SOX2 expression in IDC with other characteristics of human breast cancer, the same samples were also stained with immunohistochemical prognostic panels (estrogen and progesterone receptors, and HER-2). SOX2 overexpression was observed in 44% of the cases. Higher SOX2 expression was correlated with the worst lymph node status (p<0.001) and with positive HER-2 immunostaining (p<0,001). The present study demonstrated the association of SOX2 and tumors with worse TNM staging, as well as its overexpression in positive HER-2 tumors. In conclusion, SOX2 was identified as a potential biomarker for poor prognosis of human breast cancer. Further studies with higher patient numbers are necessary to investigate and help clarify the mechanisms underlying this association.
Keywords: SOX2; HER-2; Breast cancer; IHC; Invasive ductal carcinoma; Prognosis.
Citation: Rodrigues RC, Almeida CP, Oliveira MCMD, Ferreira E, Ribeiro TS, et al. Overexpression of SOX2 is associated with poor prognosis in human breast cancer. J Clin Images Med Case Rep. 2021; 2(3): 1182.
Introduction
Breast cancer is increasingly understood to be a heterogeneous disease. Morphologically similar breast tumors may present distinct molecular profiles that are not evident using routine histopathological examination. Consequently, the unique genetic and molecular patterns of each tumor may go undetected [1,2]. Although breast cancer diagnoses are mostly histopathological, auxiliary diagnostic tests exist to support it, as well as cancer classification, prognosis, and response to therapy.
The simple morphological classification used for breast cancer categorization is insufficient for describing these tumors. Identification and differentiation of molecular phenotypes in breast cancer through immunohistochemistry profiling is advantageous for determining cancer prognosis. The expression of these markers is important in choosing adequate treatment for each cancer type [3,4].
The study of tumor markers makes it possible to better comprehend the molecular and cellular basis of cancer initiation and progression [5]. SOX family proteins are a conserved group of transcription regulators defined by the presence of a highly conserved High-Mobility Group (HMG) domain that mediates DNA binding. The members of the SOX family may act as oncogenes, tumor suppressor genes, or both, depending on the ongoing cellular context. They can also be turned on or off by a variety of genetic and epigenetic mechanisms, including alterations in DNA copy numbers and methylation status, or by miRNA aberrant expression [6,7].
Sex-determining region Y-box transcription factor 2 (SOX2) is an embryonic transcription factor gene located on the 3q26.3-q27 chromosome that encodes a high mobility group protein, which plays an essential role in maintaining the differentiation potential and self-renewing feature of embryonic stem cells [8].
Studies from the past few years have demonstrated that SOX2 protein participates in apoptosis inhibition and promotion of cell proliferation, mediating tumor aggressiveness. This protein has played the same roles in breast cancer, in which SOX2 is suggested to be overexpressed [9].
Therefore, the present study aimed to evaluate SOX2 expression and immunolocalization in invasive ductal carcinoma (IDC) samples from female patients, as well as correlating its expression to literature-validated predictive and prognostic factors for breast cancer.
Material and methods
Patient samples
The cohort of the study was composed of female patients (n=19) diagnosed with breast cancer, registered on the Mastology Clinic of Maria Aparecida Pedrossian University Hospital at Campo Grande, Mato Grosso do Sul, Brazil.
Paraffin-embedded samples IDC tissue blocks without any other specifications [10], were obtained after histopathological examination of the surgical specimen, followed by immunohistochemistry analysis for diagnostic confirmation. The tumors were staged according to TNM classification for malignant tumors and then classified by their molecular subtype [10]. The Ethical Committee approved this study for Human Research of Federal University of Mato Grosso do Sul (document number 3.723.300), and informed consent forms were collected from all patients.
SOX2 expression and immunolocalization by immunohistochemistry
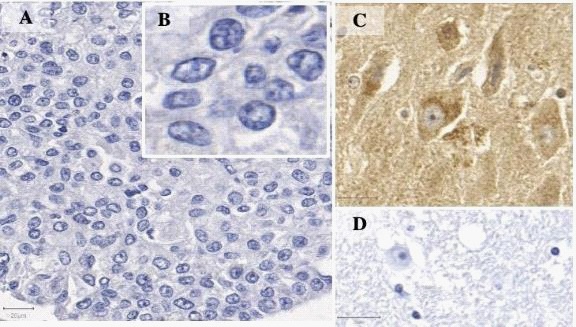
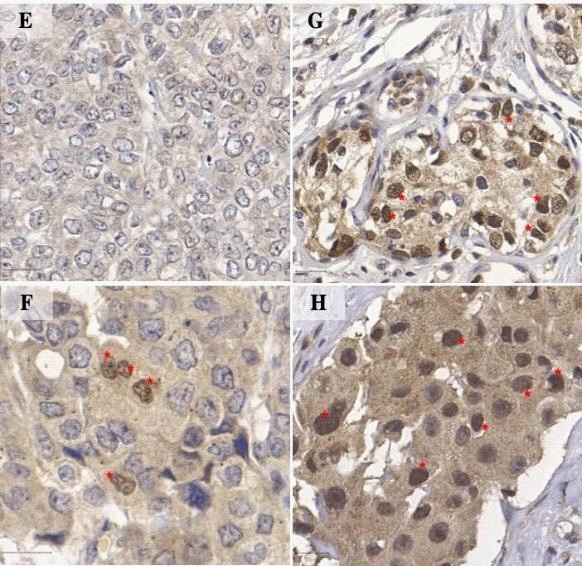
Immunohistochemistry was performed according to the peroxidase reaction technique, using a biotinylated secondary antibody for target identification (Polyclona Rabbit Anti-Goat Immunoglobulin Biotinylated; Dako North America; Via Real Carpinteria, CA, USA), and avidin-biotin detection kit (ABC Kits; Vector Lab’s VECTASTAIN; Burlingame, California USA). Heatinduced antigen retrieval was obtained under pressurized humid heat (134°C in autoclave), using a citrate buffer solution at pH 6.0. For endogenous peroxidase blockage, slides were incubated three times for five minutes each in 3% H2 O2 methanol solution. Subsequently, endogenous proteins were blocked by a 20-minute incubation with Protein Block Serum-Free Ready to Use (Dako North America; Via Real Carpinteria, CA, USA). All reagents were manually applied to the samples, which were incubated with primary antibody for 16 hours, followed by a 3-minute incubation with 3’3-diaminobenzidine chromogen (Liquid DAB+Substrate Chromogen system; Dako North America, Via Real Carpinteria, CA, USA). Finally, samples were hematoxylin stained and washed in running water for five minutes. For a negative control, primary antibody incubation was not included on a breast IDC slide (Figure 1A and 1B and human brain tissue Figure 1D); and for positive control, a sample of normal human brain tissue was used (Figure 1C). Analyses were based on SOX2 tissue expression and immunolocalization, and only nuclear staining was considered as positive staining for SOX2. Two protocols for evaluating this transcription factor expression were applied: a protocol that did not grade staining intensity; and a semi-quantitative score protocol (score 1: strong nuclear immunostaining in less than 10% of cancer cells, considered as negative for SOX2 expression; and score 2: strong nuclear immunostaining in more than 10% of cancer cells, considered as positive for SOX2 expression). The counting was performed by a single observer, out of 1000 cells scattered in the slide, with the assistance of a grid. Briefly, using ImageJ grid plug-in, six sections were defined in the images and the upper left, middle bottom and upper right sections were selected to perform the counting until the 1000 cells were analyzed.
Statistical analysis
Descriptive and inferential statistics were applied to data analysis. Shapiro-Wilk’s normality test revealed a parametric pattern of continuous variables. Mean plus or minus standard deviation (mean ± SD) of the variables was calculated for subsequent data description. Categorical variables were descriptive in a number of events and percentages, and their inferential statistics were calculated through the chi-square test. For all analyses, a significance index (SI) of 5% (p<0.05) was assumed.
Correlations between SOX2 expression and breast cancer clinical characteristics
The patients in this study averaged 56.6 years of age, with a range of 36 to 78 years. The highest prevalence of SOX2 positive expression occurred in patients aged 55 to 65 years. When also considering cancer cell vascular invasion, lymphatic embolization, and the presence of metastasis in axillary lymph nodes (Table 1), SOX2 overexpression was statistically significant (p=0.003, p=0.005, and p=0.001, respectively). In addition, subsequent to TNM staging, higher SOX2 expression was observed in patients with more advanced cancer stages (p=0.04). Regarding the expression profile of hormone receptors in the IDC analyzed samples, it was not possible to demonstrate a significant difference between the presence of SOX2 and lack of receptor expression. However, SOX2 was overexpressed in HER-2 positive tumors (p=0.034). There was higher SOX2 expression in subtypes that also overexpressed human epidermal growth factor type 2, HER-2 (p=0.001).
Table 1: Correlation between SOX2 expression and breast cancer Clinical Pathological parameters.
Clinicopathological parameters |
SOX2 high expression (%) |
p value |
Tumor size (cm) < 2 > 2 |
25 75 |
0.163 |
Lymph node metastasis Yes |
50 50 |
0.001 |
Vascular invasion Yes |
62.5 37.5 |
0.003 |
Lymphatic embolization Yes |
75.5 25.5 |
0.005 |
ER* status |
62.5 37.5 |
0.410 |
PR** status |
75 25 |
0.502 |
HER2 |
62.5 37.5 |
0.034 |
*ER: Estrogen receptor; **PR: Progesterone receptor
Discussion
Increasing evidence supports the hypothesis that cancer stem cells, or tumor initiating cells, control and maintain several types of human malignancies [12]. Healthy and cancer stem cells share phenotypes that may reflect activity of common signaling paths such as overexpression of SOX2, among others [13]. The role of SOX2 in organogenesis or breast tissue functioning is still little understood, although in a healthy normal mammary tissue, there is no significant expression of SOX2. In breast cancer cells, on the other hand, this transcription factor was found to be overexpressed by both mRNA by RT-qPCR and western blotting techniques [3]. Corroborating such findings, all IDC samples in this study exhibited strong nuclear SOX2 immunostaining, and SOX2 was overexpressed in almost half of the samples. Data based on research with breast cancer cell lines support an active role for SOX2 during mammary tumorigenesis, in which SOX2 promotes cell proliferation and tumorigenesis in vitro, partially favoring G1/S transition in cell cycle and regulating, alongside β-catenin, the expression of effector genes such as CCND1 [9]. High levels of SOX2 appear to be intimately related to several processes during tumor development, including tumor initiation, invasion, and metastasis. Thus, SOX2 seem to participate in tumor invasion and metastasis, since the expression of this transcription factor correlates to increased tumor size and stage, lymph node metastasis and substantial cancer cell invasiveness [14].
A close association among vascular invasion, lymphatic embolization, and lymph node metastasis was consistently demonstrated in this work. More importantly, a correlation among these markers of poor cancer prognosis and SOX2 expression in patients presenting high TNM staging at the initial diagnosis phase was established. This was mostly due primarily to lymph node status since in this study, the SOX2 transcription factor proved to be overexpressed in samples from patients that had IDC metastasis in their lymph nodes. Approximately 70% to 75% of breast cancer cases express estrogen receptor alpha (ERα), which is associated with a better prognosis, and studies already reported that mammary stem cells express low levels of estrogen receptors or do not express them at all [15]. A previous study indicated that cancer stem cells showing high levels of SOX2 lack or express low levels of estrogen receptors, are more resistant to Tamoxifen and have a less differentiated cell phenotype [16]. Since ER was not included in the current study, it was not possible to examine the relationship between the expression of estrogen receptors and SOX2. Nevertheless, the results consistently identified SOX2 overexpression in HER2 positive tumors. It is well known that 15% to 20% of all breast cancers express HER2 and that this oncogene is directly related to prognosis and therapeutic response, playing a pivotal role in cancer-free overall survival. HER2 expression in IDC samples was concomitant with the expression of the transcription factor SOX2 in the present study. These results partially differ from others which showed significantly higher SOX2 expression in triple-negative tumors from the basal subtype [17]. This discrepancy might derive from the samples used in the previous study that collected tumors from a larger number of patients going through pre-menopause stages, and from patients that carried BRCA1 mutations.
Breast cancer classification falls into three main subtypes, based on the presence or absence of molecular markers for estrogen or progesterone receptors, and for the human epidermal growth factor 2 receptor (HER2). The triple negative breast cancer subtype has a higher recurrence rate than the other two subtypes, associated with an 85% survival rate of five years for stage I cancer, while the subtypes expressing a receptor for hormones and HER2 have a survival rate of 94% to 99%. Additionally, there is a one-year average overall survival for the metastatic triple-negative breast cancer, compared to the five-year survival estimated for the other two subtypes
The results presented in this paper revealed SOX2 overexpression in HER2 positive tumor subtypes, in contrast with the lack of expression of this transcription factor in tumor subtypes expressing hormone receptors. These findings corroborate previous ones that relate SOX2 expression to poorer therapeutic responses, indicating that SOX2 levels are higher in patients that failed to respond to endocrine therapy, as well as in their primary tumors, in comparison to patients that achieved a successful response [18]. To summarize, all the data taken together indicate that the SOX2 transcription factor has a demonstrated potential as a biomarker for prognosis and survival of breast cancer patients and that it must be included in future lists of emerging biomarkers for this type of cancer. Hence, further studies involving SOX2 and breast cancer may provide valuable diagnostic and therapeutic insights for handling this disease
Conclusion
This study succeeded in demonstrating the presence of SOX2 protein in the analyzed IDC samples. More than half of the IDC samples exhibited a positive score for the expression of this transcription factor. Immunohistochemistry revealed SOX2 nuclear expression in cancer cells. Expression of this protein was not evaluated in other cell types from the tumor microenvironment
Importantly, an association of SOX2 and lymph node status was established by displaying the overexpression of this transcription factor in more advanced tumor stages, as well as in HER2 positive tumors. Additionally, SOX2 expression proved to be absent or reduced in patients whose tumors expressed ER and PR hormone receptors.
Acknowledgements: The microscopic data shown in this work was obtained using digital microscope Scanner 3D Histech located in the Center for Acquisition and Processing of Images (CAPI-ICB/UFMG).
Funding statement: This work was supported by Fundação de Amparo à Pesquisa de Minas Gerais (FAPEMIG), Brazil.
Author’s contribution: RCR, ASM, and HLDP designed the study. HLDP, EF, and CPA conceived and planned the experiments. CPA, ITB, and HWG performed the immunohistochemistry. MCMO, CPA, HLDP, RCR, EF, and CAO contributed to the analysis and interpretation of the results. RCR, HLDP, CPA, and TSR drafted and wrote the manuscript. All authors have read and approved the final manuscript.
References
- Barros ACSD, Leite KR. Classificação molecular dos carcinomas de mama: Uma visão contemporânea/Molecular. Revista Brasileira de Mastologia. 2015; 25: 146-155.
- Perou CM, Sorlie T, Eisen MB, Van De Rijn M, Jeffrey SS, Rees CA, et al. Molecular portraits of human breast tumours. Nature. 2000; 17: 747-52.
- Chen C, Yuan JP, Wei W, Tu Y, Yao F, et al. Subtype classification for prediction of prognosis of breast cancer from a biomarker panel: Correlations and indications. International Journal of Nanomedicine. 2014; 9: 1039–1048.
- Cheng YS, Kuo SJ, Chen DR. Sparing sentinel node biopsy through axillary lymph node fine needle aspiration in primary breast cancers. World Journal of Surgica Oncology. 2013; 9: 1039-1048.
- Gobbi H. Classificação dos tumores da mama: Atualização baseada na nova classificação da Organização Mundial da Saúde de 2012. Jornal Brasileiro Patologia Médica Laboratorial. 2012; 48: 463-474
- Castillo SD, Sanchez-Cespedes M. The SOX family of genes in cancer development: biological relevance and opportunities for therapy. Expert Opinion Ther Targets. 2012; 16: 903- 919.
- Dong C, Wilhelm D, Koopman P. Sox genes and cancer. Cytogenetic GenomeResearch. 2004; 105: 442–447.
- Lengerke C, Fehm T, Kurth R, et al. Expression of the embryonic stem cell marker SOX2 in early-stage breast carcinoma. BMC Cancer. 2011; 11: 42.
- Feng X, Lu M. Expression of sex-determining region Y-box protein 2 in breast cancer and its clinical significance. Saudi Medical Journal. 2017; 38; 685–690.
- CARLSON RW, et al. Breast cancer. Journal of the National Comprehensive Cancer Network. 2009; 7: 122-192.
- Graham V et al. SOX2 functions to maintain neural progenitor identity. Neuron. 2003; 39: 749-765.
- Diehn M, Cho RW, Clarke MF. Therapeutic implications of the cancer stem cell hypothesis. Seminars in Radiation Oncology. 2009; 19: 78 – 86.
- Simoes BM, Piva M, Iriondo O, Comaills V, Lopez-Ruiz JA, et al. Effects of estrogen on the proportion of stem cells in the breast. Breast Cancer Research and Treatment. 2011; 129: 23–35.
- Chen Y, Shi L, Zhang L, Li R, Liang J, et al. The molecular mechanism governing the oncogenic potential of SOX2 in breast cancer. Journal Biology of Chemistry. 2008; 283: 17969–17978.
- Clayton H, Titley I, Vivanco M. Growth and differentiation of progenitor/stem cells derived from the human mammary gland. Experimental Cell Research. 2004; 297: 444 – 460.
- Piva M, Domenici G, Iriondo O, Rábano M, Simões BM, et al. Sox2 promotes tamoxifen resistance in breast cancer cells. Molecular Medicine. 2014; 6; 66–79.
- Rodriguez-Pinilla SM, Sarrio D, Moreno-Bueno G, Rodriguez-Gil Y, Martinez MA, et al. Sox2: A possible driver of the basal-like phenotype in sporadic breast cancer. Modern Pathology. 2007; 20: 474–481.
- Wuebben EL, Rizzino A. The dark side of SOX2: Cancer - A comprehensive overview. Oncotarget. 2017; 8: 44917-44943.