Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
An unusual case of delayed and recurrent bleeding after renal biopsy in a patient with malignant hypertension
Jeremy JW Teng; Emmett TY Wong; Christopher CH Leo; Titus WL Lau; Pei Loo Tok*
Division of Nephrology, National University Health System, Singapore.
*Corresponding Author: Pei Loo Tok
Division of Nephrology, National University Health
System, Singapore.
Email: pei_loo_tok@nuhs.edu.sg
Received : Apr 30, 2021
Accepted : Jun 07, 2021
Published : Jun 10, 2021
Archived : www.jcimcr.org
Copyright : © Tok PL (2021).
Abstract
We report a 35-year-old Asian man who presented with symptomatic malignant hypertension with complications of acute kidney injury, thrombocytopenia and microangiopathic haemolytic anemia. A renal biopsy done led to recurrent bleeding needing repeated embolization. We highlight the importance of continued monitoring for post biopsy bleeding even weeks after the biopsy for high-risk cases and discuss the aspects of prevention of severe post biopsy bleeding.
Keywords: Renal biopsy; Malignant hypertension; Embolization; Desmopressin.
Citation: Teng JJW, Wong ETY, Leo CCH, Lau TWL, Tok PL. An unusual case of delayed and recurrent bleeding after renal biopsy in a patient with malignant hypertension. J Clin Images Med Case Rep. 2021; 2(3): 1188.
Introduction
Percutaneous ultrasound guided native renal biopsy is a common procedure done to obtain tissue for histological diagnosis of kidney diseases. Complications such as bleeding, infection and loss of kidney are uncommon [1]. Severe bleeding needing intervention is rare at 0.6% [1] and 91-98% of these cases happen within 24 hours post biopsy [2,3]. We report an unusual case which required repeated interventions to stop bleeding even weeks after the renal biopsy.
Case presentation
A 35-year-old man with past medical history of hypertension and non-adherence to antihypertensives presented with malignant hypertension and acute kidney injury. Blood pressure was 230/130 mmHg. Features of thrombotic microangiopathy such as hemolytic anemia and thrombocytopenia were present. ADAMTS 13 activity, 24-hour urine phaeochromocytoma screen, 24-hour urine cortisol, renin and aldosterone ratio were unremarkable (Table 1). Creatinine rose from 120 µmol/L on admission to 1123 µmol/L in 2 weeks despite improved blood pressure to 160 systolic. His platelet count rose from 11 x 109 /L to 150 x 109 /L over 2 weeks with only 4 units of platelet given. His right kidney was 12.5 cm; left kidney was 12.1 cm, both showing echogenicity on ultrasound. There was no renal artery stenosis. Auto-antibodies for systemic lupus erythematosus, ANCA, AntiGBM were unremarkable.
Table 1: Investigations for etiology of young-onset hypertension and kidney injury.
Urine Formed Element |
/HPF |
5 |
0-4 |
Thyroxine, Free |
pmol/L |
11.1 |
8.0-16.0 |
TSH |
mIU/L |
1.60 |
0.45-4.50 |
Renin, plasma |
ng/mL/h |
0.6 |
<= 0.6-4.3 |
Aldosterone |
ng/dL |
< 4.0 |
<= 21 |
Cortisol, U Free 24h |
nmol/day |
40 |
59-413 |
Dopamine, U |
nmol/L |
49 |
|
Dopamine, U 24h |
nmol/day |
46 |
424-2612 |
Metanephrine, U |
nmol/L |
279 |
|
Metanephrine, U 24h |
nmol/day |
263 |
325-1530 |
Normetanephrine, U |
nmol/L |
700 |
|
Normetanephrine. U 24h |
nmol/day |
659 |
885-2880 |
Anti-Nuclear Ab |
|
<1:80 |
<1:80 Negative |
Anti-ds-DNA |
IU/mL |
< 3 |
<100 Negative |
Complement 3 |
mg/dL |
90 |
85-185 |
Complement 4 |
mg/dL |
11 |
10-50 |
Anti-GBM |
U/mL |
< 20.0 |
< 20 Normal |
Anti-Myeloperoxidase |
U/mL |
1 |
<20 No antibody detected |
Anti-PR3 |
U/mL |
<1 |
<20 No antibody detected |
Rheumatoid Factor |
IU/mL |
< 10 |
0-15 |
ADAMTS-13 Activity |
IU/mL |
0.59 |
>= 0.65 |
At the time of renal biopsy, urea was 22.8 mmol/L; platelet count was 150 x 109 /L; haemoglobin 8.7 g/dl. Prothrombin time and partial thromboplastin time were normal. Blood pressure was 135/98 mmHg. He was not on antiplatelet or anticoagulant. Three cores of kidney tissue measuring 1-1.5 cm were obtained using 18 G coaxial biopsy of lower pole of left kidney. One hour after the biopsy, he developed left flank pain with hypotension. BP was 80/40 mmHg. Hemoglobin was 6 g/dl. He was given 2 units of packed red cells.
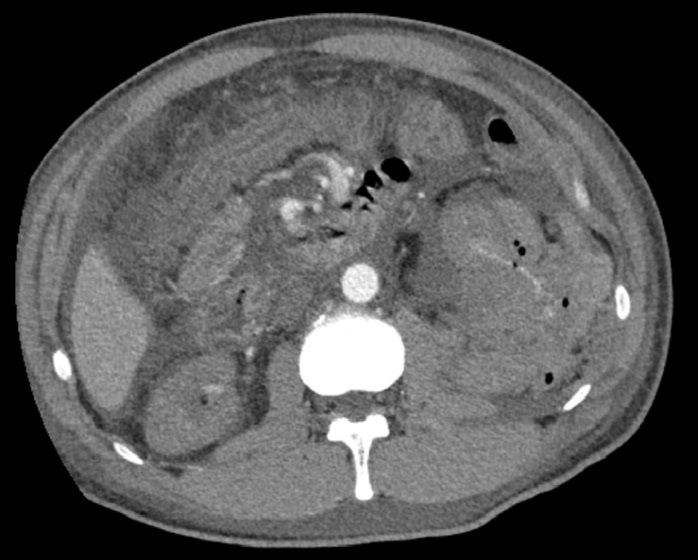
Computed Tomography (CT) angiogram revealed active arterial hemorrhage from the lower pole of the kidney (Figure 1). Angioembolization of the left renal artery was performed with two interlock coils and a small amount of gelfoam. Histology showed hypertensive nephrosclerosis with changes suggestive of malignant hypertension including thrombotic microangiopathy. He was subsequently initiated on hemodialysis via a tunneled dialysis catheter due to worsening kidney function. He also had difficult to control hypertension from the page kidney that developed as a result of the post biopsy hematoma and required five anti-hypertensives (Clonidine 50 mcg TDS, Carvedilol 12.5 mg BD, Irbesartan 300 mg OD, Hydralazine 100 mg TDS, Nifedipine LA 60 mg BD). Due to recurrent fluid overload partially contributed by fluid indiscretion, he needed additional hemodialysis sessions for ultrafiltration. His systolic blood pressure regularly fluctuated between 110 to 170 mmHg.
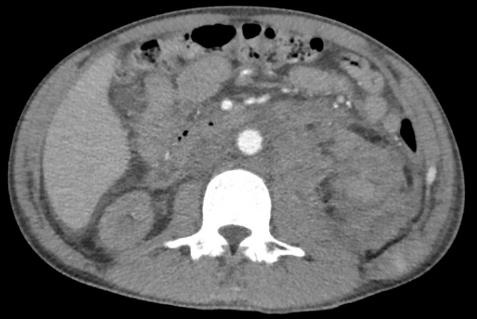
Three weeks post renal biopsy, he developed left flank pain with drop of hemoglobin to 5.6 g/dl. CT angiogram showed increase in size of the left retroperitoneal hematoma and a pseudoaneurysm in the left posterolateral abdominal wall (Figure 2). On-table angiogram demonstrated a bleeding point at lower pole of left kidney. Coil embolization was performed to lower pole vessels. As hemoglobin continued to drop, thrombin injection of the left posterolateral abdominal wall pseudoaneurysm was performed.
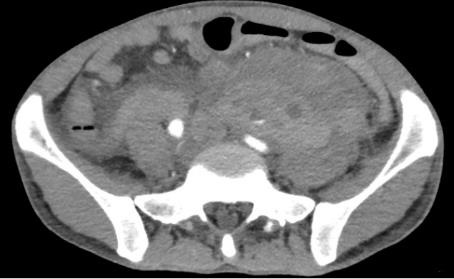
Five weeks post renal biopsy, he developed left flank pain again. Haemoglobin was 4 g/dL. A repeat CT angiogram (Figure 3) revealed significant increase in size of the left retroperitoneal hematoma, with new extension into the pelvis. Active contrast extravasation was seen in the hematoma, although no definite feeding vessel was identified. A larger left perinephric hematoma was also seen. On table angiogram revealed active bleeding from the proximal segment of the left internal iliac artery, which was embolized with coils and gelfoam.
Throughout these bleeding episodes, platelet count and coagulation profile were within normal limits and adequate dialysis was performed. Hematologist was consulted and no other bleeding diathesis was discovered except for kidney failure related platelet dysfunction. Blood pressure control was intensified to keep it less than 130 systolic constantly and additional hemodialysis was done to keep patient at dry weight. Subsequently the hematoma stabilized with no further rebleeding episodes.
Discussion
According to a large systemic review, risk factors for bleeding include use of 14-gauge needle (versus 16- or 18- gauge needle), higher mean creatinine more than 176 µmol/L, baseline hemoglobin < 12 g/dl [1]. Trends towards increased bleeding risk were observed where mean age more than 40 years old and systolic blood pressure more than 130 mmHg [1].
Risk factors for bleeding in our patient include severe renal impairment and its associate platelet dysfunction, anemia and poorly controlled hypertension. While delayed bleeding [4] and page kidney [5,6] post renal biopsy has been reported, recurrent bleeding needing repeated intervention is unusual. In our patient, the post biopsy hematoma resulted in page kidney which exacerbated hypertension and increases risk of rebleeding. Page kidney can increase blood pressure via activation of Renin Angiotensin-Aldosterone System (RAAS) and use of RAAS blockers is recommended [7].
Limited studies that evaluate the use of desmopressin to prevent bleeding post renal biopsy suggest it may decrease hematoma size but did not reduce interventions [8-9]. Due to concerns of hyponatremia [10], further trials are suggested prior to routine usage [11]. In addition, injecting hemostatic matrix in the path of renal biopsies has been used to treat bleeding [12] and to prevent post biopsy bleeding [13].
Although post renal biopsy imaging is frequently performed, studies have yet to demonstrate that post biopsy hematoma detected on ultrasound correlates with clinically significant complications [14]. A negative imaging post biopsy has high negative predictive value for major bleeding complications [14].
Conclusion
Recurrent bleeding post renal biopsy is unusual. Patients with high-risk features such as poorly controlled hypertension, renal impairment and platelet dysfunction are high risk of this unique complication. Multipronged approach including blood pressure optimization and correcting platelet dysfunction is needed. Methods with limited evidence such as gelfoam and sometimes desmopressin may be beneficial but need further study.
References
- Corapi KM, Chen JL, Balk EM, Gordon CE. Bleeding complications of native kidney biopsy: A systematic review and meta-analysis. Am J Kidney Dis. 2012; 60: 62–73.
- Whittier WL, Korbet SM. Timing of complications in percutaneous renal biopsy. J Am Soc Nephrol. 2004; 15: 142–147.
- Prasad N, Kumar S, Manjunath R, Bhadauria D, Kaul A, et al. Real-time ultrasound-guided percutaneous renal biopsy with needle guide by nephrologists decreases post-biopsy complications. Clin Kidney J. 2015; 8: 151–156.
- Y Takeuchi, Y Ojima, S Kagaya, S Aoki, T Nagasawa. Unexpected delayed bleeding after native renal biopsy: A case report. CEN Case Rep. 2018; 7: 9-12.
- Atsushi Ono, Tsutomo Inoue, Koji Tomori, Hirokazu Okada. Page kidney following renal biopsy. Intern Med 2017; 56: 875.
- Rodrick C. Zvavanjanja, Aaron Sean ashton. Page kidney secondary to subcapsular haematoma following percutaneous renal allograft biopsy. Radiol case rep. 2018; 13.
- Dopson, Shirley J et al. Page Kidney as a Rare Cause of Hypertension. Case Report and Review of the Literature. American Journal of Kidney Diseases. 54: 334–333.
- Stratta P, Canavese C, Marengo M, Mesiano P, Besso L, et al. Risk management of renal biopsy: 1387 cases over 30 years in a single centre. Eur J Clin Invest. 2007; 37: 954–963.
- Manno C, Bonifati C, Torres DD, Campobasso N, Schena FP. Desmopressin acetate in percutaneous ultrasound-guided kidney biopsy: A randomized controlled trial. Am J Kidney Dis. 2011; 57: 850–855.
- Lim CC, Siow B, Choo JCJ, Chawla M, Chin YM, et al. Desmopressin for the prevention of bleeding in percutaneous kidney biopsy: Efficacy and hyponatremia. Int Urol Nephrol. 2019; 51: 995-1004.
- Lim CC, Tan HZ, Tan CS, Healy H, Choo JCJ, et al. Desmopressin acetate (DDAVP) to prevent bleeding in percutaneous kidney biopsy: A systemic review. Intern Med J. 2020.
- Jain V, Gupta A, Gulia A, Singhal M, Gulati SS. et al. Gelatinthrombin hemostatic matrix injection to salvage refractory postrenal graft biopsy bleed. Indian J Nephrol. 2013; 23: 149–152.
- Antonio Rahal Junior, Priscila Mina Falsarella et al. Injecting hemostatic matrix in the path of biopsies: Efficacy, potential complications, and the management of such complications Radiol Bras. 2018; 51: 102–105.
- Injecting hemostatic matrix in the path of biopsies: Efficacy, potential complications, and the management of such complication Jonathan J. Hogan, Michaela Mocanu, and Jeffrey S. Berns. The native kidney biopsy: Update and evidence for best practice. Clin J Am Soc Nephrol. 2016; 11: 354-362.