Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Co-infection with nocardia and mycobacterium tuberculosis in a patient with systemic lupus erythematosus: A case report
Zhongji Jiang1; Zhikun Xu2; Jinsong Wu1; Yuemei Lu1; Jine Chen1; Yong Xu3; Xueyan Liu2*
1Department of Laboratory Medicine, The Second Clinical Medical College of Jinan University, Shenzhen People’s Hospital, China.
2Department of Intensive Care Unit, The Second Clinical Medical College of Jinan University, Shenzhen People’s Hospital, China.
3Molecular Diagnosis Center, Shenzhen Second People’s Hospital, China.
*Corresponding Author : Xueyan Liu
Department of Intensive Care Unit, The Second
Clinical Medical College of Jinan University, Shenzhen People’s Hospital, China.
Email: liuxueyan100916@163.com
Received : May 05, 2021
Accepted : Jun 15, 2021
Published : Jun 18, 2021
Archived : www.jcimcr.org
Copyright : © Liu X (2021).
Abstract
Objective: Pulmonary infection is an important cause of morbidity and death in immunosuppressed patients. Recent studies showed that infections caused by Nocardia are quite rare, especially mixed infections with Nocardia and Mycobacterium Tuberculosis (MTB). This case emphasizes the importance of being alert to the possibility of co-infection in immunosuppressed patients.
Case description: A 31-year-old female patient presented to the outpatient clinic with recurrent fever and cough. She had also suffered from Systemic Lupus Erythematosus (SLE) and type V lupus nephritis for 8 years. Additionally, she tested positive for the Nocardia nova complex and MTB following culture of the sputum/bronchoalveolar lavage fluid (BALF) and lung tissue. Initial Computed Tomography (CT) showed a bi-pulmonary massive high-density shadow with multiple cavities, Ground-Glass Opacity (GGO) and multiple miliary nodules. After admission, the patient was given Cefoperazone-Sulbactam (CSL) for empiric treatment and also received low-flow oxygen therapy. Four days later, microscopic examination suggested infection with Nocardia and CSL was replaced with Trimethoprim/Sulfamethoxazole (SXT), Moxifloxacin (MFX) and Imipenem (IPM). On the 9th day, the Nocardia nova complex was confirmed although there was a significant improvement in pulmonary symptoms and imaging results. However the images showed that there was no significant change in GGO and miliary nodules. Moreover, 18 days later she was confirmed with MTB infection and was subsequently transferred to the infectious diseases hospital for further treatment.
Conclusion: Immunosuppressed patients have higher morbidity and mortality after opportunistic infection with pulmonary pathogens such as Nocardia and MTB. Therefore, early detection of pathogens and selection of appropriate antimicrobial therapy can significantly improve the prognosis.
Keywords: Immunosuppression; nocardia nova complex; mycobacterium tuberculosis; co-infection
Citation: Liu X, Jiang Z, Xu Z, Wu J, Lu Y, et al. Co-infection with nocardia and mycobacterium tuberculosis in a patient with systemic lupus erythematosus: A case report. J Clin Images Med Case Rep. 2021; 2(3): 1191.
Background
The Coronavirus Disease 2019 (COVID-19) which causes pulmonary infection initially broke out in Wuhan, China, in December 2019. The disease mainly manifests in the form of fever and cough and has spread across the globe, making it a public health emergency. However, fever and cough are more commonly associated with pneumonia caused by bacteria or fungi, in the elderly, patients with diabetes mellitus, those with HIV/AIDS, pregnant women and immunosuppressed individuals. In addition, pulmonary infection is an important cause of morbidity and mortality in immunosuppressed individuals. On the other hand, Nocardia is a genus of Gram-positive bacteria, characterized by aerobic growth and can be observed under a microscope through modified acid-fast staining because their cell walls contain mycelial acid. Nocardia mainly exists in the soil, water and organic residues and infection arises from inhalation of the bacteria through the respiratory tract [3]. Moreover, the clinical symptoms and imaging characteristics of infection with Nocardia are similar to those of pulmonary tuberculosis , nontuberculous mycobacteria and actinomycetes. These symptoms mainly include fever, cough and expectoration. Nocardia can also cause extrapulmonary infections such as skin abscess, endophthalmitis and brain abscess. Notably, the radiographic findings mainly include abscess, cavity, Ground-glass opacity and miliary changes. Due to these characteristics, Nocardia is also known as the great masquerader as it excellently camouflages as tuberculosis [4-8]. According to current studies, Coinfection with tuberculosis and Nocardia occurs in about 1% of the population, while the co- infection rate among HIV patients ranges from 3% to 6.25 [6,9]. Furthermore, thousands of cases of Nocardia infections are reported worldwide each year, with a mortality rate of approximately 30%-50% [10-12]. It is also noteworthy that co-infection with Nocardia and Mycobacterium tuberculosis (MTB) is more dangerous and therefore deserves more attention. Consequently, the present study reports a case of pulmonary co-infection with the Nocardia nova complex and MTB [6,9].
Case report
A 31-year-old woman presented to the outpatient clinic of Shenzhen people’s hospital with fever and cough. She had experienced intermittent fever for 4 months, with a temperature range of 38.0-38.5°. She had also been coughing for 2 months, with a small amount of yellow sputum, had chills, lacked dyspnea, suffered hemoptysis and had night sweats. In consideration of the patient’s history of Systemic Lupus Erythematosus (SLE) and type V lupus nephritis, she was treated with 60mg qd of prednisone and 750 mg bid of mycophenolate mofetil. Additionally, the patient had no previously known pulmonary or thoracic diseases and had no history of smoking. She had also not traveled to Wuhan or other COVID- 19 risk areas. Moreover, Polymerase Chain Reaction (PCR) tests on pharyngeal swabs were negative for SARS-CoV-2-RNAN-gene1.
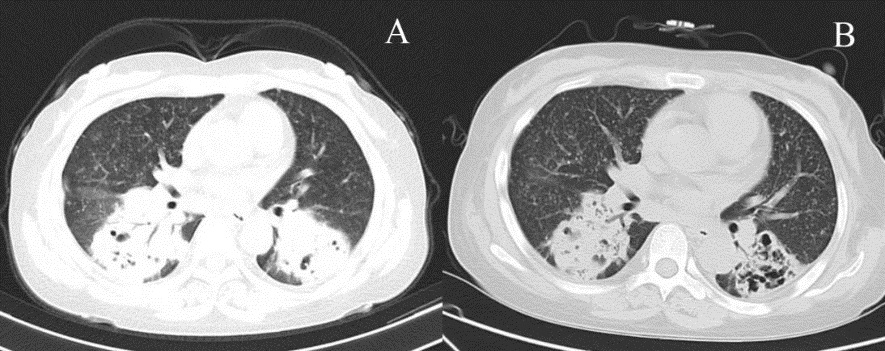
She was taken to hospital with stable vital signs. Physical examination revealed acute appearance, mental fatigue, a full moon face, a heavy breathing sound from both lungs, no dry-wet rales, mild edema on both lower limbs and no abnormality in other systems. Additionally, Computed Tomography (CT) showed bilateral lung masses with many cavities and a small amount of pleural effusion, diffuse Ground-Glass Opacity (GGO) and miliary nodules (Figure 1a). After admission, the patient received empirical treatment with Cefoperazone-Sulbactam (SCF) and low-flow oxygen inhalation through nasal catheters.
Peripheral blood analysis revealed the following results: White blood cell count, 13.53×109/L; hemoglobin, 100.00 g/L; platelet count, 172.00×109/L; Procalcitonin (PCT), 24.64 ng/ml; hs-CRP,150.2 mg/L; CO2CP, 13.1 mmol/L; IL6,16.45 pg/ml; lactate dehydrogenase (LDH), 283 × 109/L. Erythrocyte sedimentation rate(ESR), 49 μmm/h; CD4+/CD8+Th/Ts Ratio: 0.28; CD3+CD4+,12.8/ μL; CD3+CD8+T,45.8/ μL; Total protein, 40.3 mg/L, serum albumin, 22.3 g/L; globulin,18 g/L; urea nitrogen,18.11 mmol/L; creatinine, 235.1 umol/L;eGFR,3.06 ml/ (min ·1.73 m2).
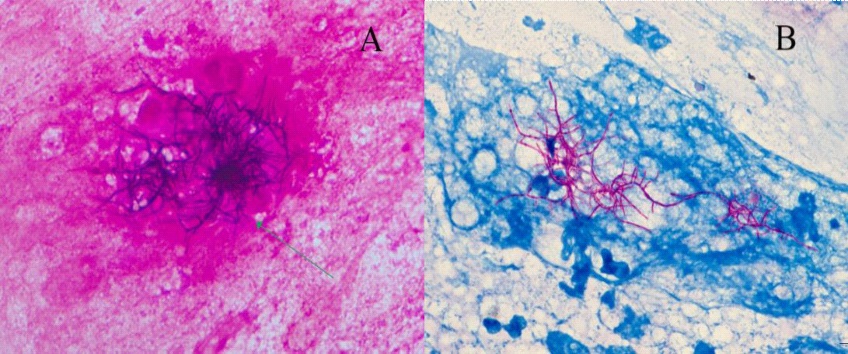
On the 3rd day of admission, coagulation function was found to be abnormal and there was progressive decrease in platelets (72×109/L), INR: 4.42. Therefore, vitamin K was injected intramuscularly to correct the coagulation function. The sputum, Bronchoalveolar Lavage Fluid (BALF) and lung tissue showed positive vertical branches by modified acid-fast staining. In addition, Nocarbacter infection was highly suspected (Figure 2), so treatment with Cefoperazone-Sulbactam (CSL) was discontinued and switched to trimethoprim sulfamethoxazole (SXT, 140 mg every 12 hours), imipenom (IPM, 500 mg every 8 hours), and moxifloxacin (MFX, 400 mg once daily). Xpert MTB/RIF (Xpert), a rapid molecular test used to detect drug resistance of Mycobacterium tuberculosis and rifampicin, tested positive for Mycobacterium tuberculosis complex DNA. However, the level of detection was so low that it was ignored. Moreover, lung biopsy showed phagocyte aggregation and neutrophil lymphocyte infiltration.
On the 9th day, the patient was still coughing up a small amount of white foamed sputum although results from routine blood tests and inflammatory indexes revealed significant improvement. CT also showed an improvement in pulmonary inflammation (it was better than before) but GGO and the nodule shadow still existed. (Figure 1b). Specimen culture gave positive results for the Nocardia nova complex (N. nova 50%, N. Africa 50%) which was confirmed through Mass Spectrometry (MS). Due to limitation by laboratory conditions, the drug sensitivity test could not be carried out according to the CLSI standards. After SXT treatments, the pulmonary infection of the patient was significantly improved. In addition, BALF was again moderately positive with Xpert detection Given the medical history, the patient was highly likely suffering from tuberculosis and the results from lung tissue culture would help to make a definitive diagnosis.
Discussion
In this case, a modified acid-fast stain showed the presence of Nocardia, which is characterized by vertical branching. One week later, the patient’s sputum and BALF were cultured and MS confirmed the presence of the Nocadia nova complex. In addition, two rounds of examination using Xpert showed that the patient was positive and MTB infection was confirmed on the 18th day of lung tissue culture. However, it is still unclear whether the patient was suffering from pulmonary Nocardiosis or pulmonary tuberculosis. This is because infection with Nocardia has no specific clinical and imaging manifestations. Additionally, the clinical and imaging manifestations of infection with Nocardia are usually similar to those of COVID-19, pulmonary tuberculosis, fungal infection and lung tumor. It is important to note that isolation and culture of patient specimens are not the only means of diagnosing pathogenic infections, as identification of genera and species and drug susceptibility tests may also be used. However, Nocardia grows slowly and usually needs to be cultured at 35-37oC for about one week making it very difficult to isolate from samples containing normal microflora, such as respiratory tract samples (e.g. sputum and BALF). Moreover, the traditional identification process through phenotypic and biochemical experiments is extremely tedious and has low accuracy [13]. Given that the different species of Nocardia have distinct levels of pathogenicity and sensitivity to drugs, it is very important to accurately identify the species. According to the most recent report, of the 105 species of Nocardia present, Nocardia asteroid is the most common while Nocardia brasiliensis is mainly associated with abscesses [14].
Furthermore, Nocardia abscess is 100% resistant to both IPM and ceftriaxone. On the other hand, Nocardia brasiliensis is resistant to most of the commonly used antibiotics. Presently, SXT is the first choice for the treatment of critically ill patients and is usually used in combination with carbapenem [15,16].
In this case, isolation and culture revealed the presence of the N. Nova complex (N. Nova 50%, N. Africa 50%) and this was confirmed through MS [17]. Notably, 16srRNA gene sequencing is the preferred method for identifying Nocardia. MS may become the gold standard in future because it is cost effective and easy to operate. In patients with suspected tuberculosis infection, Xpert uses molecular biological methods to simultaneously detect the pathogen and rifampicin resistance. This allows for timely treatment of tuberculosis and is recommended for use in combination with culture [18]. In this case, tuberculosis infection was confirmed in the patient's BALF and lung tissue through Xpert and MS. Although Nocardia and MTB are opportunistic pathogens, their incidence in immunosuppressed patients is much higher than that in patients with normal immune function [16]. Therefore, infections with Nocardia are common in patients receiving immunosuppressive therapy. Moreover, several studies have shown that in addition to pulmonary infections, disseminated infections have been reported in areas such as the brain, skin, and soft tissue [11,19,20]. Cases of mixed infection are quite rare in immunosuppressed patients. A retrospective analysis in Cambodia found that 33% of tuberculosis patients were complicated with other bacterial infections [21]. On the other hand, an Iranian study evaluated 189 patients with suspected pulmonary tuberculosis and found that of the 32 patients infected with HIV, 4 cases were infected with MTB and 2 cases were co-infected with Nocardia [6]. In addition, a rare case of pleural empyema caused by co-infection with MTB, non-tuberculosis mycobacteria and Nocardia asteroides was reported in Taiwan in a patient with SLE [22]. Nonetheless, focus should also be directed to mixed infections with bacteria and fungi in immunosuppressed patients. It is worth noting that advances in medical technology have extended the lifespan of individuals, but this has provided opportunities for opportunistic pathogens, which can lead to lung infections. Moreover, common laboratory indicators may not reflect the true status of the infection, leading to missed diagnosis [23,24]. In this case, the patient was admitted to our hospital mainly because of "Fever and Cough" and imaging revealed that there were ground glass density shadow, similar to COVID-19 at around the same time. However, infection with COVID-19 was ruled out following a nucleic acid test after admission. Considering the existence of immunosuppression in the patient, the patient was examined by staining, isolation culture, MS, molecular biology and other methods, and the co-infection of Nocardia and Mycobacterium tuberculosis was found. Therefore, clinicians should pay attention to suspected symptoms of infection in immunosuppressed patients, consider opportunistic pathogens and co- infections, and ensure timely communication with microbiology lab staff. The detection rate for opportunistic pathogens was improved by prolonging the culture time, using a special staining technique and employing a combination of various identification methods. Therefore, early diagnosis can improve patient prognosis.
Conclusion
The present report highlights a rare case of fever and cough which was finally confirmed to be a mixed pulmonary infection with the N. nova complex and MTB. It is therefore important for clinicians to be aware of opportunistic mixed infections in immunosuppressed patients and laboratory staff should combine a variety of methods for a more comprehensive diagnosis.
Funding
Supported by Shenzhen Key Medical Discipline Construction Fund (No. SZXK045)
Acknowledgements
We thank the patient for granting us permission to publish this information. Written informed consent was obtained from the patient prior to publication of this case and any accompanying images. A copy of the written consent is available for review by the Editor of this journal.
Author contribution
All the authors were involved in collection and analysis of clinical data. In addition, Jinsong Wu, Yuemei Lu and Jine Chen were responsible for the analysis of microbiological data while Zhongji Jiang and Zhikun Xu wrote the manuscript. Finally, Xueyan Liu and Yong Xu revised and approved the manuscript.
Data availability
All available information has been included in the manuscript.
Ethical approval
Ethical approval was not obtained due to clinical standard treatment.
Conflict
All the authors declare that not conflicts of interest exist.
References
- Azoulay E, Russell L, Van de Louw A, Metaxa V, Bauer P, Povoa P et al. Diagnosis of severe respiratory infections in immunocompromised patients. Intensive Care Med. 2020; 46: 298-314.
- Mueller NJ. New immunosuppressive strategies and the risk of infection. Transpl Infect Dis. 2008; 10: 379-384.
- Wilson JW. Nocardiosis: Updates and clinical overview. Mayo Clin Proc. 2012; 87: 403-407.
- Ercibengoa M, Vicente D, Arranz L, Ugarte AS, Marimon JM. Primary Cutaneous Nocardia brasiliensis in a Spanish Child. Clin Lab. 2018; 64: 1769-1772.
- Gnanam H, Rajapandian SGK, Gunasekaran R, Roshni Prithiviraj S, Ravindran RS, et al. Molecular identification of Nocardia species causing endophthalmitis using multilocus sequence analysis (MLSA): A 10-year perspective. J Med Microbiol. 2020; 69: 728-38.
- Ekrami A, Khosravi AD, Samarbaf Zadeh AR, Hashemzadeh M. Nocardia co-infection in patients with pulmonary tuberculosis. Jundishapur journal of microbiology. 2014; 7: e12495.
- Sullivan DC, Chapman SW. Bacteria that masquerade as fungi: Actinomycosis/nocardia. Proceedings of the American Thoracic Society. 2010; 7: 216-221.
- Peleg AY, Husain S, Qureshi ZA, Silveira FP, Sarumi M, et al. Risk factors, clinical characteristics, and outcome of Nocardia infection in organ transplant recipients: A matched case-control study. Clin Infect Dis. 2007; 44: 1307-1314.
- Alnaum HM, Elhassan MM, Mustafa FY, Hamid ME. Prevalence of Nocardia species among HIV-positive patients with suspected tuberculosis. Trop Doct. 2011; 41: 224-226.
- Williams E, Jenney AW, Spelman DW. Nocardia bacteremia: A single-center retrospective review and a systematic review of the literature. Int J Infect Dis. 2020; 92: 197-207.
- Jeong JH, Moon SM, Park PW, Ahn JY, Kim KH, et al. Multiple Brain Abscesses Caused by Nocardia asiatica in a Patient With Systemic Lupus Erythematosus: The First Case Report and Literature Review. Ann Lab Med. 2017; 37: 459-461.
- Mehrabadi SM, Taraghian M, Pirouzi A, Khaledi A, Neshani A, et al. Pulmonary Nocardiosis in Suspected Tuberculosis Patients: A Systematic Review and Meta-Analysis of Cross-Sectional Studies. Ethiop J Health Sci. 2020; 30: 293-300.
- Praharaj I, Sujatha S, Ashwini MA, Parija SC. Co-infection with Nocardia asteroides complex and Strongyloides stercoralis in a patient with autoimmune hemolytic anemia. Infection. 2014; 42: 211-214.
- Fatahi-Bafghi M. Nocardiosis from 1888 to 2017. Microb Pathog. 2018; 114: 369-384.
- Schlaberg R, Fisher MA, Hanson KE. Susceptibility profiles of Nocardia isolates based on current taxonomy. Antimicrob Agents Chemother. 2014; 58: 795-800.
- Steinbrink J, Leavens J, Kauffman CA, Miceli MH. Manifestations and outcomes of nocardia infections: Comparison of immunocompromised and nonimmunocompromised adult patients. Medicine (Baltimore). 2018; 97: e12436.
- Marní M, Ruiz A, Iglesias C, Quiroga L, Cercenado E, Martín -Rabadán P et al. Identification of Nocardia species from clinical isolates using MALDI-TOF mass spectrometry. Clin Microbiol Infect. 2018; 24: 1342. e5-.e8.
-
- Dorman SE, Schumacher SG, Alland D, Nabeta P, Armstrong DT, et al. Xpert MTB/RIF Ultra for detection of Mycobacterium tuberculosis and rifampicin resistance: A prospective multicentre diagnostic accuracy study. Lancet Infect Dis. 2018; 18: 76-84.
- Nakamura I, Nagakura T, Fujita H, Fukusima S, Gonoi T. Nocardia elegans infection: A case report and literature review. Int J Infect Dis. 2017; 54: 15-17.
- Wang T, Jia Y, Chu B, Liu H, Dong X, et al. Nocardiosis in Kidney Disease Patients under Immunosuppressive Therapy: Case Report and Literature Review. International journal of medical sciences. 2019; 16: 838-44.
- Attia EF, Pho Y, Nhem S, Sok C, By B, Phann D, et al. Tuberculosis and other bacterial co-infection in Cambodia: a single center retrospective cross-sectional study. BMC Pulm Med. 2019; 19:60
- Huang HC, Yu WL, Shieh CC, Cheng KC, Cheng HH. Unusual mixed infection of thoracic empyema caused by Mycobacteria tuberculosis, nontuberculosis mycobacteria and Nocardia asteroides in a woman with systemic lupus erythematosus. J Infect. 2007; 54: e25-8.
- Jain S, Self WH, Wunderink RG, Fakhran S, Balk R, Bramley A, M et al. Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults. N Engl J Med. 2015; 373: 415-27.
- Vargas E, Apewokin S, Madan R. Role of the leukocyte response in normal and immunocompromised host after Clostridium difficile infection. Anaerobe. 2017; 45: 101-105.
Conclusion
The present report highlights a rare case of fever and cough which was finally confirmed to be a mixed pulmonary infection with the N. nova complex and MTB. It is therefore important for clinicians to be aware of opportunistic mixed infections in immunosuppressed patients and laboratory staff should combine a variety of methods for a more comprehensive diagnosis.
Funding
Supported by Shenzhen Key Medical Discipline Construction Fund (No. SZXK045)
Acknowledgements
We thank the patient for granting us permission to publish this information. Written informed consent was obtained from the patient prior to publication of this case and any accompanying images. A copy of the written consent is available for review by the Editor of this journal.
Author contribution
All the authors were involved in collection and analysis of clinical data. In addition, Jinsong Wu, Yuemei Lu and Jine Chen were responsible for the analysis of microbiological data while Zhongji Jiang and Zhikun Xu wrote the manuscript. Finally, Xueyan Liu and Yong Xu revised and approved the manuscript.
Data availability
All available information has been included in the manuscript.
Ethical approval
Ethical approval was not obtained due to clinical standard treatment.
Conflict
All the authors declare that not conflicts of interest exist.
References
- Azoulay E, Russell L, Van de Louw A, Metaxa V, Bauer P, Povoa P et al. Diagnosis of severe respiratory infections in immunocompromised patients. Intensive Care Med. 2020; 46: 298-314.
- Mueller NJ. New immunosuppressive strategies and the risk of infection. Transpl Infect Dis. 2008; 10: 379-384.
- Wilson JW. Nocardiosis: Updates and clinical overview. Mayo Clin Proc. 2012; 87: 403-407.
- Ercibengoa M, Vicente D, Arranz L, Ugarte AS, Marimon JM. Primary Cutaneous Nocardia brasiliensis in a Spanish Child. Clin Lab. 2018; 64: 1769-1772.
- Gnanam H, Rajapandian SGK, Gunasekaran R, Roshni Prithiviraj S, Ravindran RS, et al. Molecular identification of Nocardia species causing endophthalmitis using multilocus sequence analysis (MLSA): A 10-year perspective. J Med Microbiol. 2020; 69: 728-38.
- Ekrami A, Khosravi AD, Samarbaf Zadeh AR, Hashemzadeh M. Nocardia co-infection in patients with pulmonary tuberculosis. Jundishapur journal of microbiology. 2014; 7: e12495.
- Sullivan DC, Chapman SW. Bacteria that masquerade as fungi: Actinomycosis/nocardia. Proceedings of the American Thoracic Society. 2010; 7: 216-221.
- Peleg AY, Husain S, Qureshi ZA, Silveira FP, Sarumi M, et al. Risk factors, clinical characteristics, and outcome of Nocardia infection in organ transplant recipients: A matched case-control study. Clin Infect Dis. 2007; 44: 1307-1314.
- Alnaum HM, Elhassan MM, Mustafa FY, Hamid ME. Prevalence of Nocardia species among HIV-positive patients with suspected tuberculosis. Trop Doct. 2011; 41: 224-226.
- Williams E, Jenney AW, Spelman DW. Nocardia bacteremia: A single-center retrospective review and a systematic review of the literature. Int J Infect Dis. 2020; 92: 197-207.
- Jeong JH, Moon SM, Park PW, Ahn JY, Kim KH, et al. Multiple Brain Abscesses Caused by Nocardia asiatica in a Patient With Systemic Lupus Erythematosus: The First Case Report and Literature Review. Ann Lab Med. 2017; 37: 459-461.
- Mehrabadi SM, Taraghian M, Pirouzi A, Khaledi A, Neshani A, et al. Pulmonary Nocardiosis in Suspected Tuberculosis Patients: A Systematic Review and Meta-Analysis of Cross-Sectional Studies. Ethiop J Health Sci. 2020; 30: 293-300.
- Praharaj I, Sujatha S, Ashwini MA, Parija SC. Co-infection with Nocardia asteroides complex and Strongyloides stercoralis in a patient with autoimmune hemolytic anemia. Infection. 2014; 42: 211-214.
- Fatahi-Bafghi M. Nocardiosis from 1888 to 2017. Microb Pathog. 2018; 114: 369-384.
- Schlaberg R, Fisher MA, Hanson KE. Susceptibility profiles of Nocardia isolates based on current taxonomy. Antimicrob Agents Chemother. 2014; 58: 795-800.
- Steinbrink J, Leavens J, Kauffman CA, Miceli MH. Manifestations and outcomes of nocardia infections: Comparison of immunocompromised and nonimmunocompromised adult patients. Medicine (Baltimore). 2018; 97: e12436.
- Marní M, Ruiz A, Iglesias C, Quiroga L, Cercenado E, Martín -Rabadán P et al. Identification of Nocardia species from clinical isolates using MALDI-TOF mass spectrometry. Clin Microbiol Infect. 2018; 24: 1342. e5-.e8.
- Dorman SE, Schumacher SG, Alland D, Nabeta P, Armstrong DT, et al. Xpert MTB/RIF Ultra for detection of Mycobacterium tuberculosis and rifampicin resistance: A prospective multicentre diagnostic accuracy study. Lancet Infect Dis. 2018; 18: 76-84.
- Nakamura I, Nagakura T, Fujita H, Fukusima S, Gonoi T. Nocardia elegans infection: A case report and literature review. Int J Infect Dis. 2017; 54: 15-17.
- Wang T, Jia Y, Chu B, Liu H, Dong X, et al. Nocardiosis in Kidney Disease Patients under Immunosuppressive Therapy: Case Report and Literature Review. International journal of medical sciences. 2019; 16: 838-44.
- Attia EF, Pho Y, Nhem S, Sok C, By B, Phann D, et al. Tuberculosis and other bacterial co-infection in Cambodia: a single center retrospective cross-sectional study. BMC Pulm Med. 2019; 19:60
- Huang HC, Yu WL, Shieh CC, Cheng KC, Cheng HH. Unusual mixed infection of thoracic empyema caused by Mycobacteria tuberculosis, nontuberculosis mycobacteria and Nocardia asteroides in a woman with systemic lupus erythematosus. J Infect. 2007; 54: e25-8.
- Jain S, Self WH, Wunderink RG, Fakhran S, Balk R, Bramley A, M et al. Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults. N Engl J Med. 2015; 373: 415-27.
- Vargas E, Apewokin S, Madan R. Role of the leukocyte response in normal and immunocompromised host after Clostridium difficile infection. Anaerobe. 2017; 45: 101-105.