Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Duplex optogenetic stimulation system modulates compulsive behaviors bidirectionally
Edenia Menezes1; David Ashurov1; Catarina Sousa Cunha1,2*
1 Emotional Brain Institute, The Nathan Kline Institute for Psychiatric Research, Orangeburg, NY, USA.
2 NYU Langone Health, Department of Child & Adolescent Psychiatry, New York, NY 10016, USA.
*Corresponding Author: Catarina Sousa Cunha
Departments of Child & Adolescent Psychiatry and
Neuroscience & Physiology, New York University
Langone School of Medicine, One Park Ave 8th Floor,
New York, NY 10016, USA.
Email: Catarina.sousacunha@nyulangone.org
Received : May 05, 2021
Accepted : Jun 23, 2021
Published : Jun 29, 2021
Archived : www.jcimcr.org
Copyright : © Cunha CS (2021).
Abstract
Decisions enable us to consider our actions while adjusting behaviors to a relentlessly changing environment. Since conscious decision making requires our full attention and is therefore expensive, we use a cheaper system for everyday and repetitive tasks that run automatically without conscious evaluations, commonly described as habits. The combination of these two systems is highly adaptive. However, if there is a disruption in these systems’ balance, mental illnesses such as Obsessive-Compulsive Disorders (OCD) or addiction may arise.
Now, how does a newly acquired memory or behavior transition to a habit?
We know that the striatum’s dorsolateral region (DLS) plays a significant role in sustaining successful behaviors and habits manifestation. Furthermore, it has been shown that the micro-circuitry of the DLS is organized into two functional opposing pathways that consist of the direct pathway striatal spiny projection neurons (dSPNs), which facilitate movement, and the indirect pathway SPNs, which inhibit actions.
Already Freud wrote in the letters to his friend Fliess that memory and motive are inseparable, and its recollection would have no force or meaning unless it would be coupled to a motive or emotion. Therefore, we hypothesize that emotional-associated cues would directly prime the DLS through the amygdala to turn newly acquired behaviors into habits. We are adding new supporting insight for this hypothesis by using a duplex optogenetic stimulation system of the amygdala input in the DLS, modulating specific compulsive behaviors bidirectionally. These behavior modulations were accompanied by spine density modulations and intrinsic excitability changes in the SPNs of the DLS.
Keywords: Compulsions; Habits; Optogenetics; Neuromodulation; Dorsolateral striatum; Amygdala.
Citation: Menezes E, Ashurov D, Cunha CS. Duplex optogenetic stimulation system modulates compulsive behaviors bidirectionally. J Clin Images Med Case Rep. 2021; 2(3): 1201.
Introduction
We make decisions every day. They allow us to evaluate the consequences of our actions and to adapt to a continually changing environment. However, conscious decision making is an expensive process and requires our full attention. For every day and repetitive tasks, our brain uses a different system to free up working memory to focus on new tasks and challenges: habits. Habits are formed based on successfully learned and applied behaviors, which turned into a routine that runs automatically without conscious evaluations [1]. The combination of both systems is efficient and highly adaptive.
However, if there is a disproportional weight on one of the two, it may lead to various illnesses such as Obsessive-Compulsive Disorders (OCD) [2,3] or addiction [4].
It has been sown in various animals such as mice, rats, nonhuman primates, and humans that the goal-directed system is centered in the Dorsomedial (DMS), and the habit system is located in the Dorsolateral Striatum (DLS) [5-8]. From inactivation and lesion studies of the DMS or DLS, we know that either action-outcome or habit strategies become disrupted [6- 9]. How does the transition from a newly acquired memory or behavior turn into a habit? In the 1890s, Freud wrote in “The Project,” which consisted of letters to his friend and colleague Fliess, that there are neuronal systems with contact barriers that are capable of selective facilitation or inhibition [10]. Those would allow permanent neuronal changes that represent information acquisition and new memories. This hypothesis of the associative process underlying action selection was right in the striatum’s spiny projection neurons, where brief glutamate uncaging at spines on distal dendrites evoked somatic upstates lasting hundreds of milliseconds [11]. Freud also wrote that memory and motive are inseparable, and its recollection would have no force or meaning unless it would be coupled to a motive or emotion [10], which facilitates long-term memory. Hebb’s work described this vital aspect of causation and added the requirement of temporal precedence [12]. Johansen et al. 2014, provided us with direct evidence supporting the idea of Hebbian processes in the amygdala and showed that it is both necessary and sufficient to trigger synaptic strengthening and behavioral associative memory formation [13]. These hypotheses and results propose a direct functional projection from the amygdala, which is mainly responsible for labeling both positive and negative emotions [14,15], to the DLS, where behaviors turn into ingrained habits. Solving this problem would bring us closer to gain a more in-depth insight into information integration mechanisms and to comprehend better aberrations in the circuitry of mental health disorders such as OCD and addiction. Striatal spiny projection neurons (SPNs) of the dorsal striatum are organized into two functional opposing pathways, the direct pathway formed by the dSPNs, which express the dopamine D1 receptor and facilitate movement, and the indirect pathway iSPNs, which express dopamine D2 receptors and inhibit activity [16-21]. Based on the cellular output consequences, the balance of these two pathways is critical for appropriate behavior and habit learning. Anatomical work showed that while SPNs in the dorsal striatum mostly receive sensory and motor-related inputs, the same SPNs also receive a small number of amygdala inputs [22]. This study shows a direct functional synaptic connection between the basal and lateral nuclei of the amygdala (BLA) and DLS. Synchronized activation of the BLA inputs with a blue laser targeted to a distal dendrite in current-clamp mode could evoke somatic action potentials. These action potentials were followed by a prolonged depolarization lasting tens of milliseconds, similar to that produced by optogenetic activation of cortical terminals, but significantly scaled-down in both amplitude and duration. BLA terminal activation in the DLS modulated mice’s behavior, and over-activation of this pathway induced OCD-like behaviors in mice and were turned back to an average frequency using a depotentiation stimulus. Our mapping results showed that in the dSPNS of the DLS of optogenetically induced OCD-like behaving mice, the spine density was increased, and intrinsic excitability was elevated compared to the iSPNs. The observed circuit changes indicate increased synaptic function of the direct pathway associated with hyperkinetic behavior, such as compulsive grooming. In summary, the results demonstrate how DLS circuitry can be plastically modulated by over activation of the amygdala circuit, contributing to repetitive motor behavior. However, most importantly, the results indicate that using a low-frequency stimulation of the BLA-DLS pathway can ameliorate compulsive behavior.
Methods
We used ~75-110-day old BAC transgenic drd1a-td Tomato transgenic mice following procedures approved by the Nathan Kline Animal Care and Use Committee. Surgeries: For stereotaxic surgery, mice were anesthetized with ketamine and xylazine and secured in a Neurostar robot stereotaxic apparatus. WT mice received unilateral injections of a synapsin-ChR2-GFP-AAV construct (UPenn vector core) at 2-5 months of age. Stereotaxic injections targeted the BLA (ML -2.99 mm, AP -1.22 mm, DV -4.38 mm), thalamus (ML 1.5 mm, AP −4.25 mm, DV 3.5 mm), and SMC (ML 1 mm, AP -2.5 mm, DV 0.75 mm). In the same surgery, a mono fiber-optic cannula (core diameter 200 um, outer diameter 245 um, 0.37 NA, Doric Lenses) was implanted in the dorsolateral striatum (0.6 mm anterior to Bregma, tip depth 2.7 mm below pia surface) and held in place with dental cement. After the implantation surgery, animals were allowed to recover for 10 to 14 days. Behavior: Two weeks following surgery, the cannula was coupled to a fiber optic patch cord attached to a 473 nm blue LASER and placed in a transparent glass cylinder. Mice were acclimated to this configuration for three days, 10 minutes per day. After acclimation and baseline measurements, the dorsolateral striatum was stimulated by the LASER using 10 ms pulses (3 mW) at 20 Hz or 2 Hz for 10 minutes during either grooming or digging behavior, once daily for five days 29. Controls included mice that received saline injections or sham cannula implants. Mice were videotaped in the glass cylinder to assess grooming during three epochs: 1) 5 minutes before laser stimulation, 2) during laser stimulation, and 3) 5 minutes after laser stimulation. Grooming was also assessed three days before the first stimulation and one week following the last LASER stimulation. All data presented is from 5 min before stimulation. Total time spent grooming was quantified by the observer’s blind to experimental conditions.
Electrophysiology
Anesthetized mice (50 mg kg−1 ketamine and 4.5 mg kg−1 xylazine) were perfused transcardially with 5–10 ml ice-cold artificial CSF (aCSF) containing (in mM): 124 NaCl, 3 KCl, 1 CaCl2 ,1.5 MgCl2 , 26 NaHCO3 , 1 NaH2 PO4 , and 16.66 glucose, continuously bubbled with carbogen (95% O2 and 5% CO2 ). Slices were then transferred to a holding chamber where they were incubated in ACSF containing (in mM) 2 CaCl2 , 1 MgCl2 , at 35°C for 60 min, after which they were stored at room temperature until recording. Identified SPNs were recorded from a patch-clamp system equipped with integrated calcium imaging capabilities. The recording electrode contained (in mM) 135 Cs-gluconate, 10 HEPES, 10 Naphosphocreatine, 4 Mg2-ATP, and 0.4 NaGTP (290–295 mOsm, pH 7.35 with CsOH) and 0.05 Alexa 568 and 0.5 Fluo-5F hydrazide Na+ salt (Invitrogen). Dendrites and calcium signals (held at 0 mV) were visualized in real-time for mapping experiments. Ca2+ signals were analyzed in Matlab and expressed as ΔF/Fo. For spine density studies, high magnification z-series images (0.2 μm steps, 5x digital zoom) were taken through 2 distal (>100 µm from the soma) and proximal (50 µm from the soma) dendritic regions per SPN. The images were deconvoluted using AutoQuant X2 (Media Cybernetics), and spine density analysis was performed with Neuron Studio. Differences were considered statistically significant if p< 0.05.
Results
Anatomical research has stressed that while striatal spiny projection neurons (SPNs) primarily receive sensory and motorrelated inputs in the DLS, a limited number of amygdala inputs also converge on the same SPNs [22]. Although such convergence may indicate a possible mechanism through which emotion-associated priming may directly influence dorsal striatummediated action selection and habit learning, clear functional evidence of these synaptic connections is scarce.
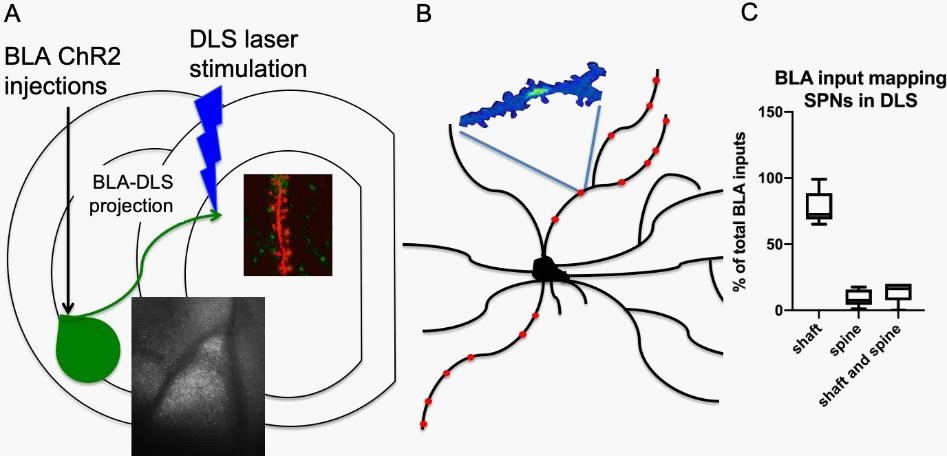
Characterization and localization of BLA inputs on dSPNs and iSPNs in the DLS
In order to characterize how the BLA innervates the DLS, Channelrhodopsin 2 (ChR2) was stereotaxically injected into the BLA. After two weeks, we voltage-clamped and filled dSPNs and iSPNs with calcium dye in the DLS of acute brain slices and visualized the BLA terminals along their dendrite while activating them by trailing blue laser spots along the dendrite. This way, we measured and imaged the location of BLA terminal activation- induced calcium transients in the dSPNs and iSPNs of the DLS simultaneously.
Our results show that the BLA functionally innervates both dSPNs and iSPNs in the DLS. While the BLA innervation is scant, it is distributed mainly along the dendritic arbor of dSPNs and iSPNs. We also observed that the BLA projection to the DLS favorably targets the dendritic shafts, not the spines, of both SPNs types (Figure 1).
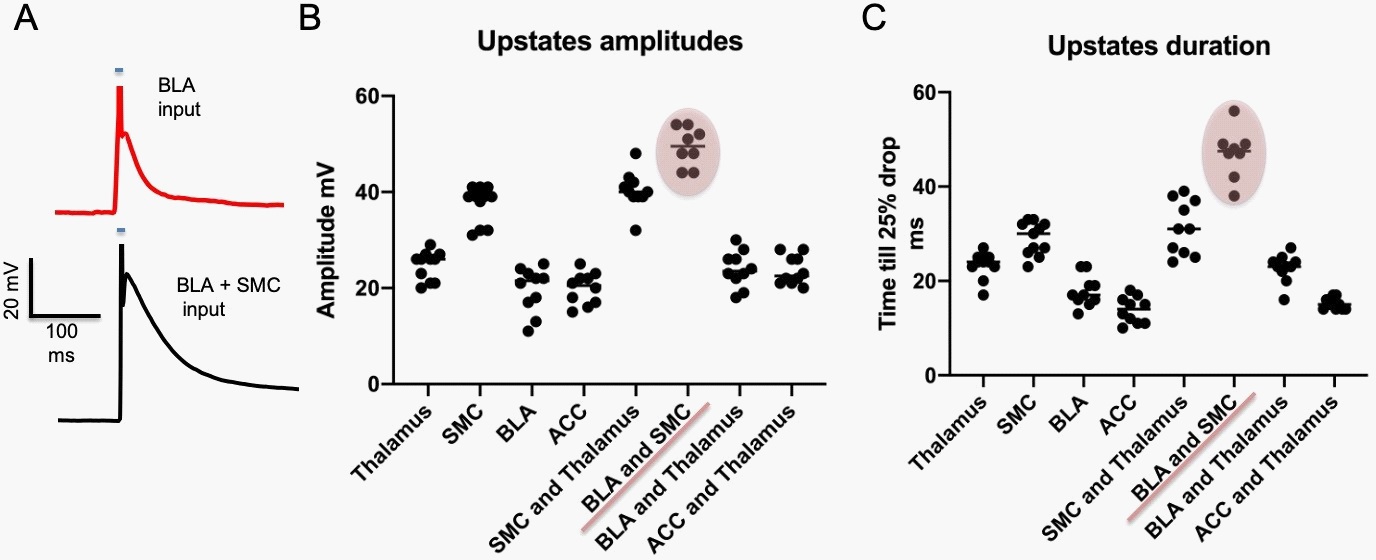
Role of BLA in shaping heterosynaptic integration in SPNs
Here we show that stimulating specific optogenetic DLS input terminal activation with just five laser pulses on distal dendrites in current-clamp mode evokes an action potential in the recorded SPNs. Furthermore, suppose we activated, using the same stimulation protocol, simultaneously the BLA and SMC inputs. In that case, we could functionally enhance the amplitude and duration of evoked synaptic responses in SPNs significantly more than with any other input stimulation grouping (Figure 2).
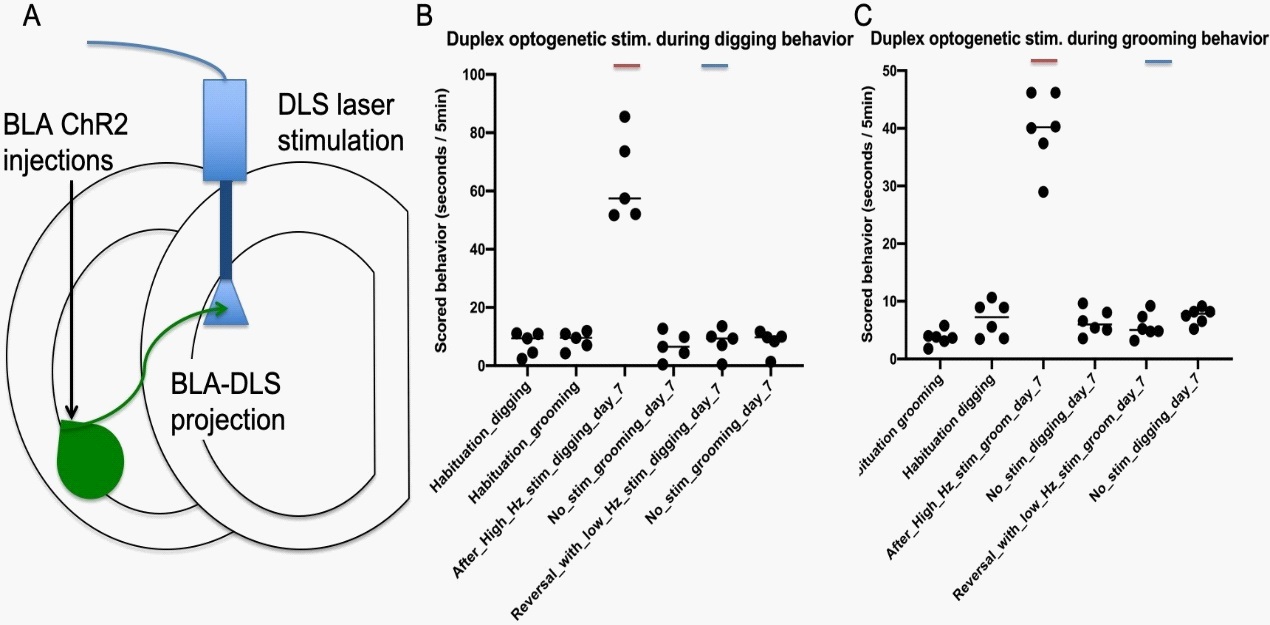
Duplex optogenetic stimulation system modulates compulsive behaviors bidirectionally
Next, we wanted to test which role the BLA-DLS pathway stimulation plays in the behaving animal. Furthermore, we hypothesized that the BLA-DLS pathway bidirectionally controls the frequency of specific behaviors. Therefore, we repeatedly stimulated optogenetically the BLA axon terminals within the DLS during specific behaviors first at a high frequency (20 Hz), and to reverse the effect, we used a depolarizing low-frequency stimulation of 2 Hz. Our data show that the stimulation of BLA axons in the DLS during specific behaviors produces a long-term and behavior-specific (grooming vs. digging and vice versa) increase in response frequency while sparing other behaviors (grooming/digging) (Figure 3). Succeeding behavior-specific low-frequency stimulation of this pathway reduced these specific targeted repetitive behavior frequencies back to baseline levels while untargeted behaviors were left unchanged (Figure 3).
Plasticity changes accompany repetitive behavior modulations
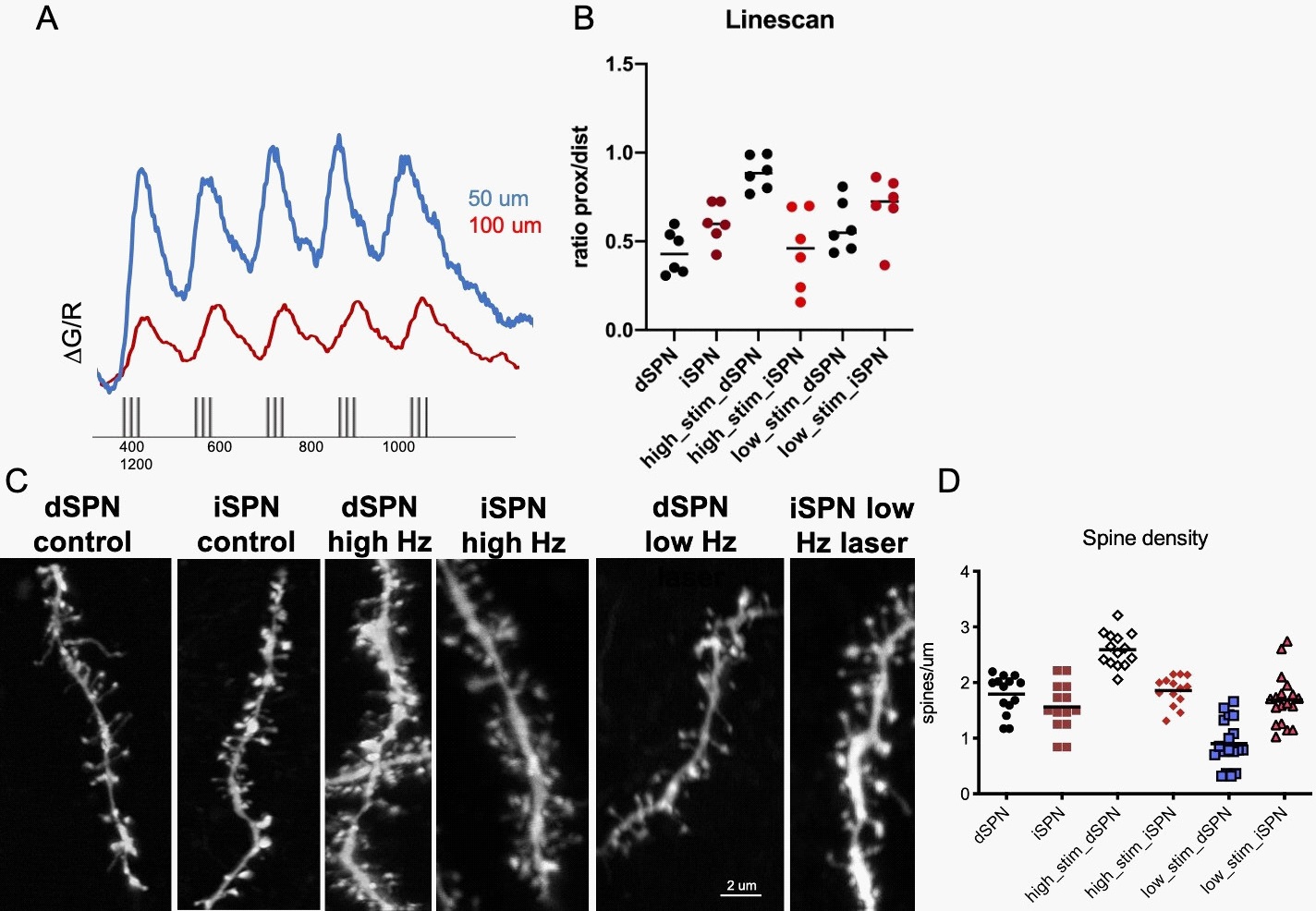
In this part of the study, we examine if the BLA inputs' behavioral effects to the DLS induce excitability and plasticity changes in the SPNs.
We conducted combined electrophysiology and imaging experiments to describe the intrinsic excitability changes in SPNs of the DLS striatum after in vivo BLA input stimulation. We evoked backpropagating action potentials (bAPs) while measuring calcium dynamics of dendrites by filling SPNs with the high-affinity Fluo-5F hydrazide Na+ salt (Invitrogen) calcium dye through the patch electrode (Figure 4). In order to elicit action potential that propagated to the dendrites and evoked calcium transients, we conducted current injections into the somata of SPNs (Figure 4A). We used five bursts of action potentials because of the better signal-to-noise ratio for trains since they have been shown to be more sensitive indicators of dendritic scaling of bAPevoked calcium transients than single action potentials [23]. We conducted experiments to compare the relative amplitudes of bAP-evoked calcium transients along the dendrite using the high- speed line-scan mode. We regularly tested transients at a reference point (50 μm distance) and at a remote position (100 μm distance) of the dendrite to normalize the magnitude of the evoked transients throughout the duration of the experiment. The ratio of transient amplitudes was higher in high-frequency BLA input stimulated dSPNs than iSPNS of the DLS. In the low frequency stimulated SPNs of the DLS through the BLA input, we could not detect a significant difference (Figure 4 A,B).
To determine if the behavioral modulation through BLA input stimulations in the DLS induced synaptic plasticity, we examined spine density in dSPNs and iSPNs. We found that dSPNs spine densities were significantly increased in high-frequency BLA-DLS pathway stimulated mice compared to both low frequency stimulated and control mice (Figure 4C,D).
Discussion
Our results show a direct and functional activation of the SPNs DLS from the BLA.
In the study by Wall et al. 2014 contextual information, encompassing sensory information (from somatosensory cortex) and valence (from amygdala) may be preferentially routed to the direct pathway to select or initiate actions based on past experiences in similar contexts [22]. However, as with any anatomical labeling technique, we must be careful extrapolating physiological significance for an entire brain structure from anatomical data alone, particularly given that we only sampled from a restricted, slightly laterally biased region in the dorsal striatum.
From behavioral studies, we know that with just a single presentation, children can learn a new word [24]. But in contrast with behavioral and in vivo physiological observations, in vitro experiments on synaptic plasticity show that long-term plasticity depends on the activation of coactive synapses [25]. Additionally, this physiological process is usually known to be incremental [26]. Furthermore, habits have been shown to occur after the repetition of performed behaviors [1]. However, we hypothesize that if emotional and sensory information coincides, long-term plasticity occurs with only a few presentations. The fact that BLA inputs to the DLS occur mainly on the dendritic shaft of the SPNs (Figure 1) while SMC inputs are known to be predominately located on the dendritic spines of the SPNs in the DLS [27], supports our hypothesis since even only a few dendritic shaft BLA inputs would amplify the incoming SMC input onto the spines [28]. We're demonstrating here that synaptic stimulation of the combined BLA and SMC input on distal dendrites evoked higher dendritic plateau potentials and somatic up-states than stimulation of other input combinations. This difference was not attributable to a higher density of inputs at distal synapses, but rather the localization of BLA input on the dendritic shaft amplifying the SMC input on the dendritic spines. We show here how emotional priming could accelerate the process of habit learning-related synaptic plasticity in vitro.
We detected long-lasting circuit alterations in the DLS that accompanied the expression of specific repetitive behavior: 1) an increase in the intrinsic excitability of dSPNs measured by eliciting bAPs while running line scans to measure dendritic propagation (Figure 4&2) a general increase in dendritic spine density in dSPNs (Figure 4). These circuit modifications indicate increased synaptic drive of the direct pathway, a symptom associated with hyperkinetic behavior. Taken together, these results explain how over- activation of the limbic-associated amygdala circuit will plastically modulate the dorsolateral striatum performance, contributing to repeated motor behavior. First, we looked at whether we could counteract repetitive behavior through low-frequency de- potentiating stimulation of the BLADLS pathway only during digging/grooming behavior, which contributed to a decrease in digging/grooming but did not affect grooming/digging behavior (Figure 2), which was followed by improvements in spine density (Figure 4). These findings support the theory that the BLA-DS system contains compulsive behaviors that contribute to plasticity modulations and improvements in the encoding of information in this network and that the depotentiation of this pathway decreases compulsive behavior. In summary, the findings show how overactivation of the amygdala circuit may plastically modulate the dorsolateral striatum, leading to compulsive behaviors.
Author contribution: Catarina Cunha conceived of the presented idea. Catarina Cunha developed the experiment design. Edenia Menezes, David Ashurov and Catarina performed experiments and oversaw the project. German Todorov analyzed and verified the data and analytical methods. David Ashurov and Edenia Menzes assisted with the blind analysis of behavior data. All authors discussed the results and contributed to the final manuscript.
Data availability: The data that support the findings of this study are available from the corresponding author (C.C.), upon reasonable request.
References
- Graybiel AM. Habits, rituals, and the evaluative brain. Annu Rev Neurosci. 2008; 31; 359-387.
- Gillan CM. et al. Enhanced avoidance habits in obsessive-compulsive disorder. Biol Psychiatry. 2014; 75: 631-638.
- Gillan CM. et al. Disruption in the balance between goal-directed behavior and habit learning in obsessive-compulsive disorder. Am J Psychiatry. 2011; 168: 718-726.
- Belin D, Belin-Rauscent A, Murray JE Everitt BJ. Addiction: Failure of control over maladaptive incentive habits. Curr Opin Neurobiol. 2013; 23: 564-572.
- Hilario M, Holloway T, Jin X, Costa RM. Different dorsal striatum circuits mediate action discrimination and action generalization. Eur J Neurosci. 2012; 35: 1105-1114.
- Yin HH, Knowlton BJ, Balleine BW. Lesions of dorsolateral striatum preserve outcome expectancy but disrupt habit formation in instrumental learning. Eur J Neurosci. 2004; 19: 181-189.
- Miyachi S, Hikosaka O, Lu X. Differential activation of monkey striatal neurons in the early and late stages of procedural learning. Exp Brain Res. 2002; 146; 122-126.
- McNamee D, Liljeholm M, Zika O, O’Doherty JP. Characterizing the associative content of brain structures involved in habitual and goal-directed actions in humans: A multivariate FMRI study. J Neurosci. 2015; 35: 3764-3771.
- Yin HH, Knowlton BJ. Balleine BW. Blockade of NMDA receptors in the dorsomedial striatum prevents action-outcome learning in instrumental conditioning. Eur J Neurosci. 2005; 22: 505-512.
- Freud S. et al. Sammlung. (Belknap Press of Harvard University Press. 1985.
- Plotkin JL, Day M, Surmeier DJ. Synaptically driven state transitions in distal dendrites of striatal spiny neurons. Nat Neurosci. 2011; 14: 881-888.
- Hebb DO. The organization of behavior; A neuropsychological theory. Wiley. 1949.
- Johansen JP, et al. Hebbian and neuromodulatory mechanisms interact to trigger associative memory formation. Proceedings of the National Academy of Sciences. 2014; 111: E5584-E5592.
- Ball T. et al. Anatomical specificity of functional amygdala imaging of responses to stimuli with positive and negative emotional valence. J Neurosci Methods. 2009; 180: 57-70.
- Beyeler A. et al. Organization of Valence-Encoding and Projection-Defined Neurons in the Basolateral Amygdala. Cell Rep. 2018; 22: 905-918.
- Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989; 12: 366-375.
- DeLong, R. Lithium treatment and bipolar disorders in childhood. N C Med J. 1990; 51: 152-154.
- Graybiel AM. The basal ganglia. Trends Neurosci. 1995; 18: 60-62.
- Hikosaka O, Takikawa Y, Kawagoe, R. Role of the basal ganglia in the control of purposive saccadic eye movements. Physiol Rev. 2000; 80: 953-978.
- Mink JW. The basal ganglia: Focused selection and inhibition of competing motor programs. Prog Neurobiol. 1996; 50: 381-425.
- Kravitz AV. et al. Regulation of parkinsonian motor behaviours by optogenetic control of basal ganglia circuitry. Nature. 2010; 466: 622-626.
- Wall NR, De La Parra M, Callaway EM, Kreitzer AC. Differential innervation of direct- and indirect-pathway striatal projection neurons. Neuron. 2013; 79: 347-360.
- Svoboda K, Helmchen F, Denk W, Tank DW. Spread of dendritic excitation in layer 2/3 pyramidal neurons in rat barrel cortex in vivo. Nat Neurosci. 1999; 2: 65-73.
- Carey, S. & Bartlett, E
- Markram H, Lubke J, Frotscher M, Sakmann B. Regulation of synaptic efficacy by coincidence of postsynaptic APs and EPSPs. Science. 1997; 275: 213-215.
- Wang HX, Gerkin RC, Nauen DW, Bi GQ. Coactivation and timingdependent integration of synaptic potentiation and depression. Nat Neurosci. 2005; 8: 187-193.
- Hintiryan H. et al. The mouse cortico-striatal projectome. Nat Neurosci. 2016; 19: 1100-1114.
- Harnett MT, Makara JK, Spruston N, Kath WL, Magee JC. Synaptic amplification by dendritic spines enhances input cooperativity. Nature. 2012; 491: 599-602.