Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Pure muscle tenosynovial giant cell tumor mimicks a metastasis in patient with melanoma
Pier Paolo Arcuri1*; Angela Commisso3; Marta John3; Giuseppe Lucio Cascini2; Domenico Laganà3
1Radiology Unit, Department of Radiology, Pugliese-Ciaccio Hospital, Catanzaro, Italy.
2Nuclear Medicine Unit, Department of Experimental and Clinical Medicine, “Magna Graecia” University, Catanzaro, Italy.
3Radiology Unit, Department of Experimental and Clinical Medicine, “Magna Graecia” University, Catanzaro, Italy.
*Corresponding Author: Pier Paolo Arcuri
Radiology Unit, Department of Radiology, PuglieseCiaccio Hospital, Catanzaro, Italy.
Email: arppaolo@alice.it
Received : Jun 12, 2021
Accepted : Jul 21, 2021
Published : Jul 29, 2021
Archived : www.jcimcr.org
Copyright : © Arcuri PP (2021).
Abstract
Tenosynovial Giant Cell Tumor (TGCT) is a rare benign synovial tumor arising from the tendon sheath, bursae, synovium or adjacent soft tissue. TGCT typical occurs in intra-articular site but can be rarely extra- articular. A 50-years-old woman already affected by malignant melanoma was submitted to PET/CT scan for routine diagnostic examinations during follow up; an intense FDG focal uptake corresponding to peritrochanteric medial part of right iliopsoas muscle was detected. Corresponding MRI images demonstrated a lesion with well-defined margins, slightly hyperintense in T1, low to intermediate signal intensity in T2 weighted scans, no significant restriction in DWI, and intense enhancement in T1 after intravenous contrast medium administration. There wasn’t evidence of extra-lesional spread.
MRI findings oriented for benign lesion, but according to intermediate signal intensity in T2, intense enhancement in T1 after intravenous contrast medium, the presence of intense FDG uptake as well as history of aggressive malignancy, a local excision was done; then the final diagnosis of TGCT has been proven. Our case shows TGCT lesions may reproduce malignant appearance on FDG-PET, while MRI may be useful tool to properly manage affected patients.
Keywords: Tenosynovial giant cell tumor; Melanoma metastasis; Diffusion-MRI; FDG-PET/CT.
Citation: Arcuri PP, Commisso A, John M, Cascini GL, Laganà D. Pure muscle tenosynovial giant cell tumor mimicks a metastasis in patient with melanoma. J Clin Images Med Case Rep. 2021; 2(4): 1242.
Introduction
Tenosynovial Giant Cell Tumor (TGCT) is a benign synovial tumor arising from the tendon sheath, bursae, synovium or adjacent soft tissue. It can be localized (single lesion) or diffuse (multiple lesions) [1]. The first is a benign and well defined lesion that occurs most commonly in the distal extremities such as hand, wrist, ankle, foot and knee. The incidence is about 1/50.000 cases for year with recurrence rate ranging between 0% and 6% after treatment.
Diffuse TGCT also named PVNS (pigmented villonodular sinovitis), shows unclear margins with massive infiltration and growth into the entire synovial membrane and sometimes into adjacent structures. Knee, hip, ankle, wrist and shoulder are the most common affected joints. Its incidence is about 2/1.000.000 case for year while recurrence rates range between 14% and 67%.
The onset of TCGT is usually intra-articular with adjacent soft tissues involvement (ligaments, tendons and muscles) resulting in swelling and functional limitation of the affected joint. Extraarticular presentations are uncommon and may involve solely the muscles [2,3]; diagnosis in these cases is incidental because the absence of specific symptoms although potential diagnostic clues as well as overlying skin’s tension and adjacent nerve structures compression may be recognized [4].
The diagnostic work should be defined according to the clinical presentation and articular involvement, and generally includes MRI and US to identify the number of lesion and extension; histological assessment is still required when diagnostic pattern persists equivocal [5].
However differential diagnosis between localized and diffuse TGCT cannot be done by using histology because cellular component and histopathological characteristics are similar in both; the diagnosis may be done on the basis of number of involved sites and extra-capsular diffusion recognized by MRI and US.
In case of specific symptoms, US is the first level diagnostic investigation to characterize the lesion and the relationship with adjacent tissues. Typically TGCT appears as a homogeneously hypoechoic mass, with internal vascularity, joint effusion and synovial membrane’s thickening in intra-articular form.
MRI can identify site, characteristics, extension of the tumor mass and may be useful for differential diagnosis with other conditions such as hemophilic arthropathy, synovial chondromatosis, synovial hemangioma, synovial sarcoma [6]. TGCT usually appears with low signal in T1 and T2 sequences and moderate enhancement in T1 after intravenous contrast injection.
Case report
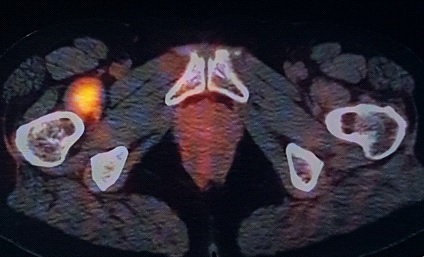
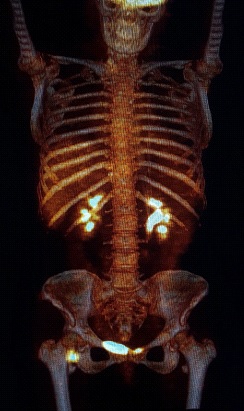
A 50-year-old woman, affected by ulcerated dorsal melanoma (Breslow 1,3 mm; Clark’s level IV, stage IIA, T2b N0 M0) treated with excision surgery and sentinel node biopsy in 2017, was admitted to our institution for unspecific rise of CEA (Carcinoembryonic Antigen). Follow-up FDG PET-CT (Discovery ST8, GEHC) was performed for whole body evaluation, demonstrating an intense focal uptake (SUV max 10.9) corresponding to the peritrochanteric medial part of right iliopsoas muscle. The FDG finding was suspected for malignancy, then, Ultrasonography (US) was required to guide further imaging and biopsy. US showed weak hypoechoic area in the iliopsoas muscle without significant diagnostic contributions.
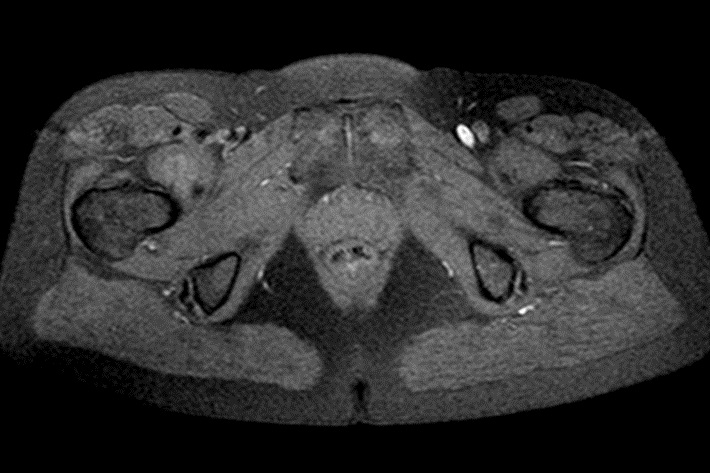
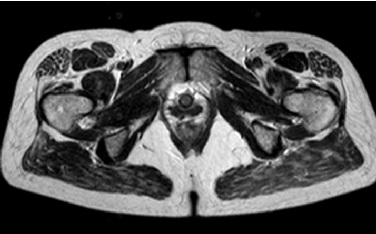
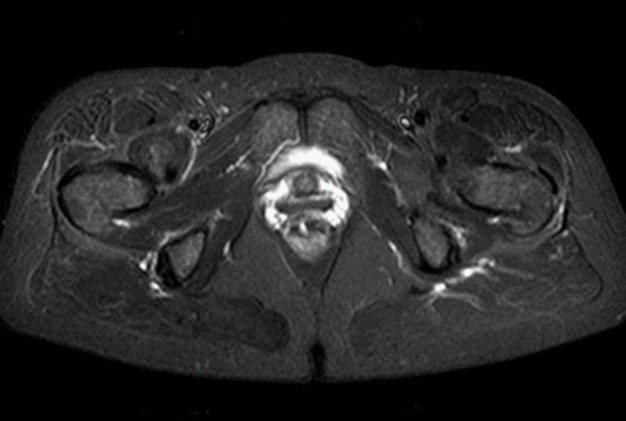
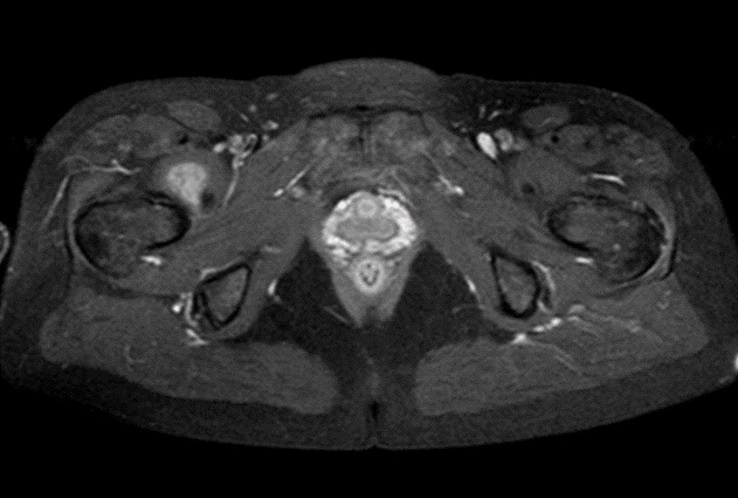
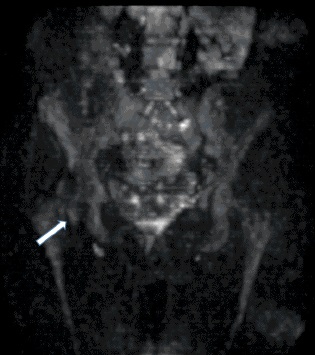
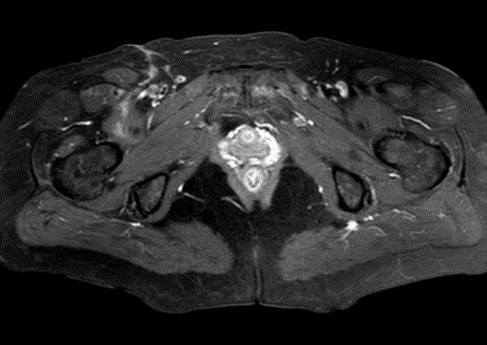
MRI examination was performed (Philips Achieva 1.5 T, release 5.3.1.3) showing, in the same area, a lesion with slightly high signal intensity in T1 (Figure 2a), isointense lesion compared with the surrounding muscle in TSE T2-weighted sequence (Figure 2b) in which only the “Mass-Effect” can be observed in the right iliopsoas muscle, low to intermediate signal (with small foci of hyperintensity included) in T2 FAT-SAT (Figure 2c). Intense ad homogeneous enhancement after intravenous contrast medium in T1 Fat-Suppressed sequence was observed (Figure 3). The lesion presents well-defined margins without infiltration in the surrounding tissue and no peri-lesional oedema. We also performed the Diffusional sequence (DWIBS) with B-value = 800 (Figure 4), the only case of TGCT studied with Diffusion Imaging, based on our knowledge. The diffusional study showed a non-significant restriction (1.2 × 10-3 mm2 /s) inside the lesion. Typical melanoma pattern on MRI images is: variably hyperintense in T1 (due to different melanin content and consequent paramagnetic effect), hypointensity in T2, inhomogeneous hyperintensity in T2 Fat-Sat, significant restriction in DWI (<1 x 10-3 mm2 /s), intense enhancement after contrast medium [7,8,9,10]. Regarding DWI, the ADC values approximately or less than 1 x 10-3 mm2 /s have been described as a threshold for predicting malignancy in various tumors [11,12]. In particular, Katharina Erb-Eigner, Gregor Willerding et all. suggest that the ADC of ocular melanoma is well below 1 x 10-3 mm2 /s [13]. Therefore, the non-univocal interpretation of the findings, in particular for the MRI signal in T2 Fat-Sat (irregular hypointensity instead of high hyperintensity) and in DWI (absence of significant restriction instead of a restrictive value <1 x 10-3 mm2/s) made it necessary to be excised.
The histologic examination on macroscopically showed a white-grey tumor mass (3 x 2.9 x 1.8 cm) with tones from white to yellow in the histological section. The microscopic exam showed small stromal mononuclear cells, stromal fibrosis, osteoclast-like a multinucleated giant cells, consistent with diagnosis of Tenosynovial Giant Cell Tumor. The patient is actually free of disease, showing no signs of loco-regional recurrence of TGCT (Figure 5).
Discussion
Our case shows a rare benign lesion of iliopsoas muscle coexisting in a patient with history of aggressive malignancy. The patient on the basis of FDG PET-CT, performed for routine follow up, was suspected for disease recurrence. Subsequent US and MRI suggested for benign muscular lesion but were inconclusive to exclude a metastasis from cutaneous melanoma. The diagnosis of TGCT was finally obtained after surgical removal. The FDG PET-CT is widely used as imaging tool for melanoma patients but false positive findings are frequently reported in benign disease such as infection, inflammation or vascular abnormalities. In presence of focal pattern FDG uptake with intensity higher than surrounding tissues, aggressive malignancies are generally suspected. In this case SUV max was particularly high (10.9) supporting the hypothesis of muscular metastases from ulcerated aggressive melanoma. In order to obtain a definitive diagnosis and to guide biopsy, further imaging was required. In particular contrast enhanced MRI was considered the imaging of choice to confirm the muscular metastases. Another sequence that has a certain specificity is the T2 sequence. Although MRI appearance of soft tissue malignant lesions may be indeterminate, often displaying varying degrees of T2 hyperintensity; in particular a subset of tumor-like lesions may exhibit prominent areas of T2 hypointensity relative to skeletal muscle. These include endometriosis, tenosynovial giant cell tumor, arteriovenous malformation, tumoral calcinosis, amyloidosis, low-grade fibromyxoid sarcoma, myxofibrosarcoma, peripheral nerve sheath tumors, dedifferentiated liposarcoma. Low-intensity signals from Diffuse-TGCT are frequently seen on T2-weighted MRI. This is attributed to hemosiderin deposition, known as the “Blooming Effect”[14]. Although there are no references in the literature on the behavior of TGCT in Diffusion MRI, the absence of significant restriction showed in our case, is compatible with non- aggressive lesion.
The MRI has revealed a formation, probably benign and, above all, has allowed us to exclude the melanoma metastasis. The melanoma metastasis findings in MRI are hyperintense signal in T1, variously hypointense signal in T2, hyperintensity in T2 Fat-Sat, significant restriction in DWI and peripheral rim enhancement with a heterogeneous pattern in T1 weighted scan after intravenous contrast enhancement. Instead, our lesion was slightly hyperintense in T1, low to intermediate signal intensity in T2 weighted scan and in T2 Fat-Sat (with small foci of hyperintensity included), no significant restriction in DWI, intense and homogeneous enhancement in T1 after intravenous contrast medium.
Our case report shows that in the differential diagnosis of malignant lesion with FDG uptake there is also TGCT and for this reason can be useful continue the diagnostic process with MRI. So, when we have lesions with FDG uptake in PET/CT, but dubious nature due to unusual site, it would be useful to continue with MRI examination to make a differential diagnosis.
Furthermore, our case report expands the few data available in literature about the Tenosynovial Giant Cell Tumor (TGCT) involving exclusively the muscles. Somerhausen et al [15] reviewed 8 cases of TCGT involving exclusively muscles. More recently cases were reported in two papers in 2015 ( Yoo Jin Lee et al) [16] and 2016 (Takeuchi et al) [1]. It’s clear that there is a lack of information about these rare forms of TGCT. It would be desirable to describe the rare cases encountered to improve the knowledge about these tumors that represent a real diagnostic challenge for Radiologists and Pathologists. We managed to diagnose of this exclusively muscular TGCT using MR and PETCT. With MR we could identify the characteristic of the lesion and we have the diagnostic suspicion of TGCT, that was finally confirmed by histopathologic examination. Using MR, we also classified the neoplasm according to a severity-scale, created by Mastboom et al in 2018. This classification is of meaningful prognostic importance.
The severity‘s classification of TGCT by Mastboom et al. is based on the definition of 4 MRI parameters:
- type of TGCT
- articular involvement, cartilage-covered bone invasion
- involvement of muscular/tendinous tissue
- involvement ligaments or neurovascular structures.
On the basis of these 4 parameters, TGCT severity subtypes were established:
1. Mild localized, either intra- or extra-articular involvement, without involvement of muscular/tendinous tissue/ligaments.
2. Severe localized, either intra - or extra-articular lesions and either or both involvement of muscular/tendinous tissue/ligaments.
3. Moderate diffuse, with intra - and/or extra-articular disease without and involvement of muscular/tendinous tissue/ ligaments.
4. Severe diffuse, including intra - and extra-articular involvement and involvement of at least one of the three structures (muscular/tendinous tissue/ligaments).
These four subtypes showed a clinically relevant or significant prognostic value for recurrent disease. In our case report, we describe a severe localized subtype, because it’s localized, extra-articular, with muscular involvement. These subtypes have a Recurrent Free Survival at 4 years of 88%. This tumor must be to consider benign. The follow-up can be less frequent than the one of diffuse subtypes.
Conclusion
In the differential diagnosis of malignant lesion with FDG uptake in PET/CT there is also TGCT and for this reason can be useful continue the diagnostic process with MRI. It is important to consider the possibility that a lesion of soft tissue with FDG uptake at PET/CT, slightly hyperintense in T1, low to intermediate signal intensity in T2 weighted scans, no significant restriction in DWI and intense enhancement in T1 after intravenous contrast medium, could be a rare benign lesion, as a muscular TGCT. In particular, we underline that the determining sequences in excluding malignant lesions were the DWI and T2 Fat-Sat. Therefore, based on our experience, in the suspicion of malignant lesions, it is appropriate to include the above sequences in the MRI study protocol.
Conflict of interest: The authors disclosure of any personal or financial support or author involvement with organization with financial interest in the subject matter or any actual or potential conflict of interest.
Acknowledgements: special thanks to Andrea Teodoro Radiographer.
References
- Takeuchi A, Yamamoto N, Hayashi K. Tenosynovial giant cell tumors in unusual locations detected by positron emission tomography imaging confused with malignant tumors: report of two cases. BMC Musculoskelet Disord. 2016; 17: 180.
- Mastboom MJL, Verspoor FGM, anff DFH, Dijkstra PDS. Severity classification of Tenosynovial Giant Cell Tumours on MR imaging. Surgical Oncology. 2018; 27: 544-550.
- Lee YJ, Kang Y, Jung J, Kim S, Kim CH. Intramuscular Tenosynovial Giant Cell Tumor, Diffuse-Type. J Pathol Transl Med. 2016; 50: 306-308.
- Gouin F, Noailles T. Localized and diffuse forms of tenosynovial giant cell tumor (formerly giant cell tumor of the tendon sheath and pigmented villonodular synovitis) Orthopaedics & Traumatology: Surgery & Research. 2017; 103: S91-S97.
- Mastboom MJL, Verspoor FGM, Verschoor AJ. Higher incidence rates than previously known in tenosynovial giant cell tumors. Acta Orthop. 2017; 88: 688–694.
- A. Greenspan J. Beltran “Orthopaedic Imaging- A pratical approach”.Verduci Editore. 2016; 23: 913-918.
- Gómez-León N, Pacheco-Barcia V, Ballesteros AI, Fraga J, Colomer R. Skeletal muscle and solitary bone metastases from malignant melanoma: multimodality imaging and oncological outcome. Melanoma Res. 2018; 28: 562-570.
- Namikawa K, Yamazaki N. Metastatic melanoma on the abdominal wall. Jpn J Clin Oncol. 2009; 39: 336.
- Yoshioka H, Itai Y, Niitsu M, Fujiwara M, Watanabe T, Satomi H, Otsuka F. Intramuscular metastasis from malignant melanoma: MR findings. Skeletal Radiol. 1999; 28: 714-716.
- Falappa P, Pennasilico GM, Arcuri PP, Laschena F, Concolini F. Intramuscolar metastasis from malignant melanoma: MR findings. A case report. Radiol Med. 2000; 100; 62-64.
- Sepahdari AR, Aakalu VK, Setabutr P, Shiehmorteza M, Naheedy JH, et al. Indeterminate orbital masses: Restricted diffusion at MR imaging with echo-planar diffusion-weighted imaging predicts malignancy. Radiology. 2010; 256: 554-564.
- Fornasa F, Nesoti MV, Bovo C, Bonavina MG. Diffusion-weighted magnetic resonance imaging in the characterization of axillary lymph nodes in patients with breast cancer. J Magn Reson Imaging. 2012; 36: 858-864.
- Erb-Eigner K, Willerding G, Taupitz M, Hamm B, Asbach P. Diffusion-weighted imaging of ocular melanoma. Invest Radiol. 2013; 48: 702-707.
- Verspoor FGM, Mastboom MJL, Weijs WLJ, Koetsveld AC, Schreuder HWB, et al. Treatments of tenosynovial giant cell tumours of the temperomandibular joint: A report of three cases and a review of literature. Int J Oral Maxillofac Surg. 2018; 47: 1288-1294.
- omerhausen NS, Fletcher CD. Diffuse-type giant cell tumor: clinicopathologic and immunohistochemical analysis of 50 cases with extraarticular disease. Am J Surg Pathol. 2000; 24: 479-492.
- Lee YJ, Kang Y, Jung J, Kim S, Kim CH. Intramuscular Tenosynovial Giant Cell Tumor, Diffuse Type - J Pathol Transl Med. 2016; 50: 306–308.