Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Diminished distal dorsal finger wrinkling in patients with sporadic inclusion body myositis
Ava Yun Lin1; Benjamin Koo2; Leo H Wang3*
1 Department of Pediatrics, Division of Pediatric Neurology, University of Michigan, Ann Arbor, MI, USA.
2 Kaiser Permanente, 7101 NE 137th Ave, Vancouver, WA 98682, USA.
3 Department of Neurology, University of Washington, Seattle, Washington, USA.
*Corresponding Author: Leo H Wang
Department of Neurology, University of Washington,
Seattle, Washington, University of Washington Medical Center, 356465, 1959 NE Pacific Street. Seattle, WA
98195-6465, USA.
Email: leowang@uw.edu
Received : Jun 25, 2021
Accepted : Aug 04, 2021
Published : Aug 10, 2021
Archived : www.jcimcr.org
Copyright : © Wang LH (2021).
Abstract
Sporadic Inclusion Body Myositis (IBM) is the most common acquired myopathy after the age of 45. Often there is a delay in diagnosis as the disease may be mistaken for other inflammatory myopathies and other neuromuscular disorders such as motor neuron disease. One of the hallmarks of the disease is distal finger flexor weakness but non-neurologists who see IBM may not have the expertise to make this assessment. Other clinical signs that require less expertise may be of value in identifying this disorder.
We present three cases of patients with IBM who had loss of wrinkling of the dorsal distal interphalangeal joints of the fingers that was not seen in any control patients who had no finger flexor weakness.
The loss of wrinkling of the dorsal distal interphalangeal joints of the fingers is an additional physical exam finding that could distinguish IBM from other neuromuscular disorders.
Keywords: Clinical neurology examination; all neuromuscular disease; muscle disease; inclusion body myositis.
Citation: Lin AY, Koo B, Wang LH. Diminished distal dorsal finger wrinkling in patients with sporadic inclusion body myositis. J Clin Images Med Case Rep. 2021; 2(4): 1260.
Introduction
Sporadic Inclusion Body Myositis (IBM) is the most common acquired myopathy after the age of 45 [1,2]. However, the diagnosis can be hard to make, with frequent delay in diagnosis of five to eight years from symptom onset [3,4]. Most patients receive another diagnosis - with same percentage of patients being first diagnosed with motor neuron disease as IBM [4]. Both present as painless weakness that can be asymmetric and/or accompanied by dysphagia.
Finger flexor weakness is a hallmark of IBM. In particular, selective weakness of the flexor digitorum profundi and flexor pollicis longus with sparing of long finger extensors and intrinsic hand muscles typically distinguishes IBM from amyotrophic lateral sclerosis [5,6]. Ideally, providers should always test for finger flexor weakness in these types of patients, but this is not always done as part of the routine physical/neurological exam and can be missed by non-muscle specialists. We present a case series of three patients that suggest that looking at loss of creases may be an additional exam finding that raises suspicion for finger flexor weakness, and in the appropriate clinical scenario, aid in diagnosing IBM.
Case presentations
Case 1
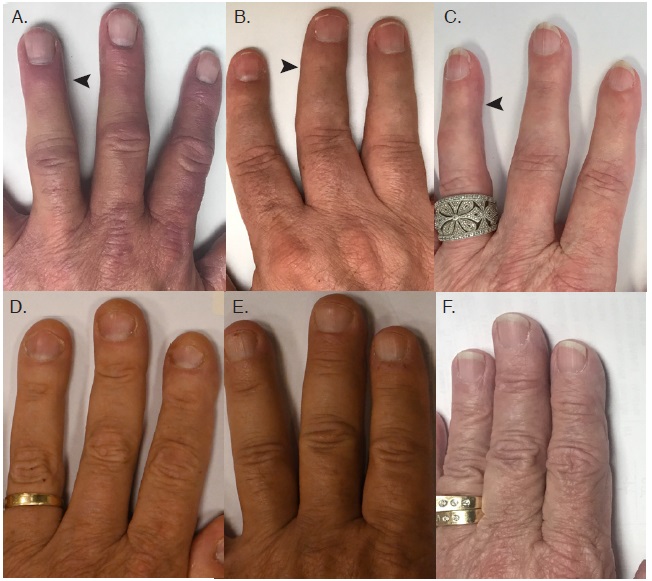
61-year-old male with type 2 diabetes mellitus presented with swallowing difficulty for 4-5 months. Upon further questioning, the patient reported seven years of progressive proximal weakness involving lower extremities more than upper. Amyotrophic lateral sclerosis was the initial top differential until the attending physician noticed striking loss of dorsal Distal Interphalangeal (DIP) creases (Panel A). This finding prompted further examination, which revealed significant weakness in distal finger flexors. The left 4th digit was most affected with at least a 1/5 strength on the MRC scale. The other digits shown in Panel A were less affected (at least 3/5), but still showed dorsal crease loss compared to healthy age-matched control (Panel D). Grip strength by Jamar handheld dynamometry showed diminished grip strength - 9 kg - in the left hand. There were no clear limitations to the passive range of motion at the DIP. Creatine kinase levels were in the upper 200s. His Electromyogram (EMG) showed myopathic changes. Muscle biopsy showed nearly end-stage myopathy, myonecrosis and regeneration, focal endomysial T-lymphocytic infiltration and rimmed vacuoles, consistent with sporadic Inclusion Body Myositis (IBM).
Case 2
61-year-old male who presented with three-year history of difficulty arising from a chair. He noted weakness in the right finger flexors (panel B) that was insidious for 10 years spreading to the left finger flexors in the prior year. His EMG showed myopathic changes; muscle biopsy endomysial inflammation without rimmed vacuoles. He was treated with multiple immunosuppressants to no avail. His slow progression, striking involvement of deep finger flexors, and lack of response to immunosuppression, was most consistent with sporadic IBM.
Case 3
75-year-old female who presented with 13-year-old history of slowly progressive difficulty getting up from the floor or a chair. This started insidiously without significant exacerbations or clear improvements. Over the last ten years, she has had progressive weakness of her knee extensors and her arms, especially her left-hand grip which was most noticeable after breaking her wrist. Over the years, she has also developed dysphagia for solids such as bananas or muffins but not liquids. Her creatine kinase was reportedly over 2000. A muscle biopsy of right quadriceps showed endomysial inflammation, and she was treated with immunosuppressants such as prednisone, azathioprine, methotrexate, mycophenolate, rituximab with no clear improvement in strength. Prednisone caused avascular necrosis of the left hip requiring a hip replacement; azathioprine increased liver function tests; methotrexate oral ulcers; mycophenolate urinary tract infections. Her left fingers also showed loss of finger creases (panel C). Her slow progression, deep finger flexors and knee extensor weakness, and lack of response to immunosuppression made her clinical diagnosis to be sporadic IBM.
All three patients had finger flexor weakness and loss of dorsal finger creases (arrows pointing the loss of distal finger creases). The fingers appeared thinner with sometimes almost translucent skin overlying the distal interphalangeal joint. In comparison, three control patients (D-F) matched for sex and age with no finger flexor weakness did not show loss of dorsal finger creases.
Discussion
Selective distal finger flexor weakness is a hallmark of IBM, but can be challenging to identify. Manual strength testing is complicated by degree of patient cooperation, which is influenced by pain (arthritis, past fractures, etc.), mental status, and other hand deformities. An adjunctive, easy to visualize, physical exam sign associated with finger flexor weakness can greatly help in early appropriate detection of IBM.
Kobayashi et al. Previously reported the fist sign [7] – an inability to bury the finger nails into the palm of the hand when attempting to form a fist due to selective distal finger flexor weakness in IBM. Our patients also had subtle positive fist sign, along with a striking loss in dorsal DIP creases.
Acquired reduction in skin wrinkles can result from induced muscle weakness, eg. the cosmetic use of botulinum toxin [8]. Decreased palmar, plantar and finger creases are reported in several genetic disorders with congenital weakness that is irrespective of ethnic origin [9]. Our crease sign (or lack of) is a feasible adjunct exam finding, along with the fist sign, for detection of distal finger flexor weakness. Using this sign to think about inclusion body myositis early may prevent the treatment of these patients with immunosuppressants, which have no clinical benefit and possible side effects.
References
- Lloyd TE, Mammen AL, Amato AA, et al. Evaluation and construction of diagnostic criteria for inclusion body myositis. Neurology. 2014; 83: 426-433.
- Badrising UA, Maat-Schieman ML, van Houwelingen JC, et al. Inclusion body myositis. Clinical features and clinical course of the disease in 64 patients. J Neurol. 2005; 252: 1448-1454.
- Needham M, Corbett A, Day T, et al. Prevalence of sporadic inclusion body myositis and factors contributing to delayed diagnosis. J Clin Neurosci. 2008; 15: 1350-1353.
- Badrising UA, Maat-Schieman M, van Duinen SG, et al. Epidemiology of inclusion body myositis in the Netherlands: a nationwide study. Neurology. 2000; 55: 1385-1387.
- Menon P, Bae JS, Mioshi E, et al. Split-hand plus sign in ALS: differential involvement of the flexor pollicis longus and intrinsic hand muscles. Amyotroph Lateral Scler Frontotemporal Degener 2013; 14: 315-318.
- Shemesh A, Arkadir D, Gotkine M. Relative preservation of finger flexion in amyotrophic lateral sclerosis. J Neurol Sci. 2016; 361: 128-130.
- Kobayashi Z, Fukatsu E, Itaya S, Akaza M, Ota K, Numasawa Y, et al. Fist sign in inclusion body myositis. Neuromuscul Disord. 2017; 27: 385–386.
- Gart MS, Gutowski KA. Overview of Botulinum Toxins for Aesthetic Uses. Clin Plast Surg 2016; 43: 459–471.
- Schaumann BA, Kimura S. Palmar, plantar, and digital flexion creases: morphologic and clinical considerations. Birth Defects Orig Artic Ser. 1991; 27: 229–252.