Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
A rare case of transformation of autoimmune thyroiditis into Grave’s disease after COVID-19 disease
Kravchenko V*; Rakov O
SI “Institute of Endocrinology and Metabolism. VP Komissarenko National Academy of Medical Sciences of Ukraine “, 04114 Kyiv, street Vyshgorodskaya, 69, Ukraine.*Corresponding Author: Kravchenko Victor
Radiology Department, Fann University Hospital
Center, 5035 Avenue Cheikh Anta DIOP, Dakar,
Senegal.
Email: endocrinolog@ukr.net
Received : Jun 02, 2021
Accepted : Aug 16, 2021
Published : Aug 20, 2021
Archived : www.jcimcr.org
Copyright : © Victor K (2021).
Abstract
Autoimmune Thyroiditis (AIT) and Graves’ disease are autoimmune diseases with the opposite state of thyroid function: hypothyroidism and thyrotoxicosis. Typically, thyrotoxicosis can progress to thyroiditis and eventually hypothyroidism. The transformation of autoimmune thyroiditis into Graves’ disease is a very rare event. SARS-CoV-2 viral infection shares autoimmune mechanisms with common diseases of the thyroid gland and may be the cause of their diseases. We have described a case of Graves’ disease after suffering Covid-19 in a patient with autoimmune thyroiditis and hypothyroidism.
Abbreviations: TSH: Thyroid Stimulating Hormone; T4: Total Thyroxine; T3: Triiodothyronine; FT4: Free Thyroxine Level; Tsab: Thyroid Stimulating Antibodies; Abrtsh Antibodies To The TSH Receptor.
Citation: Kravchenko V, Rakov O. A rare case of transformation of autoimmune thyroiditis into Grave’s disease after COVID-19 disease. J Clin Images Med Case Rep. 2021; 2(4): 1272.
Introduction
Autoimmune diseases of the thyroid gland are the most common among organ autoimmune diseases [1-3]. Among them are autoimmune thyroiditis and Graves’ disease. Various links of acquired and innate immunity, cellular and humoral components are involved in the pathogenesis of these diseases [4,5]. The main antigens for autoimmune reactions are thyroglobulin, thyroxine peroxidase and thyroid stimulating hormone receptor. For autoimmune thyroiditis, a characteristic feature is the destruction of thyroid cells as a result of a cellular immune response, followed by the development of hypothyroidism. In Graves’ disease, stimulation of the thyroid gland due to the presence of humoral antibodies to the thyroid-stimulating hormone receptor on thyrocytes and the development of thyrotoxicosis comes to the fore. Among the diseases associated with the SARS-CoV-2 coronavirus, attention is drawn to those related to the thyroid gland [6-8]. The number of reports of such an association is rapidly increasing. The thyroid gland and viral infection are involved in a complex interaction through hormones and immunomodulatory signaling molecules. These connections have been established under physiological and pathological conditions [9-11]. In this article, a case of transformation of autoimmune thyroiditis with hypothyroidism into Graves’ disease after a disease on Covid is considered.
Case report
March 21, 2018 Patient N., aged 39, was registered at the polyclinic of the Institute of Endocrinology with complaints of general weakness of joint pain, fatigue, drowsiness, memory loss, decreased ability to work, dry skin, progressive weight gain, facial swelling, hair loss on the head. Ultrasound examination showed that the thyroid gland was not enlarged, the heterogeneity of its echostructure and a diffuse decrease in its echogenicity were noted. In a laboratory study, the TSH content in the blood increased to 26.9 µU/ml (reference values 0.4-4.0), the level of free T4 in the blood serum was reduced to 0.34 ng/ dL (reference values 0.61-1.12 ng/dL), free T3 - 1.42 pg/ml (reference values 2.3-4.2 ng/ml), there was a significant increase in antibodies to TPO> 1000 U/ml (reference values <35 U/ml) and thyroglobulin 277 U/ml (reference <40 U/ml). The patient was diagnosed with AIT combined with hypothyroidism. She was prescribed thyroxine therapy with a gradual increase in the dose to 100 mg per day. Substitution therapy was carried out for 3 years with a satisfactory state of health.
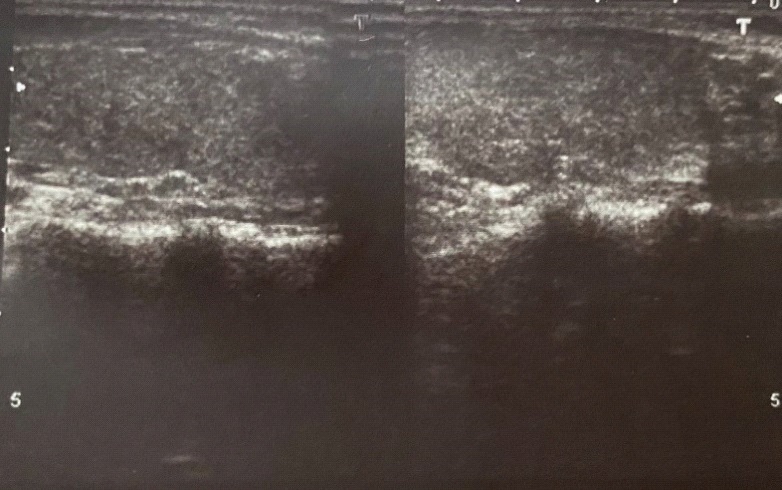
In February 2021, patient N. again turned to an endocrinologist at the polyclinic of the Institute of Endocrinology with complaints of severe general weakness, irritability, sweating, palpitations, tremors in the hands, high blood pressure, weight loss of 8 kg over the past 3 weeks. A general examination revealed a diffuse increase in the thyroid gland of the 2nd degree (according to the WHO classification), palpitations up to 100 beats per minute. Examination of the eye retraction revealed no abnormalities in the oculomotor function. Reflexes are unchanged. Neurological status was normal. Moderate postural tremor. An outpatient thyroid ultrasound examination was performed, revealing a diffusely heterogeneous structure (Figure 1). With a history, it turned out that the patient had been ill with COVID-19 about a month ago and was mainly worried about dry cough, muscle pain, loss of smell. At the time of illness, saturation was within 99%. Reverse Transcription Polymerase Chain Reaction (RT-PCR) for SARS-CoV-2 tested positive. COVID-19 treatment took place in home quarantine for 14 days. Subsequently, the patient went to work in a satisfactory condition, but after a month she felt a deterioration in her health and turned to an endocrinologist. Due to the fact that chronic autoimmune thyroiditis is often the cause of subacute thyroiditis and the latter’s disease increases with viral infections, including COVID-19 [12,13], with high titers of antibodies to viruses. We analyzed the possibility of developing this disease. Although the thyroid gland was enlarged, it was painless on palpation. Reverse transcription polymerase chain reaction (RT-PCR) for COVID-19 was negative.
Laboratory blood test results showed free thyroxine (T4) and free triiodothyronine (T3) levels increased to 7.3 ng/dL and 2.74 pg/ml, respectively. The level of thyroid Stimulating Hormone (TSH) was low - 0.01 µU/ml.
At the time of the examination, iatrogenic thyrotoxicosis (medication) was suspected, as a result of which the patient was discontinued from taking levothyroxine and was prescribed β-blockers (anaprilin 20 mg x 2 times a day) and sedatives. 4 weeks after the next consultation, the patient’s condition remained the same, trembling in the whole body intensified, palpitations, sweating, and severe irritability worried.
The results of repeated laboratory tests revealed an abnormal increase in the level of free T3 and T4, the values of which were 11.6 pg/ml and 4.56 ng/dL, respectively. The content of Thyroid Stimulating Hormone (TSH) was low - 0.001 µU/ml. The indicator of antibodies to the TSH receptor (AbrTSH) significant y exceeded the reference values - 8.2 U/L (reference values <0.55 - negative). Tests for the presence of antibodies to thyroid peroxidase (AbTPO) continued to be elevated.
Graves’ disease was diagnosed. We assume that its development against the background of autoimmune thyroiditis, combined with hypothyroidism, is associated with COVID-19.
The patient was prescribed tyrosol at a dose of 30 mg per day and 5 weeks after taking the drug, the T4 s values were 0.98 ng/dL, free T3 - 3.78 pg/ml, and the TSH level remained suppressed and amounted to 0.06 µU/ml. Clinically, the patient noted a significant improvement in overall well-being. At the moment, the patient continues to take tyrosol at a dose of 5 mg per day and, based on the deficiency of vitamin D in the diet of the population of Ukraine, vitamin D at a dose of 2000 IU per day [14].
Discussion
In this case, the patient received replacement therapy with 100 μg of levothyroxine per day for 3 years for autoimmune thyroiditis and hypothyroidism, which were confirmed by clinical and laboratory studies. When the patient 3 years later turned to the polyclinic of the institute with symptoms of thyrotoxicosis, the first thing that we suspected was iatrogenic hyperthyroidism, i.e. thyrotoxicosis caused by an overdose of levothyroxine. However, some time after the drug was discontinued, there was no improvement, and the clinical and laboratory parameters only worsened. In this regard, the levels of AbrTSH in the blood serum were determined and the diagnosis of hypothyroidism against the background of autoimmune thyroiditis was replaced by Graves' disease, which arose a month after suffering COVID-19.
Graves' disease and AIT are two spectrum of autoimmune thyroid disease. Both nosoological forms are polyetiological, with little-studied pathogenesis. Confirmed primary hypothyroidism is a stable or gradually progressive disease that requires lifelong replacement therapy with levothyroxine drugs. But in rare cases, stability is impaired and hypothyroidism can transform into hyperthyroidism.
In the literature available to us, there are rare reports of cases of transition from hypothyroidism to hyperthyroidism [15-18]. In this transformation, none of the described cases was associated with the influence of an infectious factor. In our example, the indicated transition of primary hypothyroidism to Graves' disease, which began a month after the confirmed COVID-19, and requires thyrostatic therapy.
To date, it is not known why a disease with established hypothyroidism subsequently progresses to hyperthyroidism. One of the important putative mechanisms is the presence of various autoantibodies, including antibodies that stimulate the thyroid gland, antibodies that block thyroid stimulation, and the response of the thyroid gland to these antibodies [19]. We assume that the direct action of the SARS-CoV-2 virus on the follicular cells of the thyroid gland through the ACE2 receptor with subsequent stimulation of TSAb (thyroid-stimulating antibodies to the TSH receptor) played a role in the transformation of hypothyroidism into hyperthyroidism.
References
- Rodriguez Y, Rojas M, Monsalve DM et al. Latent autoimmune thyroid disease J Transl Autoimmun. 2020.
- McLeod DS, Cooper DS. The incidence and prevalence of thyroid autoimmunity. Rew. Cooper Endocrine 2012; 42 : 252-265.
- Кravchenko V, Тоvkay O, Rakov O, Тronko M. Epidemiology of autoimmune thyroiditis. International Journal Of Endocrinology (Ukraine). 2021; 2 : 61-69.
- Ramos-Levi A, Marazuela M. Pathogenesis of thyroid autoimmune disease: the role of cellular mechanisms Endocrinol Nutr. 2016; 63: 421-429 .
- Zakharchenko T, Kravchenko V. Peculiarities of innate and adaptive immunity in the pathogenesis of thyroid autoimmune diseases. Immunocorrection (part 1) INTERNATIONAL JOURNAL OF ENDOCRINOLOGY (Ukraine). 2020; 16: 564-576.
- Gorini F, Bianchi F, Iervasi G. COVID-19 and Thyroid: Progress and Prospects Int J Environ Res Public Health. 2020; 17: 6630.
- Muller I, Cannavaro D, Dazzi D, Covelli D, Mantovani G et al. SARS-CoV-2-related atypical thyroiditis Lancet Diabetes Endocrinol. 2020; 8: 739-741.
- Lisco G, Tullio A, Jirillo E, Giagulli V, Pergola G et al. Thyroid and COVID-19: A review on pathophysiological, clinical and organizational aspects J Endocrinol Invest. 2021; 1-14.
- De Vito P, Incerpi S, Pedersen JZ, Luly P, Davis FB, et al. Thyroid hormones as modulators of immune activities at the cellular level. Thyroid. 2011; 21: 879-890.
- Tomer Y, Davies TF. Infection, thyroid disease, and autoimmunity. Endocr Rev. 1993; 14: 107-20.
- Franceschi C, Ostan R, Mariotti S, Monti D, Vitale G. The aging thyroid: A reappraisal within the geroscience integrated perspective. Endocr Rev. 2019; 40: 1250-1270.
- Brancatella A, Ricci D, Viola N, Sgro D, Santini F, Latrofa F. Subacute thyroiditis after SARS-CoV-2 infection. J. Clin. Endocrinol. Metab. 2020; 105: 1-4.
- Ippolito S, Dentali F, Tanda ML. SARS?CoV?2: a potential trigger for subacute thyroiditis? Insights from a case report. J. Endocrinol. Investig. 2020; 43: 1171-1172.
- Kravchenko V, Rakov O, Kovzun E. Status vitamin D in Ukraine patients with Grave’s Disease Journal of Endocrinology and Thyroid Research. 2021; 5.
- Ruhla M, Jepanek E, Skiba A, Chepczynski R, Sovinski J. Gravideosis Graves after primary hypothyroidism due to Hashimoto’s thyroiditis in the case of thyroid haemiaenesis: A clinical case. Neuro Endocrinol Lett. 2008; 29: 55-58.
- Bell P, Sinnamon D, Smyth P, Drexhage H, Haire M, Bottazzo G, Atkinson A. Hyperthyroidism following primary hypothyroidism in association with polyendocrine autoimmunity. Acta Endocrinol (Copenh). 1985; 108: 491-497.
- Cho B, Shong Y, Lee H, Koh C, Min H. Graves’ hyperthyroidism following primary hypothyroidism: sequential changes in various activities of thyrotropin receptor antibodies. Acta Endocrinol (Copenh). 1989; 120: 447-450.
- Fatourechi V, McConahey W, Woolner L. Hyperthyroidism associated with histological Hashimoto’s thyroiditis. Mayo Clinic Proceedings. 46; 682-686.
- Takasu N, Yamashiro K, Ochi Y, Sato I, Nagata A, et al. TSBAb and patients with Graves hyperthyroidism. Horm Metab Res. 2001; 33: 232-237.