Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Acute longitudinally extensive transverse myelitis: A case report
Asma Ben Mabrouk1,3*; Jihene Chelli1,3; Sondess Arfa1,3; Houcem Elomma Mrabet1,3; Rafik Machraoui2,3; Fatma Larbi Ammari1,3; Olfa Berriche1,3,4
1 Internal Medicine and Endocrinology Department, University Hospital Taher Sfar 5100 Mahdia Tunisia.
2 Neurology Department, University Hospital Taher Sfar 5100 Mahdia Tunisia.
3 Faculty of Medicine -University of Monastir, Monastir
4 Biochemistry Laboratory, LR12ES05 LR-NAFS Nutrition-Functional Food and Vascular Health, Faculty of Medicine, University of Monastir.
*Corresponding Author: Asma Ben Mabrouk
Internal Medicine and Endocrinology Department, University Hospital Taher Sfar 5100 Mahdia
Tunisia.
Email: asma.ben.mabrouk@gmail.com
Received : July 06, 2021
Accepted : Aug 17, 2021
Published : Aug 20, 2021
Archived : www.jcimcr.org
Copyright : © Ben Mabrouk A (2021).
Abstract
Introduction: Acute Longitudinally Extensive Transverse Myelitis (ALETM) poses a problem of etiological diagnosis. The etiologies are diverse; infectious, degenerative, inflammatory and auto-immune.
Case report: We are presenting the case of a 19 years old patient, with no prior medical history. He presented with acute flaccid paraplegia, urinary retention and fecal incontinence. The magnetic resonance imaging concluded to ALETM and the etiological investigation allowed us to confirm the diagnosis of myelitis inaugurating systemic lupus erythematosus. The treatment was based on the association of corticoids, cyclophosphamides and four plasma exchanges. No real improvement in the neurological deficiency was noted.
Conclusion: The lupus origin of ALETM, although rare, should be considered in young patients since it could, as in our case, the diagnostic circumstance of systemic lupus erythematosus.
Keywords: myelitis; lupus erythematosus; systemic; paraplegia.
Citation: Ben Mabrouk A, Chelli J, Arfa S, Mrabet HE, Machraoui R, et al. Acute longitudinally extensive transverse myelitis: A case report. J Clin Images Med Case Rep. 2021; 2(4): 1276.
Introduction
Acute Longitudinally Extensive Transverse Myelitis (ALETM) is an inflammation of the spinal cord in which lesions extend vertically over three or more vertebral segments. The Magnetic Resonance Imaging (MRI) is essential to diagnose this affection and help orientate the etiological quest [1]. The etiologies are diverse; infectious, degenerative, inflammatory and auto-immune [2].
Myelitis is a rare complication of Systemic Lupus Erythematosus (SLE) and an exceptional circumstance of diagnosis of SLE [3]. We are presenting the case of myelitis inaugurating systemic lupus erythematosus in a 19 years old male patient.
Case report
A 19 years old patient, with no prior medical history, was admitted in April 2019 after presenting with acute flaccid paraplegia, urinary retention and fecal incontinence. The patient presented, 10 days earlier, with fever, cough and vomiting. He had received antipyretics and doxycycline without any improvement.
On physical examination, the patient had a temperature of 38.5°, severe paraplegia with areflexia, and a sensory level T11 T12. No other anomaly of the neurological examination was found and there was no meningeal syndrome.
The complete blood count showed a total white blood cell count of 5930/mm3 with neutrophils count of 4670/mm3 and lymphocytes count of 810/mm3 . The patient had anemia with an hemoglobin level of 10.7g/dl and a mean corpuscular volume of 82.4 fl and platelet count of 297000/mm3.
The lumbar puncture removed a cloudy cerebrospinal fluid. Cell count showed a white blood count of 400/mm3 with neutrophils predominance (95%) associated with a high protein level (1.3 g/l) and low glucose level (1 mmol/l).
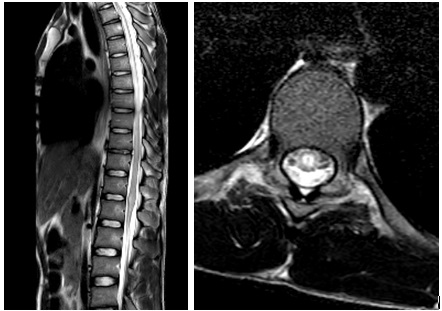
The magnetic resonance imaging was congruent with longitudinally extensive transverse myelitis, the lesions extend vertically from the vertebral disc T2-T3 to T9 with medullary cone involvement (Figure 1).
Initially, the infectious origin of the myelitis was highly suspected. Antibiotic treatment was initiated with a meningeal dosage of ceftriaxone and ofloxacin associated with dexamethasone 40 mg per day for 4 days. Blood and spinal fluid cultures were sterile.
The apyrexia was obtained at first but fever restarted after discontinuation of corticosteroids with right knee arthritis. Joint puncture removed a cloudy exudate with a protein level of 40 g/l and a white blood cells count of 1500/mm3 with neutrophils predominance (90%).
The screening for tuberculosis (skin test, Polymerase Chain Reaction (PCR) in the cerebrospinal fluid, sputum, urine and cerebrospinal fluid’s tests were negative for Mycobacterium tuberculosis), the serological tests for bacteria (Wright, Chlamydia, Mycoplasma, Lyme’s disease, syphilis) and the serological tests for virus (Human Immunodeficiency Virus (HIV), Cytomegalovirus (CMV), Epstein Barr Virus (EBV), West Nile, hepatitis A, B and C) were negative.
The autoimmune origin of the affection was suspected, screening for autoantibodies showed an elevated rate of Antinuclear Antibodies (ANA) 1/200 with positive anti-DNA (++), anti nucleosome (+++) and anti-SM/RNP(+). Perinuclear antineutrophil cytoplasmic antibodies p-ANCA were also positive (+) with negative antiphospholipid antibodies. Serum complement C3 and C4 concentrations were very low to be detected.
Since the patient presented 6 criteria of the SLICC classification for SLE: Myelitis, arthritis, urine protein >600 mg protein/24 h, lymphopenia, positive ANA and positive anti-DNA, and a score of 25 (>10) according to the 2019 European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) Classification Criteria for Systemic Lupus Erythematosus, the diagnosis of SLE was retained.
The treatment was started with 3 boli of methylprednisolone (1 g/day) relayed with high dose oral corticosteroids 1mg/kg/ day and hydroxychloroquine 400 mg/day, 5/7 days. Apyrexia was obtained with the second bolus of methylprednisolone and we noted a regression of the sensory level to L3-L4 with no motor improvement. The patient underwent 4 sessions of therapeutic plasma exchange and received 3 cures of cyclophosphamide, but this treatment was stopped after recurrent urinary tract infections. The relay treatment was azathioprine 100 mg/ day and prednisone 10 mg/day. The patient also followed a program of physical rehabilitation to ensure self-catheterization and cope with the motor deficiency. After a year follow-up, no change was noted in the patient’s sensory or motor impairment.
Discussion
The originality of this case is the acute onset of longitudinally extensive transverse myelitis, inaugurating SLE, in a young male with no prior medical history and the misleading findings of the spinal fluid analysis that orientate toward an infectious origin.
Acute transverse myelitis is, by definition, an inflammation of the spinal cord with a nadir that ranges between 4 hours and 21 days after the onset of symptoms [2]. Its incidence is estimated to 1 to 8 cases/million/year [3]. The longitudinally extensive aspect is established in the case of extension over three or more vertebral segments in T2-weighted MRI [4].
ALETM poses the problem of etiological diagnosis since this affection could be infectious, degenerative, inflammatory, and auto-immune. The most common cause is Devic's neuromyelitis optica, followed by vascular causes, then infections [2].
In our case, the infectious origin of the myelitis was initially evoked based on lumbar puncture results. This explains the prescription of antibiotics and corticosteroids for probable bacterial meningitis. In fact, neutrophils’ predominance in the cerebrospinal fluid is suggestive of bacterial or mycobacterial infection but could be found in sarcoidosis, Devic's neuromyelitis optica, autoimmune affections and viral infections. The elevated protein level is frequent. It is found in half cases of myelitis all etiologies included. The low glucose level is frequently seen in myelitis of infectious origin, bacterial or fungal. But it could be found in neuro-sarcoidosis, subarachnoid hemorrhage and SLE [5].
The infectious etiologies of ALETM could be due to bacteria, viruses, fungi and parasites [6].
Bacterial etiologies reported in the literature are syphilis, if there is a history of unprotected sex, tuberculosis in endemic zones, brucellosis in at-risk profession individuals, Lyme borreliosis even without the specific erythema, and chlamydia or mycoplasma especially in the presence of respiratory infection symptoms [1,6,7]. Viral etiologies could be due to DNA viruses (Herpes simplex virus, varicella zona virus, CMV, EBV and hepatitis B) or RNA viruses (mumps, dengue, rabies, Influenza Virus A, coxsachievirus, enterovirus, echovirus, West Nile virus, HIV and des hepatitis A and C viruses) [6,8,9]. A case of longitudinally ALETM was reported following acute COVID-19 infection [10].
Myelitis due to fungi (actinomycosis, aspergillosis and blastomycosis) and parasites (neurocystiarcasis, schistosomiasis, angiostrongylosis) are less common [1,9].
Transverse myelitis is a common manifestation of acquired demyelinating diseases, such as multiple sclerosis. Devic's neuromyelitis optica is to keep in mind even in the absence of optic lesions that could be delayed months or even years after medullar affection. The old vaccines of rabies were incriminated in the pathogenesis of myelitis [1,5,6,9,11–13]. The inflammatory causes of ALETM are mainly Behçet’s disease and sarcoidosis. The most reported autoimmune etiologies are SLE and Gougerot Sjogren syndrome. Idiopathic myelitis is the final diagnosis to retain after an exhaustive negative screening [14].
For our patient, the diagnosis of SLE was retained because of the association of 6 criteria of the SLICC classification and a score of 25 (>10) according to the 2019 European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) Classification Criteria for Systemic Lupus Erythematosus [15]. The male gender seems to predispose patients to neuropsychiatric, renal and cardiovascular manifestations of SLE [16].
The incidence of myelitis ranges between 1 and 3% of lupus patients. No predominance of either of the genders was reported [3]. When it occurs, myelitis is the circumstance of diagnosis of SLE in half cases. Longitudinally extensive forms consist of 71 to 75% of the cases. The mean age of patients, presenting with lupus myelitis is 29+/- 9.4 years [17]. The different studies concluded to an association between lupus myelitis and positive antiphospholipid antibodies. (64% to 80%). Microthrombi are incriminated in the genesis of medullar lesions [3,17,18]. The rapid onset of the neurological symptoms is a prognostic factor in lupus myelitis. The more rapid is the onset, the worst is the prognosis [19]. In cases of flaccid paraplegia, as in our patient, where the onset of a complete motor, sensory, and autonomic neurologic dysfunction is very rapid, the recuperation rates, whatever is the treatment, are about 10% [3,20].
The management of lupus myelitis remains controversial. Corticosteroids and cyclophosphamides are the most used treatments. Plasma exchanges were tried with a favorable outcome [21,22]. In refractory cases, Rituximab, and immunoglobulins were tried with promising results [23].
Conclusion
The etiologies of ALETM are diverse. LSE myelitis, although rare, should be considered in young patients with no prior medical history, since it be could, as in our case, the diagnostic circumstance of systemic lupus erythematosus.
Disclosure statement: The authors have no conflicts of interest relevant to this manuscript to disclose.
References
- Román GC, Barnes G, Benjamin S, Bowen JD, Cutter N, et al. Proposed diagnostic criteria and nosology of acute transverse myelitis. Neurology. 2002; 59: 499–505.
- Tobin WO, Weinshenker BG, Lucchinetti CF. Longitudinally extensive transverse myelitis. Curr Opin Neurol 2014; 27: 279–89.
- Li X, Xiao H, Pai P. Myelitis in systemic lupus erythematosus. 2017; 44: 18–22.
- Mirbagheri S, Eckart Sorte D, Zamora CA, Mossa-Basha M, Newsome SD. Evaluation and management of longitudinally extensive transverse myelitis: A guide for radiologists. Clin Radiol 2016; 71: 960–71.
- West TW, Hess C, Cree BAC. Acute transverse myelitis: Demyelinating, inflammatory, and infectious myelopathies. Semin Neurol. 2012; 32: 97–113.
- Collongues N, Papeix C, Zéphir H, Audoin B, Cotton F, et al. Cadre nosologique et stratégie diagnostique de la myélite aiguë transverse longitudinalement étendue. Rev Neurol (Paris) 2014; 170: 6–12.
- Erol I, Klçarslan B, Saygi S, Demir Ş, Alehan F. Acute transverse myelitis in a child with lyme disease and a review of literature. Pediatr Neurol. 2013; 48: 325–328.
- Andrade P, Figueiredo C, Carvalho C, Santos L, Sarmento A. Transverse myelitis and acute HIV infection: A case report. BMC Infect Dis. 2014; 14: 149.
- Bhat A, Naguwa S, Cheema G, Gershwin ME. The epidemiology of transverse myelitis. Autoimmun Rev. 2010; 9: A395–9.
- Fumery T, Baudar C, Ossemann M, London F. Longitudinally extensive transverse myelitis following acute COVID-19 infection. Mult Scler Relat Disord. 2021; 48.
- Boukasri C, Elalaoui O, Mebrouk Y. La myélite aiguë transverse longitudinalement étendue (MATLE): étude d’une cohorte de 12 cas. Rev Neurol (Paris). 2017; 173: S125.
- Lynn J. Myélite Transverse: Symptômes, Causes et Diagnostic · Symptômes cliniques: Causes de la Myélite transverse. 2010: 12–6.
- de Sèze J. Myélites aiguës transverses. Rev Med Interne 2009; 30: 1030–7.
- Choudhary A, Bhargava A, Khichar S, Pradhan S. Etiological spectrum, clinico-radiological profile and treatment outcomes of longitudinally extensive transverse myelitis–A prospective study from Northwest India. J Neuroimmunol. 2021; 351.
- Aringer M, Costenbader K, Daikh D, Brinks R, Mosca M, et al. 2019 European League Against Rheumatism/American College of Rheumatology Classification Criteria for Systemic Lupus Erythematosus. Arthritis Rheumatol. 2019; 71: 1400–12.
- Tan Tc, Fang H, Magder Ls, Petri Ma. Differences between Male and Female Systemic Lupus Erythematosus in a Multiethnic Population. J Rheumatol. 2012; 39: 759–69.
- Nardone R, Fitzgerald RT, Bailey A, Zuccoli G. Longitudinally extensive transverse myelitis in systemic lupus erythematosus : Case report and review of the literature. Clin Neurol Neurosurg 2015; 129: 57–61.
- Flores-silva FD, Sentı H, De ED. Natural history of longitudinally extensive transverse myelitis in 35 Hispanic patients with systemic lupus erythematosus: Good short- term functional outcome and paradoxical increase in long-term mortality. 2018: 1–8.
- White C, Leonard B, Patel A. Longitudinally extensive transverse myelitis: A catastrophic presentation of a flare-up of systemic lupus erythematosus. CMAJ. 2012; 184.
- Lefèvre G, Zéphir H, Warembourg F, Michelin E, Pruvo J, et al. Neurolupus (1 re partie). Description et démarche diagnostique et thérapeutique dans les manifestations neurologiques centrales et psychiatriques au cours du lupus érythémateux systémique Neuropsychiatric systemic lupus erythematosus (1st part). Cases. La Rev m?®decine Interne 2012; 33: 491–502.
- Mehmood T, Munir I, Abduraimova M, Antonio Ramirez M, Paghdal SM. McFarlane I. Longitudinally Extensive Transverse Myelitis Associated With Systemic Lupus Erythematosus: A Case Report and Literature Review. Am J Med Case Reports. 2019; 7: 244–9.
- Zahid AS, Mubashir A, Mirza SA, Naqvi IH, Talib A. Systemic Lupus Erythematosus Presenting as Longitudinally Extensive Transverse Myelitis and Nephritis: A Case Report. Cureus. 2018; 10.
- Lefèvre G, Zéphir H, Michelin E, Semah F, Warembourg F, et al. Neurolupus (2 e partie). Description des outils diagnostiques et thérapeutiques devant une manifestation psychiatrique ou neurologique centrale au cours du lupus érythémateux systémique Neuropsychiatric systemic lupus erythematosus (2nd part). Diagnos. La Rev m?®decine Interne. 2012; 33: 503–13.