Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Repair of tracheoesophageal fistula with pedicled sternocleidomastoid muscle flap after minimally invasive esophagectomy: A case report
Yixing Li1,2; Kun Fan1; Jizhao Wang1; Hongyi Wang1; Heng Zhao1,2; Bohao Liu1,2; Zhiyu Wang1,2; Yanpeng Zhang1,2; Junke Fu1; Jia Zhang1; Yonghao Du3; Weiwei Wu4*; Guangjian Zhang1*
1 Department of Thoracic Surgery, First Affiliated Hospital, Xi’an Jiaotong University, Xi’an 710061, China.
2 Health Science Center, Xi’an Jiaotong University, Xi’an710061, China.
3 Department of Imaging, The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, 710061, People’s Republic of China.
4 Interdisciplinary Research Center of Smart Sensors, Academy of Advanced Interdisciplinary Research, Xidian University, Xi’an, 710061, China.
*Corresponding Author: Zhang Guangjian1 & Weiwei Wu2
1Department of Thoracic Surgery, First Affiliated Hospital, Xi’an Jiaotong University, Xi’an 710061, China.
2Interdisciplinary Research Center of Smart Sensors,
Academy of Advanced Interdisciplinary Research,
Xidian University, Xi’an, 710061, China.
Email: michael8039@163.com
Received : July 13, 2021
Accepted : Aug 26, 2021
Published : Aug 31, 2021
Archived : www.jcimcr.org
Copyright : © Guangjian Z & Wu W (2021).
Abstract
Background: Tracheoesophageal Fistula (TEF) is a rare complication after Minimally Invasive Esophagectomy (MIE). Various surgical methods are available for repairing TEF. In this report, we have shown the importance and feasibility of pedicled Sternocleidomastoid Muscle (SCMM) flap in dealing with TEF.
Methods and results: A 57-year-old woman with esophageal squamous cell carcinoma underwent MIE in our hospital. TEF was diagnosed based on some clinical manifestations, like coughing after swallowing, a month after MIE. During the repair operation, we have utilized pedicled SCMM flap to cover the fistula after suturing the esophagus and trachea separately. No major complications occurred after the operation, and the prognosis was good.
Conclusions: Pedicled sternocleidomastoid muscle flap was convenient, reliable and efficient in covering the fistula, therefore, we recommend it as the routine surgical method. However, randomized controlled trials are further needed to confirm this recommendation. TEF can be reconstructed with a pedicled SCMM flap. This method can effectively avoid further complications.
Keywords: minimally invasive esophagectomy; esophageal cancer; tracheoesophageal fistula; sternocleidomastoid muscle.
Citation: Zhang G, Wu W, Li Y, Fan K, Wang J, et al. Repair of tracheoesophageal fistula with pedicled sternocleidomastoid muscle flap after minimally invasive esophagectomy: A case report. J Clin Images Med Case Rep. 2021; 2(4): 1288.
Introduction
In recent years, the concept of minimally invasive has been continuously developed. Minimally invasive esophagectomy has gradually become the first-line treatment option for resectable esophageal cancer, and more and more clinicians have begun to choose MIE. Compared with open surgery, MIE can effectively reduce the occurrence of postoperative complications and improve postoperative survival [1]. TEF is a rare complication after minimally invasive esophagectomy. The incidence of TEF during esophagectomy is less than 5%, but its mortality rate reaches 50% [2]. Surgical treatment is considered to be the best way to cure TEF. At present, there is no clear standard for the treatment of TEF after MIE. In this study, we used a pedicled sternocleidomastoid myocutaneous flap to repair TEF in patients receiving MIE.
Methods
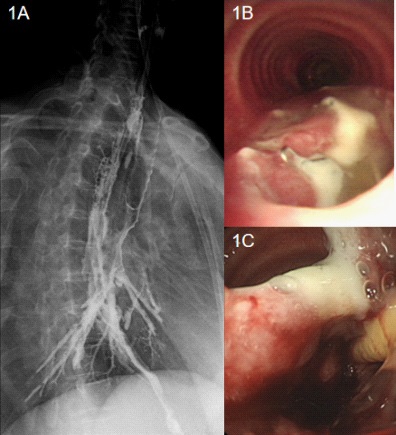
A 57-year-old woman with progressive dysphagia for more than one month was admitted to hospital and was final diagnosed with esophageal squamous cell carcinoma (T2N2M0). She underwent MIE by McKeown method. The tubular stomach was used for digestive tract reconstruction. Half a month later, the patient showed coughing after swallowing but there are no abnormal results upon physical examination. We carried out further examination and finally found she has TEF (Figure 1). After fully communicating with the patient, we decided to repair the TEF by operation.
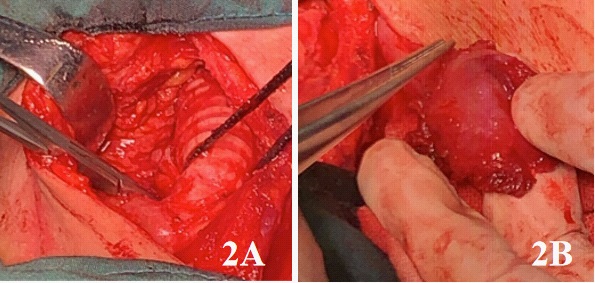
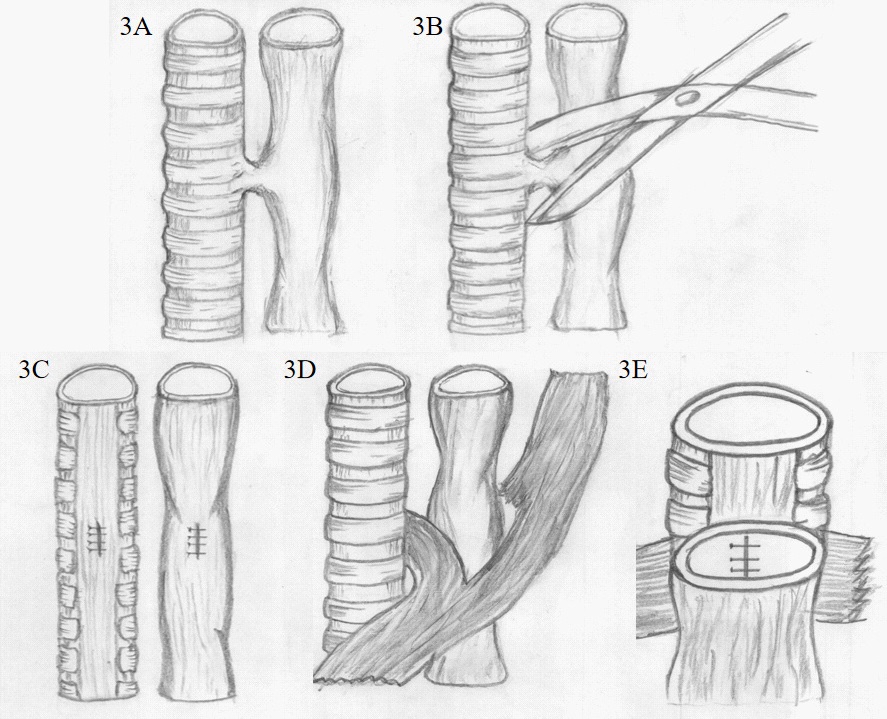
She was intubated by orotracheal intubation after general anesthesia. A 8 cm incision was made in the middle of the neck and then the subcutaneous tissue were dissociated. We dissected separately both sides of the trachea to expose the space between the trachea and the esophagus. The TEF was found at about 5cm under the glottis. At operation, the tracheal fistula was found about 1 cm X 0.8 cm, and the esophageal fistula was about 1 cm X 1 cm. The fistula was resected, and then, trachea and esophageal were repaired with absorbable sutures respectively by the way of double-layer suture. Subsequently, we dissected the right SCMM along the cervical fascia and separated part of the SCMM to form a pedicled muscle flap at the proximal end. The muscle flap was passed through the gap between the sternothyroid muscle and the scapulohyoid muscle, and finally entered the gap between the trachea and the esophagus. We compared the distance of the SCMM from the fistula during the operation, so that the length of the separated muscle flap could ensure that the muscle flap covers the fistula. The trachea and esophagus were isolated with pedicled SCMM flap to cover the surface of fistula, and the position of the muscle flap was fixed by sutures (Figure 2,3). After washing the surgical field, indwelling drainage tube and endotracheal intubation, we close the neck wound layer by layer. The operation time was about 320 minutes, the intraoperative blood loss was less than 10 ml. No transfusion was required and no intraoperative complications were encountered.
Results
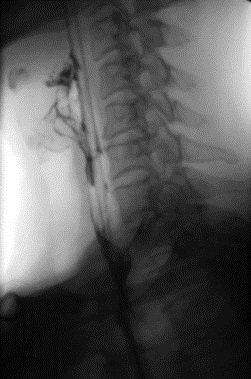
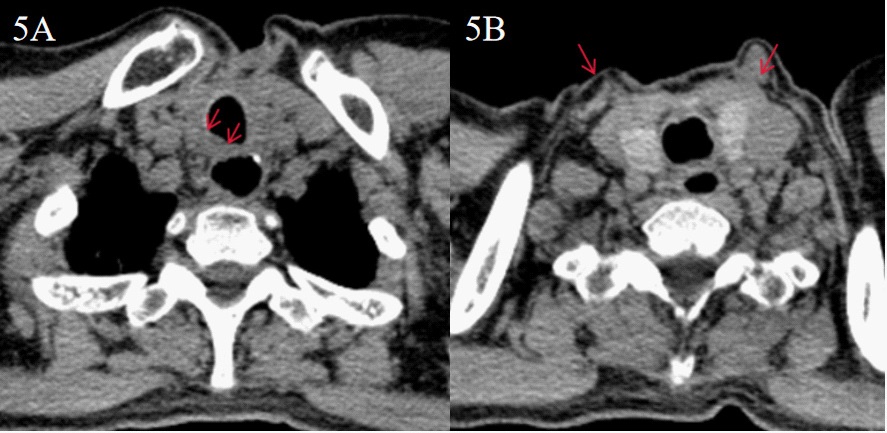
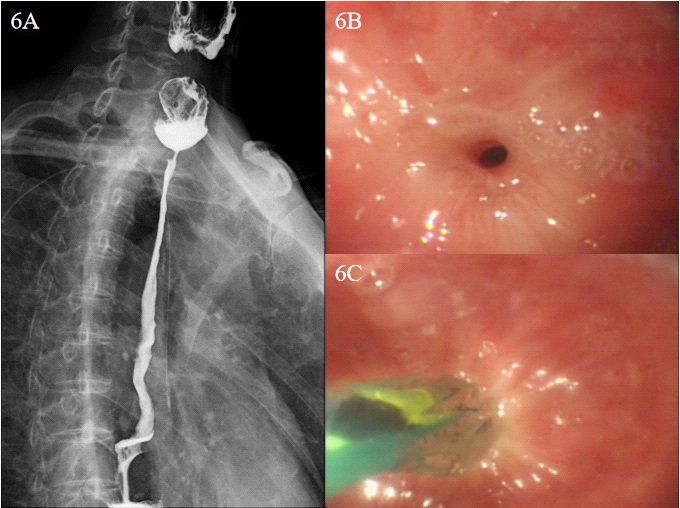
The vital signs of the patients were stable after operation, and the symptoms of coughing after swallowing disappeared. Barium esophagography showed no leakage of anastomosis (Figure 4). The patient was ventilated with ventilator after operation, and the tracheal intubation was removed 4 days later. The neck wound healed well. One month after the surgery, she was discharged. When the patient came to our hospital for review one month later, CT revealed that the right SCMM was partially absent, and the musculature was visible between the esophagus and trachea (Figure 5). Esophagoscopy revealed that the anastomotic stoma was narrow after operation so the endoscopic esophageal dilatation was performed (Figure 6). Subsequently, the anastomosis had an excellent and good recovery and there were no other complications.
Discussion
Esophagectomy is currently the first choice for radical treatment of resectable esophageal cancer [3]. Traditional open surgery has many postoperative complications and low patient survival rate [4,5]. In recent years, MIE has developed rapidly. MIE can effectively reduce postoperative complications and postoperative pain, and has a better postoperative survival rate [6,7]. However, there is still a risk of TEF after surgery. Most patients with TEF after MIE generally belong to non-malignant acquired TEF. The causes of adult non-malignant acquired TEF include injury after intubation, trauma, laryngectomy, esophagectomy and so on, of which esophagectomy accounts for 11% [8]. At present, we have not seen any systematic report or big data study on the complication of TEF after MIE. Anastomotic leakage is one of the main causes of TEF after operation of esophageal cancer [9]. In a large sample study of 2704 cases, it was found that the incidence of anastomotic leakage after esophagectomy was 11.4% [10]. In a study, there is a 14.8% chance of anastomotic leakage after MIE, and the occurrence and severity of anastomotic leakage are related to the prognosis of patients [11]. We can infer that TEF is not a common postoperative complication after MIE. Although reconstruction of the esophagus on the chest has a lower rate of anastomotic leakage than anastomosis on the neck [12]. But for upper esophageal cancer, the esophageal anastomosis after MIE is often lifted up to the neck. This may cause TEF more easily. However, the risk of TEF is very high, which seriously affects the prognosis of patients undergoing MIE and needs intervention as soon as possible.
There are many ways to treat TEF, including surgery and endoscopic treatment. Endoscopic treatment is to close the TEF by stents, clips, biological glue and so on. Some studies claim that endoscopy can treat TEF well. But we have not been able to achieve desired results in clinical practice. For achieving thorough treatment, we recommend to take the surgical approach. The success rate of surgery to fix TEF can reach more than 90% [8]. But differences in surgical methods lead to different possibilities for recurrence of TEF after surgery. In order to promote the healing of wound after repair, tissue such as pedicled muscle flap and fascia is often used to separate trachea and esophagus. At present, there have been clinical reports of pedicled forearm fascia, latissimus dorsi, serratus anterior, intercostal muscle, pectoralis major muscle and so on to treat TEF [13-15].
The different position of the TEF causes the different methods to select muscle flaps. Intercostal muscle, serratus anterior, and pectoralis major muscle are often used for the repair of chest TEF. The hyoid muscle, sternocleidomastoid muscle and latissimus dorsi can be used for TEF in the neck. Compared with the SCMM, the hyoid muscles are closer to the gap of the trachea and esophagus anatomically, but they are more slender and have fewer muscle flaps available. Excessive anatomy will affect their original physiological functions. The latissimus dorsi can offer more materials, but it is farther away from the TEF, so the blood supply of the muscle flap may be restricted. Compared with the blood supply of the pedicled muscle flap, the free and the transplanted muscle flaps are completely destroyed, which may not achieve the purpose of promoting the healing of the fistula. In addition, the surgical procedure is more difficult for transplanted skin flaps from other parts, and some enlarged incisions may increase postoperative pain. Chuang FC et al [16]. reported that the SCMM rotation flap was used to repair acquired TEF in the neck. They believed that the SCMM rotation flap repairing 1~2 cm TEF has a better effect to close the fistula. Liu [17] reported a case of repairing a large TEF with pedicled myocutaneous flap of SCMM. They used the skin flap to repair the trachea membrane and covered the huge fistula with the muscle flap to promote wound healing. The results showed that the wound healed well after operation. There is also a consistent result in our research.
The patient we reported has just undergone MIE and the esophagus has been reconstructed, so it is necessary to guard against recurrence after TEF resection. At the same time, this TEF is not too large, so there is no need for excessive local resection of the trachea or esophagus, nor does it need to be sutured with a skin flap. Therefore, we consider using only SCMM flap for repair. After simply closing the fistula of trachea and esophagus, the SCMM flap with abundant blood supply was used to cover the surface of the fistula. The muscle flap can prevent the formation of a tracheoesophageal fistula once the anastomosis leaks, and at the same time, the abundant blood supply helps healing. The results showed that the patient recovered well and there was no recurrence of TEF. Follow up the results of postoperative reexamination, esophagoscopy showed that the fistula healed well, but there was anastomotic stenosis. This may be caused by the contraction of scar tissue after operation. After endoscopic esophageal dilatation, we effectively solved the problem of stricture.
Conclusion
TEF may occur after MIE. For the surgical treatment of this kind of TEF, the pedicled SCMM flap is a convenient and effective repair material. Using pedicled SCMM flap to separate tracheoesophageal space can contribute to the reconstruction of trachea and esophagus, help fistula repair as soon as possible, and reduce the possibility of postoperative recurrence. Our study is only one case, but whether it is applicable to more patients with tracheoesophageal fistula needs further research.
Disclosure: The authors declare that there are no conflicts of interest.
References
- Sakamoto T, Fujiogi M, Matsui H, Fushimi K, Yasunaga H. Comparing Perioperative Mortality and Morbidity of Minimally Invasive Esophagectomy Versus Open Esophagectomy for Esophageal Cancer: A Nationwide Retrospective Analysis. Annals of surgery. 2019.
- Palmes D, Kebschull L, Bahde R, Senninger N, Pascher A, et al. Management of Nonmalignant Tracheo- and Bronchoesophageal Fistula after Esophagectomy. Thorac Cardiovasc Surg. 2020.
- Schweigert M, Dubecz A, Stein HJ, Oesophageal cancer-an overview. Nature Reviews Gastroenterology & Hepatology. 2013: 10: 230-244.
- Shanmugasundaram R, Hopkins R, Neeman T, Beenen E, Fergusson J, et al. Minimally invasive McKeown‘s vs open oesophagectomy for cancer: A meta-analysis. Ejso. 2019; 45: 941-949.
- Sihvo E, Helminen O, Gunn, J, Sipila JOT, Rautava P, et al. Longterm outcomes following minimally invasive and open esophagectomy in Finland: A population-based study. Ejso. 2019; 45: 1099-1104.
- Sakamoto T, Fujiogi M, Matsui H, Fushimi K, Yasunaga H. Comparing Perioperative Mortality and Morbidity of Minimally Invasive Esophagectomy Versus Open Esophagectomy for Esophageal Cancer: A Nationwide Retrospective Analysis. Ann Surg. 2019.
- Gottlieb-Vedi E, Kauppila JH, Malietzis G, Nilsson M, Markar SR, et al. Long-term Survival in Esophageal Cancer After Minimally Invasive Compared to Open Esophagectomy: A Systematic Review and Meta-analysis. Ann Surg. 2019; 270: 1005-1017.
- Muniappan A, Wain JC, Wright CD, Donahue DM, Gaissert H, et al. Surgical treatment of nonmalignant tracheoesophageal fistula: a thirty-five year experience. Ann Thorac Surg. 2013; 95: 1141-6.
- Bartels HE, Stein HJ, Siewert JR, Tracheobronchial lesions following oesophagectomy: prevalence, predisposing factors and outcome. British Journal of Surgery. 1998; 85: 403-406.
- Low DE, Kuppusamy MK, Alderson D, Cecconello I, Chang AC, et al. Benchmarking Complications Associated With Esophagectomy. Annals of Surgery. 2019; 269: 291-298.
- Fransen LFC, Berkelmans GHK, Asti E, van Berge Henegouwen MI, Berlth Fet al. The Effect of Postoperative Complications After Minimally Invasive Esophagectomy on Long-term Survival: An International Multicenter Cohort Study. Ann Surg. 2020.
- Schroder W, Raptis DA, Schmidt HM, Gisbertz SS, Moons J, et al. Anastomotic Techniques and Associated Morbidity in Total Minimally Invasive Transthoracic Esophagectomy: Results From the EsoBenchmark Database. Ann Surg. 2019; 270: 820-826.
- Teixeira S, Costa J, Monteiro D, Bartosch I, Insua-Pereira I, et al. Pharyngocutaneous and tracheoesophageal fistula closure using supraclavicular artery island flap. Eur Arch Otorhinolaryngol 2018; 275: 1921-1926.
- Azuma S, Sakuraba M, Azumi S, Fujita T, Daiko H. Repairing Bronchoesophageal Tube Fistula Using A Contralateral Latissimus Dorsi Musculocutaneous Flap. Plastic and reconstructive surgery. Global open. 2017; 5: e1484-e1484.
- Siu KF, Wei WI, Lam KH, Wong J. Use of the pectoralis major muscle flap for repair of a tracheoesophageal fistula. American Journal of Surgery. 1985; 150: 617-619.
- Chuang FC, Tung KY, Huang WC, Yu CM, Yao WT. Using a Sternocleidomastoid Muscle Flap to Close an Iatrogenic Tracheoesophageal Fistula in a Patient With Advanced Laryngeal Cancer: A Case Report and Literature Review. Ann Plast Surg. 2019; 82: S126-S129.
- Liu, X, Li, Q, Huang W, Liu M, Xu X, Jiang G. Use of a Pedicled Sternocleidomastoid Musculocutaneous Flap in a Large Tracheoesophageal Fistula. Ann Thorac Surg. 2020; 110: e233- e236.