Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
A case of severe multi-system disease in a sickle cell SC patient caused by parvovirus B19 infection
Mohamed Almuqamam1*; Swetha Madhavarapu1; Nataly Apollonsky2
1 Section of Pediatric Critical Care Medicine, St. Christopher’s Hospital for Children, 160 E Erie Ave Philadelphia PA 19134, USA
2 Section of Pediatric Hematology, St. Christopher’s Hospital for Children, 160 E Erie Ave Philadelphia PA 19134, USA.
*Corresponding Author: Mohamed Almuqamam
Section of Pediatric Critical Care Medicine,
St. Christopher’s Hospital for Children, 160 E Erie
Ave Philadelphia PA 19134, USA.
Email: Mohamed.almuqamam@towerhealth.org
Received : Aug 02, 2021
Accepted : Sep 10, 2021
Published : Sep 17, 2021
Archived : www.jcimcr.org
Copyright : © Almuqamam M (2021).
Abstract
Sickle Cell Disease (SCD) is an inherited hemoglobinopathy, which results in production of abnormal hemoglobin S. HbSC disease is a variant of SCD, which shares a similar clinical complication profile to HbSS disease, but often thought to be a milder condition. In patients with SCD, Hb S in deoxygenated state undergoes polymerization, leading to hemolysis, vaso-occlusive events, and eventually end-organ damage. Among other complications in patients with SCD is increased risk of complications caused by parvovirus B19.
We present a case of a 14-year-old female with HbSC disease who presented to the emergency room with complaint of abdominal pain and found to have splenic sequestration. Splenic sequestration progressed rapidly, Hemoglobin (hb) dropped to 4.6 g/dl and acute chest syndrome (ACS) developed. She was treated following the ACS protocol, received 4 units of Packed Red Blood Cells (PRBC) and subsequently underwent a single volume PRBC exchange transfusion. Considering her unusual presentation, with severe ARDS from alveolar hemorrhage requiring mechanical ventilation and multi-organ injury, several autoimmune and infectious conditions with a cytokine storm component including COVID-19 disease, were considered. Results of viral testing revealed parvovirus B19 IgM antibodies signifying an acute infection. She fully recovered with supportive care and was discharged home.
Multisystem involvement simulating connective tissue disorders or malignancies with acute parvovirus B19 infection has been reported and is considered extremely rare. To our knowledge, there were no reports of pediatric patients with SC disease presenting with splenic sequestration and ACS in the setting of parvovirus B19 multisystem disease.
Keywords: sickle cell disease; acute respiratory distress syndrome; acute chest syndrome; parvovirus B19.
Citation: Almuqamam M, Madhavarapu S, Apollonsky N. A case of severe multi-system disease in a sickle cell SC patient caused by Parvovirus B19 infection. J Clin Images Med Case Rep. 2021; 2(5): 1314.
Introduction
Sickle Cell Disease (SCD) is an autosomal recessive genetic disorder characterized by a mutation in the beta globin gene resulting in the production of sickle hemoglobin (HbS). It is the most prevalent disorder identified by neonatal blood screening. HbSC disease is the second most frequent hemoglobinopathy after homozygous SS disease. There are an estimated 54,736 babies born with HbSC disease each year worldwide [1]. HbSC disease is generally considered to be a variant of SCD, and despite sharing similar clinical complications to SS disease, it is thought to be a milder disease.
SCD is characterized by hemolytic anemia, vaso-occlusive episodes and acute and chronic organ damage leading to shortened life span. Acute Chest Syndrome (ACS) is a serious complication of sickle cell disease and is responsible for a significant morbidity and mortality [4]. It can be defined as the presence of fever and/or new respiratory symptoms accompanied by a new pulmonary infiltrate on chest X-ray. ACS is the most common cause of death and the second most common cause of hospitalization in patients with SCD [2].
Infection with human parvovirus B19 in the pediatric population has a wide range of presentations. It classically causes a self-limited febrile illness that is characterized by a rash called erythema infectiosum or fifth disease. The range of clinical manifestations is wide and includes arthritis, arthralgia, multisystem vasculitic disease, hyperimmune syndrome and transient aplastic crisis in patients with chronic hemolytic anemia like SCD and hydrops fetalis in neonates [3].
Case presentation
A 14-year-old female with sickle cell SC disease presented to the emergency room with three days of chest and abdominal pain along with multiple episodes of emesis and decreased oral intake. In the ED, she had normal vital signs and her exam was notable for a reproducible chest pain and generalized abdominal pain with moderate guarding. Initial laboratory work up revealed mildly decreased Hb of 9.8 g/dl and platelet count of 141,000 cells/mcL comparing to her baseline and an abdominal x-ray showed moderate fecal burden so she was admitted for management of vaso-occlusive crisis and constipation.
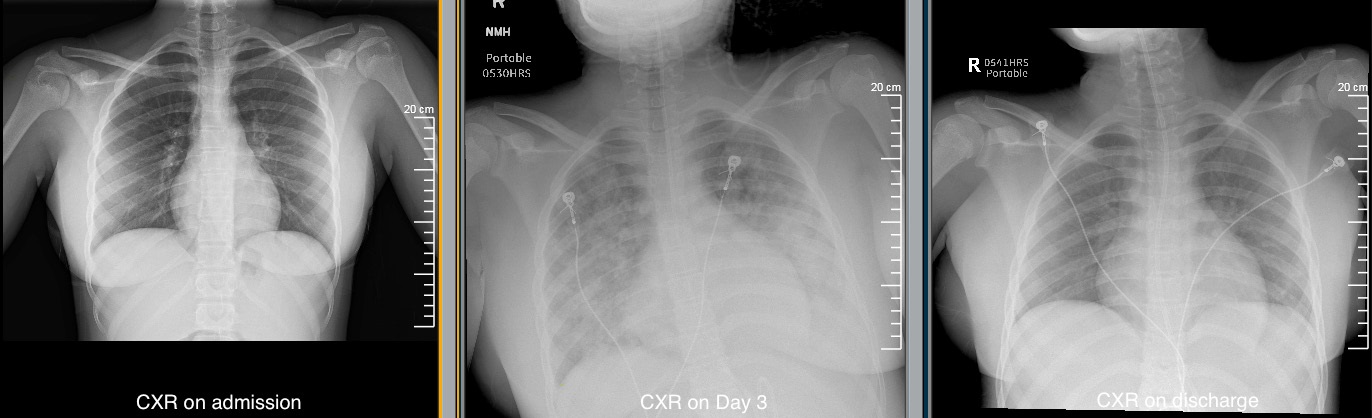
Over the course of 8 hours, she reported an increase of her abdominal pain with worsening tenderness and guarding which prompted evaluation with an abdominal CT scan due to concerns for an evolving acute abdominal process. The abdominal CT showed splenomegaly. The following morning, she developed a fever and was noted to be tachycardic, tachypneic and hypoxic with oxygen saturation in lower 80s requiring supplemental oxygen. Repeat Hb revealed a drop to 4.3 g/dl and the chest x-ray showed a new left lower lobe infiltrate (Figure 1). In lieu of the clinical decline, she was transferred to the PICU for further management.
Patient was treated following ACS/splenic sequestration protocol with broad spectrum antibiotics, fluid restriction, PRBC transfusions, single volume exchange transfusion and respiratory support, initiated with CPAP and escalating to BiPAP with increasing Fio2 requirement up to 0.8. Repeat chest imaging at 72-hour mark of hospitalization showed a new involvement of the right lung (Figure 1). CT angiogram of the chest was performed to further evaluate the parenchymal lung disease and to exclude Pulmonary Embolism (PE) given the elevated d-dimers. The CT-PA showed multiple patchy and centrally alveolar opacities and no PE. Due to worsening respiratory status and wide A-a gradient, she was intubated and placed on mechanical ventilation. Soon after intubation, she was noted to have frank blood and bloody secretions from the endotracheal tube along with brown to red colored urine. Her urinalysis showed proteinuria in the nephrotic rage with protein creatinine ratio > 3.0 along with hematuria. She also developed hypertension requiring a Nicardipine drip. Due to the multisystem involvement and elevated inflammatory markers, an extensive rheumatological panel was sent. In light of the persistent fevers while being on a broad-spectrum antimicrobial coverage with Ceftriaxone, Azithromycin and Vancomycin, extensive infectious work up was sought.
The patient remained intubated for 7 days and despite a progressively improving lung compliance, disappearing bloody endotracheal secretions and decreasing proteinuria she continued to be febrile. Rheumatological work up showed a negative SLE panel with normal serum complement levels. Vasculitic antibodies were weakly positive with Anti proteinase 3 Ab at 6.9 units/ml and Anti myeloperoxidase at 10.4 units/ml. Anticardiolipin Ab IgG was low-medium positive at 32 U/mL, anticardiolipin IgM was low-medium positive at 41 U/ml and anticardiolipin IgA was negative. Overall, she finished a 5-day course of Azithromycin for the suspected ACS and remained on Ceftriaxone for 14 days. Blood and urine cultures remained negative. Multiple nasopharyngeal swabs and deep tracheal aspirates were negative for COVID-19 and the rest of the respiratory viral panel including, EBV and CMV panels were negative. Eventually, Parvovirus IgM/IgG antibodies were positive at 17.1 and 5.7 respectively and serum Parvovirus PCR was positive. She was extubated on day 7 uneventfully and discharged on Day 18. By discharge, proteinuria, hematuria and hypertension had resolved. Considering severity of splenic sequestration and persistent splenomegaly, the plan is to consider splenectomy in the future.
Discussion
Although hemoglobin SC disease is a common hemoglobinopathy, surprisingly few studies have been dedicated to this disease. Most of the available literature on the complications and outcomes of hemoglobin SC disease is derived from longitudinal observational studies which are largely confined to centers outside North America. Naessens et al found that most common complication encountered in this patient population was retinopathy at a rate of 55.8% [4]. Rates for acute painful vaso-occlusive crises ranged between 23.1%-36% [5]. Acute chest syndrome does not seem to be a common finding in these patients with a rate of 7.7% [4].
For patients with chronic hemolytic anemia like SCD, encountering parvovirus B19 can have a myriad of serious complications. Majority of parvovirus B19 infections compromise the bone marrow and end up causing a severe but transient aplastic crisis. There are multiple reports of severe complications including acute encephalopathy, nephrotic syndrome, splenic sequestration and fatal bone marrow embolism [6-9]. It is worth noting that all of those complications were reported with SS or SE disease. ACS on the other hand, has been reported as a complication of parvovirus B19 infection in pediatric patients with SC disease like our patient. In fact, Coates et al found a clear causal relation between parvovirus B19 seroconversions and acute chest syndrome with an odd ratio of 6.4 [10].
Our patient developed ACS and splenic sequestration in the setting of acute parvovirus B19 infection, however she also had multi-organ vasculitic involvement with alveolar hemorrhage and glomerulonephritis resulting in hematuria and nephrotic range proteinuria. Moreover, multisystem involvement simulating a connective tissue disorder or malignancy with acute parvovirus B19 infection has been reported and is considered extremely rare. Parvovirus infection can be associated with a variety of renal manifestations like collapsing glomerulonephritis, endocapillary glomerulonephritis, mesangiolysis, focal segmental glomerular sclerosis, and in some cases transient urinary abnormalities [3,11-15]. It should be noted that majority of these patients had normal renal function and spontaneously recovered. Our patient also had normal renal function panels throughout her hospitalization but was transiently hypertensive requiring an intravenous antihypertensive drip.
Lung disease with parvovirus B19 is not as common of an association as renal disease and has not been previously described in conjunction with renal disease in an SC disease patient. Bousvaros et al reported a case of an 8-year-old girl with parvovirusassociated interstitial lung disease, hepatitis, and myositis [16]. Wardeh and Marik described a 49-year-old woman with acute lung injury secondary to parvovirus infection that progressed to respiratory failure and recovered with supportive therapy only [17]. Morris and Smilack reported a healthy patient with respiratory failure and bilateral pulmonary infiltrates, new onset anemia and multiple joint effusions who also improved with supportive care [18]. The exact mechanism of parvovirus B19 associated renal and pulmonary disease is not fully known. It is postulated that the disease can be due to direct invasion by the virus or the consequence of the host’s immune response.
To our knowledge, there were no reports of pediatric patients with SC disease presenting with severe ACS with alveolar hemorrhage, hypertension, proteinuria, hematuria, fevers and elevated inflammatory markers. Although some case reports showed improvement with the use of steroids in selected patients, our patient recovered spontaneously with supportive care only [19].
Conclusion
Parvovirus B19 infection presenting with concomitant ACS, splenic sequestration and multisystem disease is an uncommon but potentially serious complication in children with SCD. Healthcare providers especially intensivists should be aware of the wide spectrum of manifestations of Parvovirus B19 infection in patients with SCD to ensure effective management. This case shows that Parvovirus B19 infection maybe associated with more than a self-limited illness in SCD patients. Therefore, it is of paramount importance to develop preventive strategies including vaccination against Parvovirus B19 virus in order to mitigate its serious sequelae.
Acknowledgements: The authors would like to thank the parent for providing consent to publish the case and enclosed studies. The authors would also like to thank Dr. Svetlana Lvovich, Pediatric Rheumatologist, who provided valuable input and helped manage the patient.
Disclosure: The authors have no funding or conflict of interest to disclose.
All authors wrote and edited the manuscript.
All authors reviewed and approved final draft of the manuscript.
The case laboratory and radiological data is available and can be released upon request. The patient’s guardian gave consent for de-identified data to be published and shared publicly.
References
- Weatherall DJ. The inherited diseases of hemoglobin are an emerging global health burden. Blood. 2010; 115: 4331–6
- Farooq S, Abu Omar M, Salzman GA. Acute chest syndrome in sickle cell disease. Hosp Pract. 2018; 46:144-51
- Mori Y, Yamashita H, Umeda Y, et al. Association of parvovirus B19 infection with acute glomerulonephritis in healthy adults: case report and review of literature. Clin Nephrol. 2002; 57: 69- 73.
- Lionnet F, Hammoudi N, Stojanovic KS, et al. Hemoglobin sickle cell disease complications: a clinical study of 179 cases. Haematologica. 2012; 97: 1136-41
- Naessens V, Ward R, Kuo K. A North American Experience Of Hemoglobin SC Disease, Its Complications, and Management. Blood. 2013; 122: 2221.
- Bakhshi S, Sarnaik S, Becker C, et al. Acute encephalopathy with parvovirus B19 infection in sickle cell disease. Arch Dis Child. 2012; 87: 541–42.
- Quek L, Sharpe C, Dutt N, et al. Acute human parvovirus B19 infection and nephrotic syndrome in patients with sickle.cell disease. Brit J Haematol. 2010; 149: 289–91
- Yates A, Hankins J, Mortier N, Aygun B, Ware R. Simultaneous acute splenic sequestration and transient aplastic crisis in children with sickle cell disease. Pediatr Blood Cancer. 2009; 53: 479–81
- Rayburg M, Kalinyak K, Towbin A, et al. Fatal bone marrow embolism in child with hemoglobin SE disease. Am J Hematol. 2010; 85: 182–84.
- Coates TD, Dampier C, Kalinyak K, et al. Acute Chest Syndrome Is Strongly Associated Parvo Virus B19 Seroconversion in Patients with Hemoglobin SC Disease. Blood. 2004; 104: 1664.
- Takeda S, Takaeda C, Takazakura E, et al. Renal involvement induced by human parvovirus B19 infection. Nephron. 2001; 89: 280-85.
- Moudgil A, Nast CC, Bagga A, at al. Association of parvovirus B19 infection with idiopathic collapsing glomerulopathy. Kidney Int. 2001; 59: 2126-33.
- Komatsuda A, Ohtani H, Nimura T, at al. Endocapillary glomerulonephritis in a patient with parvovirus B19 infection. Am J Kidney Dis. 2000; 36: 851-54
- Nakazawa T, Tomosugi N, Asaka M, et al. Acute glomerulonephritis after human parvovirus B19 infection. Am J Kidney Dis. 2000; 35: E31.
- Tanawattanacharoen S, Falk RJ, Jennette JC, et al. Parvovirus B19 DNA in kidney tissue of patients with focal segmental glomerulosclerosis. Am J Kidney Dis. 2000; 35: 1166-74
- Bousvaros A, Sundel R, Thorne GM, et al. Parvovirus B19 associated interstitial lung disease, hepatitis and myositis. Pediatr Pulmonol. 1998; 26: 365-69
- Wardeh A, Marik P. Acute lung injury due to parvovirus pneumonia. J Intern Med. 1998; 244: 257-60.
- Morris CN, Smilack JD. Parvovirus B19 infection associated with respiratory distress. Clin Infect Dis. 1998; 27: 900.01
- Dass R, Ramesh P, Ratho RK, et al. Parvovirus B19.induced multisystem disease simulating systemic vasculitis in a young child. Rheumatol Int. 2005; 25: 125–29.