Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Therapeutic potential of ibuprofen inhalation for COVID-19 pneumonia: Report of two first cases
Germán Ambasch1; Esteban Coscia1; Jorge Luis Tambini Díaz1; Germán David Bueno1; Luis Alberto Argañarás2; Nicolás Martínez Ríos2; Daniela Josefina Porta5; Dante Miguel Beltramo3,4; Néstor Horacio García5*
1 Sanatorio Mayo Privado SA, Ciudad de Córdoba, Córdoba, Argentina.
2 Química Luar SRL, Ciudad de Córdoba, Córdoba, Argentina.
3 CEPROCOR, Santa María de Punilla, Córdoba, Argentina
4 CONICET, Santa María de Punilla, Córdoba, Argentina
5 Instituto de Investigaciones en Ciencias de la Salud, Ciudad Universitaria, Córdoba, Argentina
*Corresponding Author: Néstor H García
Institute for Research in Health Sciences, Ciudad
Universitaria, Boulevard de la Reforma, Cell Biology
Pavilion, University City, X5000, Córdoba, Argentina.
Email: garcia.nestor@conicet.gov.ar
Received : Aug 20, 2021
Accepted : Sep 24, 2021
Published : Oct 01, 2021
Archived : www.jcimcr.org
Copyright : © García NH (2021).
Abstract
No specific and effective antiviral treatment has been approved for COVID-19 so far. Systemic corticosteroid and remdesivir have shown to decrease mortality in COVID-19 patients, but mortality still is elevated. We propose that nebulized hypertonic ibuprofen solution which has bactericidal, virucidal, mucolytic and anti-inflammatory properties to be used for COVID-19 pneumonia and prevent the classical evolution to respiratory failure and mechanical respiration. We report here the first two cases of the COVID-19 pneumonia successfully treated with nebulized hypertonic ibuprofen solution (NaIHS). Rationale of the treatment is to mitigate the local inflammation with inhaled NIH that stays in the lung and may inhibit proliferation of the virus, inflammation and successfully reverts the hypoxia observed in these clinical cases. Mild adverse events were observed. Larger and further studies are warranted to confirm the result of these cases.
Keywords: hypertonic ibuprofen solution; inhalation; coronavirus disease 2019.
Abbreviations: COVID-19; coronavirus disease 2019; SARS-CoV-2; severe acute respiratory syndrome coronavirus.
Citation: Garcia NH, Ambasch G, Coscia E, Díaz JLT, Bueno GD, et al. Therapeutic potential of ibuprofen inhalation for COVID-19 pneumonia: Report of two first cases. J Clin Images Med Case Rep. 2021; 2(5): 1336.
Introduction
Disease due to severe acute respiratory syndrome coronavirus 2 (SARS CoV-2), COVID-19, has taken the world by storm. More than 201 million cases and greater than 4 million deaths have been reported by the World Health Organization [1] and effective therapy options remain limited. To date, dexamethasone, remdesivir, tofacitinib, and monoclonal antibody therapies have demonstrated benefit in patients with severe illness [2], while evidence of benefit for many other drugs remains unknown [3] or disproven [4-7].
A classical presentation of severe COVID-19 is profound hypoxia, progressing to Acute Respiratory Distress Syndrome (ARDS). Pulmonary release of autacoids leading to vascular injury and permeability results in excess alveolar fluid protein accumulation. This phenomenon, coupled with vasodilation and intrapulmonary shunting of non-oxygenated blood has been characterized as a ‘bradykinin storm’ [8]. This condition contributes to ventilation/perfusion mismatch and the consequence of hypoxia. Importantly, ibuprofen can prevent inflammation induced by bradykinin at the respiratory mucosa [9] and it has also been demonstrated to prevent bradykinin induced vascular extravasation and angioedema in humans [10]. These data suggest that ibuprofen may mitigate this aspect of the inflammatory response in COVID-19.
Previously we hypothesized that nebulized hypertonic ibuprofen has antibiotic, viricidal and anti-inflammatory effects that are expected to prevent viral replication in the respiratory tract, minimize bacterial superinfections and reduce inflammatory cytokines which appear to be the major problems in COVID-19 [11]. Ibuprofen with the highest FDA performance side effect profile had showed to be safe given orally or ev, and can be given at any age. Based on that ethical approval was obtained from the Institutional Independent Ethics Committees and district regulatory agencies of Córdoba Province on April 4th, 2020, for the compassionate use of Luarprofeno® (sodium ibuprofenate in hypertonic saline for nebulization) (hereafter, NaIHS).
The 2 patients were hospitalized by April, 2020 had physical symptoms and radiological findings of pneumonia. The possible efficacy of NaIHS (trade name: Luarptofen®) was evaluated. These 2 patients with poor oxygenation and radiologic signs of COVID-19 pneumoniae revert rapidly. In these cases, clinical courses revealed favorable.
Case reports
Case 1: Female, 77 years old
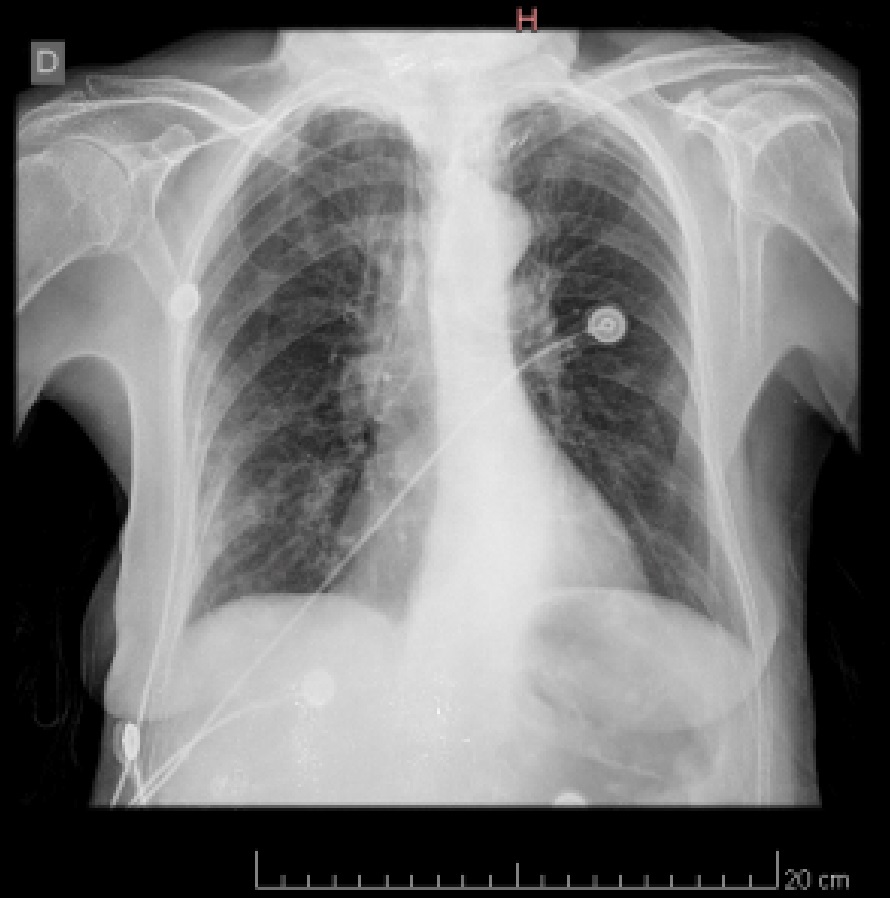
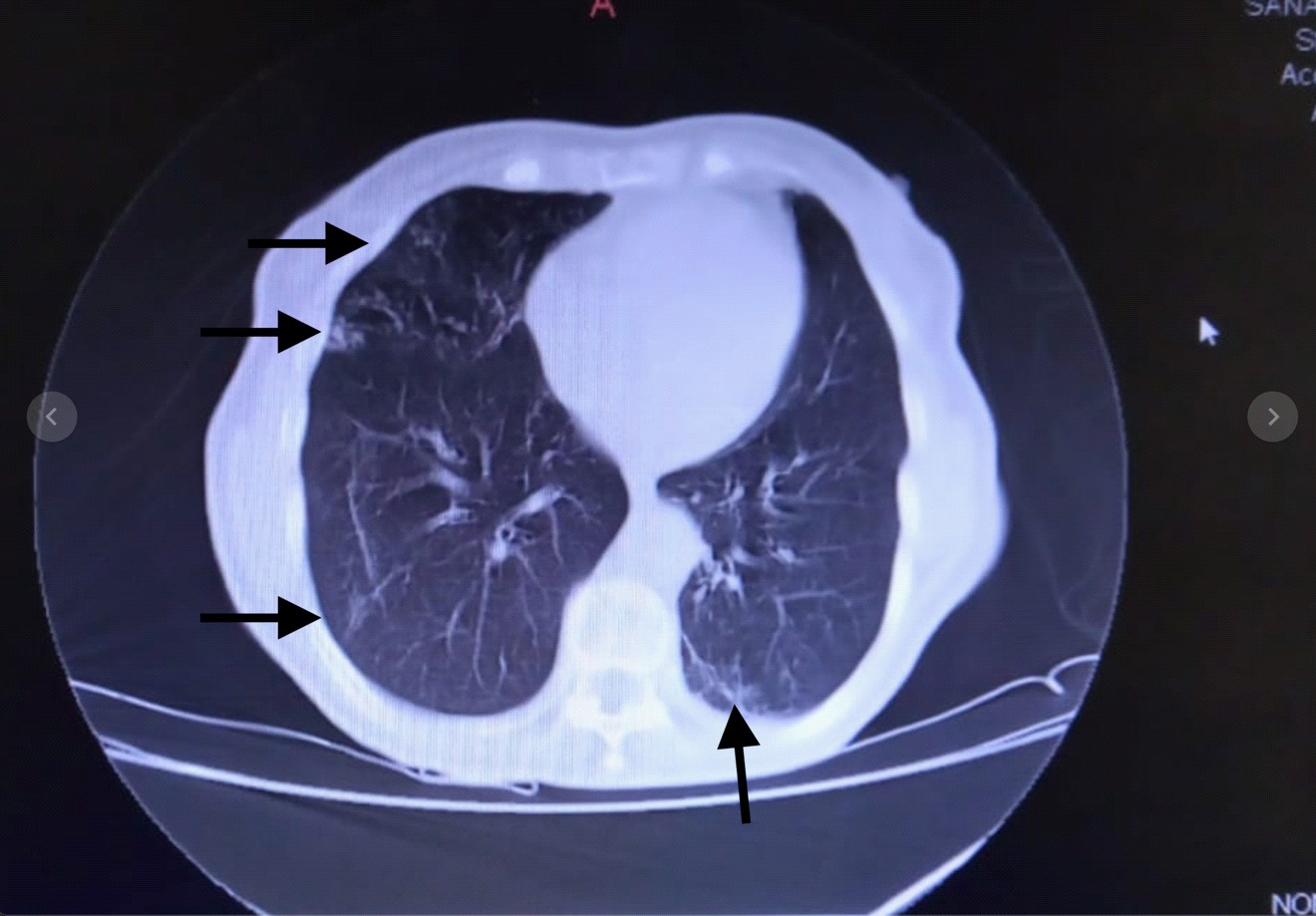
She was admitted on April 20, 2020 due to sore throat, malaise, loss of appetite, and dyspnea without fever derived from a home care center from Villa Saldán, Córdoba suspecting COVID-19 pneumoniae, the patient had a positive throat swab PCR test on April 18, 2020 for SARS-CoV-2. Past history: Hypertension, Dyslipidemia. Physical findings: No pharyngeal redness, no lymphadenopathy, rhonchus, wheezes and fine crackles could be heard in both lung fields at the end of inspiration. Rest of the examination was normal. Blood pressure 140/60 mmHg, pulse rate 85/min, respiratory rate 23/min, body temperature 36.5 °C, SpO2 86%/RA. EKG with left atrial overload. Laboratory findings were listed in Table 1. Chest X-ray on admission revealed mild infiltration shadow in lower right lung field (Figure 1) and chest CT on the same day demonstrated ground-glass opacities (GGO) along the pleura over both middle and lower lung fields (Figure 2). Based on these findings, oxygen supplementation (4 L/min), enoxaparin 40 mg sc, bid, ceftriaxone 1gr ev bid, clarithromycin 500 mg ev bid and respiratory kinesiotherapy were started.
Table 1: Results of laboratory tests conducted on the day of admission (Case 1).
Hematology |
Biochemistry |
||
WBC (μL) |
4260 |
Glucose (mg/dl) |
107 |
Neutrophils |
65% |
Urea (mg/dl) |
46 |
Lymphocytes |
30% |
Creatinine (mg/dl) |
0.84 |
Monocytes |
4% |
Total Bilirubin (mg/dl) |
4.5 |
Eosinophils |
1% |
AST (U/L) |
38 |
Basophils |
0% |
ALT (U/L) |
30 |
Red Blood Cells (106/ μL) |
426 |
LDH (U/L) |
200 |
Hemoglobin (g/dl) |
11.4 |
ALP (U/P) |
450 |
Hematocrit (%) |
35.6 |
Sodium (mEq/L) |
134 |
Platelets (103/ μL) |
110 |
Potassium (mEq/L) |
3.2 |
D-dimer (ng/ml) |
1464 |
Chloride (mEq/l) |
102 |
Ferritin (mcg/l) |
792 |
||
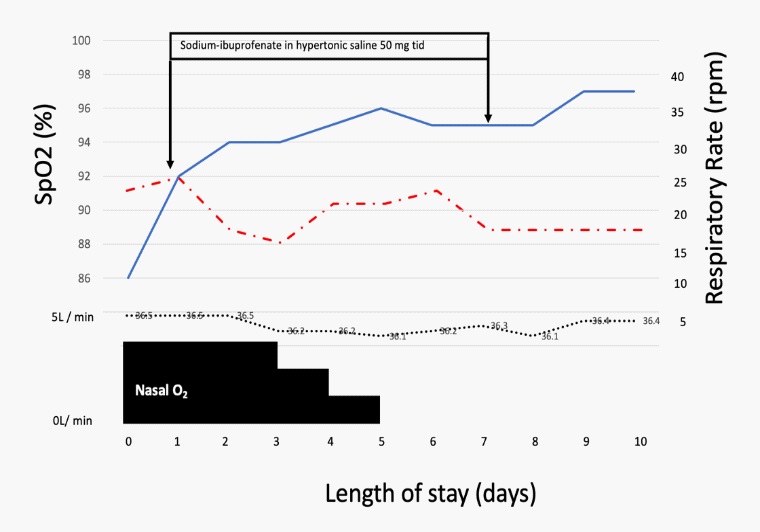
The clinical course is illustrated in Figure 3. On admission, she started with oxygen 4L/min nasal but SpO2 increase slightly and it did not reach 92%. She felt fatigue, lying down almost all the time, and could hardly take any food. There was poor communication and disorientation. After 24 hours of admission, oxygen supplementation at 4 L/min was required but SpO2 was not >95%, and 50 mg NaIHS was started each 8 hours. Oxygenation gradually improved, and after the 24 hours, SpO2 was 94%. Each dose was administered using a high flow oxygen by face-mask. To reduce health care worker infection risk in the setting of nebulized therapy use, a novel containment hood was utilized with air venting via antiviral filters, designed to prevent particle aerosolization. All health care workers utilized Personal Protective Equipment (PPE) provided. Each nebulized dose was administered over approximately 15 minutes, the time required to drain the liquid nebulization vessel. Fever was never present. After the start NaIHS oxygenation improved, respiratory rate decreased progressively every day.
Within 24-48 hours after starting NaIHS, there was no body temperature above 36.5°C, oxygenation had improved. SpO2 was maintained at >94% with less O2 requirements, and after the 4th day of NaIHS SpO2 was maintained at >94% with RA. Hypoxemia during bodily movement also improved. NaIHS was very good tolerated, but it induced during the first nebulization, mild cough, but after the first 5 minutes, it was minimal. Her appetite remarkably recovered, her fatigue improved, and she became able to walk indoors without any aid. The patient was judged to have improvement from COVID-19. By the 10th day, the patient was ready to be discharge home care. Nasal swab PCR tests was negative for SARS-CoV-2 but she was discharged on May 20, 2020 due to social difficulties.
Case 2: Male, 59 years old
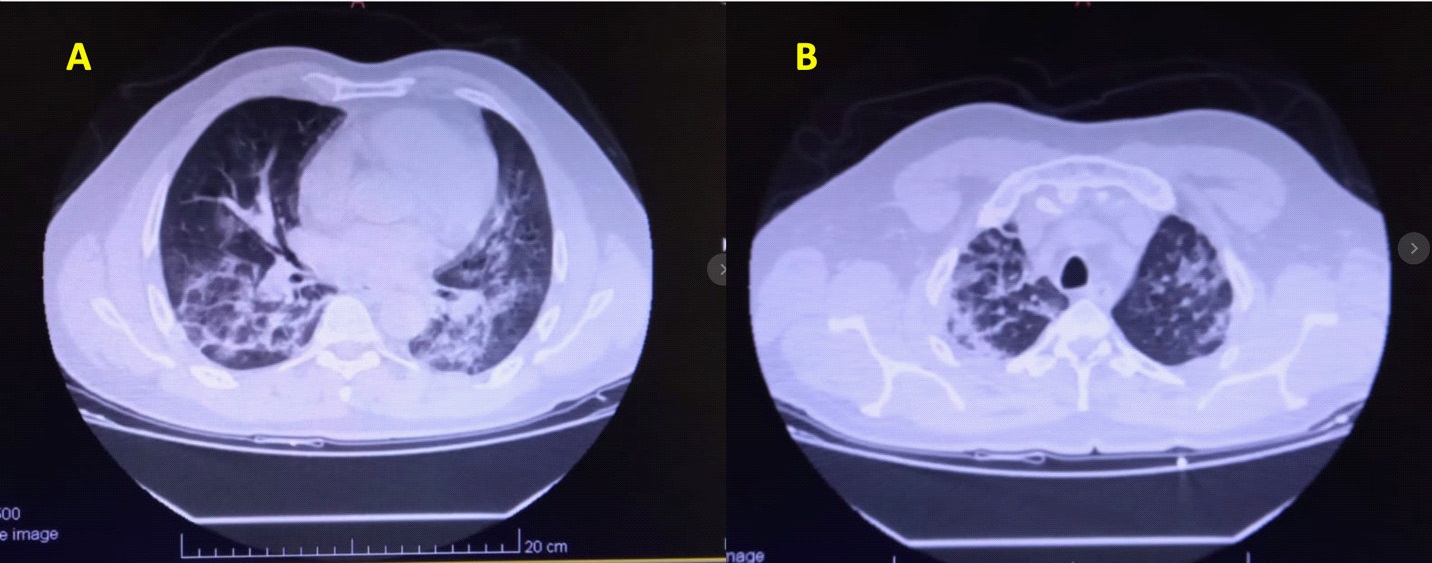
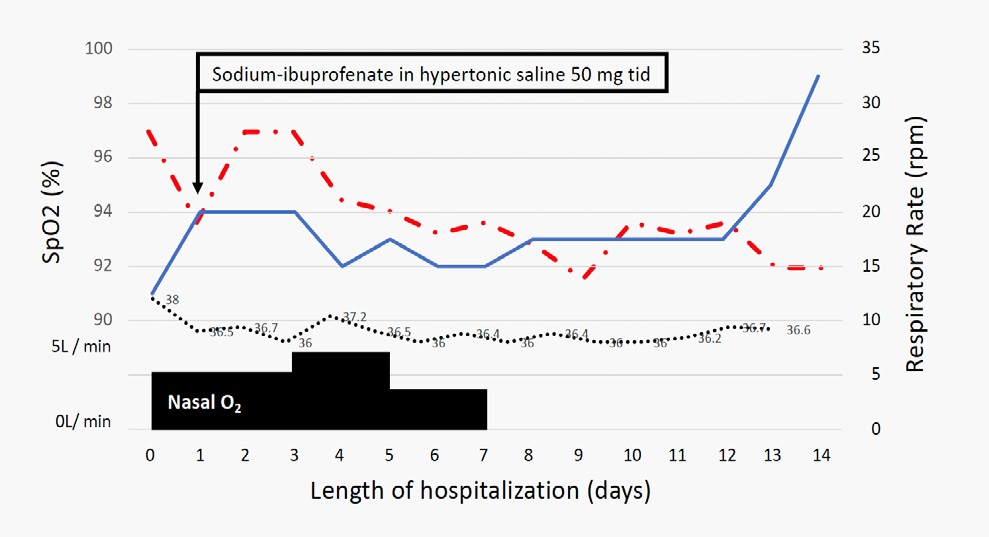
This 59 years old man was brought by the emergency service because of dry cough, fatigue, respiratory difficulties having a recent throat swab PCR positive. A fever of 38.4°C was noted. Status at the first consultation: Alert and conscious, but marked fatigue, blood pressure 126/80 mmHg, pulse 93/min, respiratory rate 32/min, body temperature 38.0°C, SpO2 91%/ RA. Physical findings: No pharyngeal redness, no lymphadenopathy, no edemas, but he presented respiratory distress findings (dyspnea, intercostal retraction, cyanosis etc.) associated to crackles, wheezes and stridor. Past history: Hypertension, no smoking history. Laboratory findings were listed in Table 2. Chest X-ray on admission showed bilateral infiltrative shadow in lower lung fields. Chest CT on the same day documented typical appearance of COVID-19 with bilateral and extensive GGO and lobe consolidation in both lung fields (Right = Left) (Figure 4, A and B). Initially the patient received enoxaparin 40 mg sc, bid, ceftriaxone 1 gr ev bid, clarithromycin 500 mg ev bid, and respiratory kinesiotherapy. Post-admission progress: The clinical course is illustrated in Figure 4. His body temperature on admission was 38.0°C, but then fluctuate between 37.2°C and 36.0°C. Oxygenation saturation on admission was 91-88% with nasal O2 4 L/min, after 24 hours, NaIHS was suggested to the patient. NaIHS was administered during 15 minutes using a high flow oxygen by face-mask at 6 L/min, health care worker utilized PPE.
Table 2: Results of laboratory tests conducted on the day of admission (Case 2).
Hematology |
Biochemistry |
||
WBC (μL) |
11000 |
C-reactive protein (mg/l) |
509 |
Neutrophils (%) |
83 |
Urea (mg/dl) |
36 |
Lymphocytes (%) |
14 |
Creatinine (mg/dl) |
1.0 |
Monocytes (%) |
2 |
Total Bilirubin (mg/dl) |
0.7 |
Eosinophils (%) |
1 |
AST (U/L) |
55 |
Basophils (%) |
0 |
ALT (U/L) |
56 |
Red Blood Cells (106/ μL) |
396 |
LDH (U/L) |
237 |
Hemoglobin (g/dl) |
11.7 |
ALP (U/P) |
69 |
Hematocrit (%) |
35.8 |
Sodium (mEq/L) |
132 |
Platelets (103/ μL) |
298 |
Potassium (mEq/L) |
2.9 |
D-dimer (ng/ml) |
2918 |
Chloride (mEq/l) |
96 |
Figure 5 shows the SpO2 along the hospitalization during NaIHS treatment, oxygen saturation initially was maintained in normal values with nasal prolongs at 3L, 4L, high flow oxygen by face-mask and after 6 days of treatment, no oxygen supplementation was required to maintain SpO2 >94%. NaIHS was given during 7 days 50 mg via nebulization tid. His appetite gradually improved, and after 7 days of treatment, oxygen was stopped, fatigue was improved and the patient tolerate walking without fatigue, desaturation occurred and it was associated to the presence of remanent wheezes until he was discharged (Day 14). A pharyngeal swab PCR was performed and the patient was ready for discharge.
Discussion
This report describes clinical outcomes in 2 cases of mild to moderate-stage COVID-19 with NaIHS, inhaled hypertonic ibuprofen, and obtained favorable results under a compassionate use program. The first described patient came to our institution after an intense spread of the virus in a home care setting of geriatric population. All these patients had poor oxygenation and pneumonia showing ground-glass shadow on chest CT. In our case, the course of multiple critical patient outcome parameters (NEWS2 Scores, RR and SpO2 ) improved rapidly following the initiation of NaIHS. Once admitted, it was clear in these patients, that respiratory failure was progressive, since SpO2 did not improve during the first 24 hours associates, as expected with an increase of respiratory rate. Once NaIHS was initiated, a rapid increase of SpO2 was observed but to maintained this oxygen improvement, NaIHS was given 3 times a day. By Day-6-8 following initiation of therapy, mean NEWS2 scores improved to <4, pulse oximetry had improved to ≥94% with diminishing supplemental oxygen needs, and respiratory rates had returned to normal. This presentation is different than the classical COVD19 pneumonia, which became rather serious, and as noted in many reports, it worsens rapidly 7–10 days after onset. These were impressive cases.
Today it is clear that inflammation is the hallmark of COVID-19 lung injury induced by viral infection, and it is expected that the antiviral and anti-inflammatory effects of NaIHS will be effective in treating lung injury caused by coronavirus, which is becoming more severe. The pulmonary delivery of NaIHS diminished bradykinin-storm physiology, likely reduced local prostaglandin and thromboxane synthesis and acutely improved the ventilation-perfusion mismatch which is a stamp of this disease.
Ibuprofen is not commercialized as inhaled drug in Argentina, but, due to the pandemic, Heath Authorities approved the compassionate use of some drugs. At that moment, our preclinical data provided information to speculate that ibuprofen continue to be safe even via nebulization. We tested the safety of higher doses in animals [12] during 1 hours nebulization period during 4 months and no significant histological changes were observed in rat lungs. Additionally, in humans’ serum ibuprofen increased from 0 to 1.09 ± 0.7 μg/ml and also the reactive oxygen species decreased from 1043 ± 252 to 441 ± 48 ΛU determined 30 min after the nebulization, demonstrating in spite of the low serum concentration, this presentation still maintains the anti-inflammatory property [13]. Finally, this beneficial effect induced by ibuprofen via nebulization in lung diseases was also observed with other NSAIDs such as indomethacin [14] evaluating the control of bronchiectasis severity, other pulmonary disease are under investigation [15,16]. The adverse events induced by NaIH were mild in these two patients. Only mild cough during the first nebulization. After 24 hours, these two patients tolerated very well NaIH, suggesting that ibuprofen via nebulization maintains the excellent safety profile observed previously.
Today, more than 15000 patients were treated with NaIH via a Compassionate Program in Argentina, a report will be available soon and based on these results a Phase II trial was authorized by the National Regulatory Authority (ANMAT #Expte 1-0047-0002-000699-20-9; Pegasus) hoping to end by the end of the year.
Conclusion
In conclusion, we report 2 cases of pneumonia COVID-19 with rapid reverse of hypoxia, complete recovery and excellence tolerance after NaIHS administration. After 7-10 days of treatment, hypoxia was recovered rapidly. These beneficial effects must be confirmed with the Phase 2 protocol, Pegasus, since early administration of NaIHS is safe, simple and inexpensive.
Authorship statement: Prof. Dr. German Ambasch and his team treated all the patients. Dr Néstor H. García contributed to design the protocol treatment for the patients based on the basic and clinical information about NaIHS. All other authors have contributed with the writing, editing and critically reviewed the manuscript.
All authors approved the final version of the manuscript, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors meet the ICMJE authorship criteria.
Declaration of competing interest: Authors certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
References
- WHO Coronavirus (COVID-19) Dashboard. 2021.
- The RECOVERY Collaborative Group. Dexamethasone in Hospitalized Patients with Covid-19. N Engl J Med. 2021; 384: 693– 704.
- Repurposed Antiviral Drugs for Covid-19 — Interim WHO Solidarity Trial Results. N Engl J Med. 2021 Feb 11; 384: 497–511.
- Simonovich VA, Burgos Pratx LD, Scibona P, Beruto MV, Vallone MG, Vázquez C, et al. A Randomized Trial of Convalescent Plasma in Covid-19 Severe Pneumonia. N Engl J Med. 2021; 384: 619–29.
- Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, et al. A Trial of Lopinavir–Ritonavir in Adults Hospitalized with Severe Covid-19. N Engl J Med. 2020; 382: 1787–99.
- Cavalcanti AB, Zampieri FG, Rosa RG, Azevedo LCP, Veiga VC, Avezum A, et al. Hydroxychloroquine with or without Azithromycin in Mild-to-Moderate Covid-19. N Engl J Med. 2020; 383: 2041–52.
- Stone JH, Frigault MJ, Serling-Boyd NJ, Fernandes AD, Harvey L, Foulkes AS, et al. Efficacy of Tocilizumab in Patients Hospitalized with Covid-19. N Engl J Med. 2020; 383: 2333–44.
- Garvin MR, Alvarez C, Miller JI, Prates ET, Walker AM, Amos BK, et al. A mechanistic model and therapeutic interventions for COVID-19 involving a RAS-mediated bradykinin storm. Elife. 2020; 9: e59177.
- Baraniuk JN, Lundgren JD, Mizoguchi H, Peden D, Gawin A, Merida M, et al. Bradykinin and Respiratory Mucous Membranes: Analysis of Bradykinin Binding Site Distribution and Secretory Responses In Vitro and In Vivo. Am Rev Respir Dis. 1990; 141: 706–14.
- Gholamreza-Fahimi E, Bisha M, Hahn J, Straßen U, Krybus M, Khosravani F, et al. Cyclooxygenase activity in bradykinin-induced dermal extravasation. A study in mice and humans. Biomedicine & Pharmacotherapy. 2020; 123: 109797.
- Nile SH, Nile A, Qiu J, Li L, Jia X, Kai G. COVID-19: Pathogenesis, cytokine storm and therapeutic potential of interferons. Cytokine & Growth Factor Reviews. 2020; 53: 66–70.
- Muñoz A, Alasino R, Garro A, Heredia V, García N, Cremonezzi D, et al. High Concentrations of Sodium Chloride Improve Microbicidal Activity of Ibuprofen against Common Cystic Fibrosis Pathogens. Pharmaceuticals. 2018; 11: 47.
- García NH, Porta DJ, Alasino RV, Muñoz SE, Beltramo DM. Ibuprofen, a traditional drug that may impact the course of COVID-19 new effective formulation in nebulizable solution. Medical Hypotheses. 2020; 144: 110079.
- Pizzutto SJ, Upham JW, Yerkovich ST, Chang AB. Inhaled nonsteroid anti-inflammatories for children and adults with bronchiectasis. Cochrane Airways Group, editor. Cochrane Database of Systematic Reviews. 2016.
- Sheikh Z, Ong HX, Pozzoli M, Young PM, Traini D. Is there a role for inhaled anti-inflammatory drugs in cystic fibrosis treatment? Expert Opinion on Orphan Drugs. 2018; 6: 69–84.
- Muralidharan P, Malapit M, Mallory E, Hayes D, Mansour HM. Inhalable nanoparticulate powders for respiratory delivery. Nanomedicine: Nanotechnology, Biology and Medicine. 2015; 11: 1189–99.