Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Coronary arteries and aortic valve calcifications in COVID-19: Not only lungs at chest CT
Marco Fogante, MD1*; Enrico Cavagna, MD2; Giovanni Rinaldi, MD2
1 Azienda Ospedaliero-Universitaria “Ospedali Riuniti” Ancona, Italy.
2 Ospedale “Infermi” Rimini, Italy
*Corresponding Author: Marco Fogante
University Hospital “Umberto I - Lancisi - Salesi”,
Department of Radiology, Ancona, Italy.
Email: marco.fogante89@gmail.com
Received : Sep 02, 2021
Accepted : Oct 05, 2021
Published : Oct 12, 2021
Archived : www.jcimcr.org
Copyright : © Fogante M (2021).
Abstract
Objectives: Cardiovascular diseases cause worse outcomes in patients affected by COVID-19. High coronary artery calcium score (CACS) and aortic valve calcifications (AC) increase cardiovascular risk. Our purpose is to evaluate CACS, measured by Weston Score (WS), and presence of AC in chest CT of COVID-19 patients and possibly to investigate their prognostic role.
Material and methods: This retrospective case-control study includes 150 hospitalized COVID-19 patients who underwent a chest CT at admission. The case group (Group A) was formed by 50 in-patients in Intensive Care Unit (ICU) under invasive ventilation (IV), while the matching control group for sex, age and BMI (Group B) was formed by 100 in-patients in non-ICU units, not under IV. After, a second case-control selection was originated from these two Groups: the case group (Group A1) composed by 30 patients selected from Group A, and the matching control group for age, sex, BMI and CT lung severity score (LSS) (Group B1) composed by 60 patients selected from Group B. WS and the presence of AC were compared between Groups A and B, and between Groups A1 and B1. Moreover, LSS and WS were correlated. Parametric tests were used for statistical analysis.
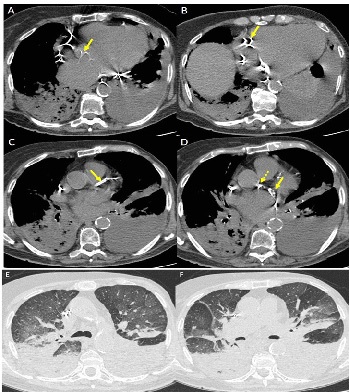
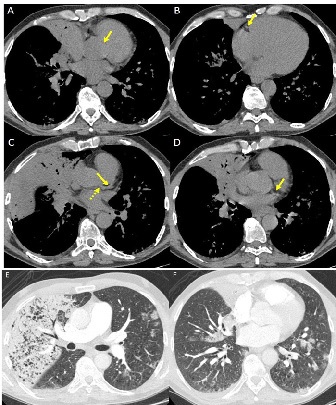
Material and methods: This retrospective case-control study includes 150 hospitalized COVID-19 patients who underwent a chest CT at admission. The case group (Group A) was formed by 50 in-patients in Intensive Care Unit (ICU) under invasive ventilation (IV), while the matching control group for sex, age and BMI (Group B) was formed by 100 in-patients in non-ICU units, not under IV. After, a second case-control selection was originated from these two Groups: the case group (Group A1) composed by 30 patients selected from Group A, and the matching control group for age, sex, BMI and CT lung severity score (LSS) (Group B1) composed by 60 patients selected from Group B. WS and the presence of AC were compared between Groups A and B, and between Groups A1 and B1. Moreover, LSS and WS were correlated. Parametric tests were used for statistical analysis.
Results: Among the 150 patients, 105 (70.0%) were male and 45 (30.0%) female. The age was 70.2 ± 13.3 years. LSS was 7.5 ± 3.9, WS was 6.4 ± 3.0; AC was present in 97/150 (64.7%). WS was significantly higher in Group A than in Group B, respectively, 7.4 ± 4.0 and 5.7 ± 2.6 (p=0.0146), and also the presence of AC, respectively, 41/50 (82.0%) vs 56/100 (56.0%) (p=0.0016). In Group B, LSS and WS had a linear positive correlation (r=0.2240, p=0.0405). Finally, WS and AC were significantly higher in Group A1 compared to Group B1.
Conclusions: Both WS and AC were higher in ICU COVID-19 patients than in non-ICU COVID-19 patients: they could have a predictive role in the pejorative evolution of the disease.
Keywords: COVID-19; chest CT; coronary artery calcium score; aortic valve calcifications.
Citation: Fogante M, Cavagna E, Rinaldi G. Coronary arteries and aortic valve calcifications in COVID-19: Not only lungs at chest CT. J Clin Images Med Case Rep. 2021; 2(5): 1355.
Introduction
Lower respiratory tract disease is the leading clinical manifestation of the COVID-19 pathology, and diffuse alveolar damage represents the histopathological condition of the severe acute respiratory syndrome (SARS) related to the new coronavirus (CoV-2) [1-3].
However, mortality in COVID-19 is often caused by major cardiovascular events, whose exact pathophysiological mechanisms are not well-understood [4]. COVID-19 might cause injuries to the cardiovascular system with different mechanisms: the SARS-CoV-2 host cell receptors, the angiotensin-converting enzyme 2, are expressed in the myocardial and endothelial cells; this could possibly be a reason for a direct damage; moreover, the excessive production of proinflammatory cytokines, induced by the virus, can lead to widespread cardiac and endothelial indirect damage [5,6]. As a matter of fact, in 8%-28% of COVID-19 patients, troponin increases in the early course of the disease, due to acute myocardial injury [7]. This event could be due to cardiac ischemia secondary to an imbalance between oxygen supply and demand, a condition that can occur in acute lung infections, such in COVID-19, especially in patients with chronic coronary heart disease [8]. Moreover, acute myocardial injury could be due to pre-existing coronary artery plaques that become unstable due to inflammatory activation, microvascular dysfunction and a pro-coagulatory state secondary to SARSCoV-2 infection [9-11]. The concomitance or a new onset of cardiac disease can worsen the prognosis of severe COVID-19, causing admission to the Intensive Care Unit (ICU), invasive ventilation (IV) and death [12-14].
Many papers show that Coronary Artery Calcium Score (CACS) is a strong independent predictor of mortality from cardiac disease [15,16]. Moreover, aortic valve calcifications (AC) is an important risk factor for cardiac events [17,18]. A visual CACS system, the Weston Score (WS), applied to non-gated chest CT, strongly correlates with the Agatston Score (AS), which is the traditional and most reliable method to evaluate CACS [19].
Up to now, pericardial effusion and cardiomegaly in chest CT have been reported as a possible cardiac involvement in COVID-19 patients [20]; however, to our knowledge no studies have described the presence of CACS and AC in these patients yet.
The purpose of our study is to evaluate CACS and AC in COVID-19 patients and to investigate their possible prognostic role in this disease.
Materials and methods
Study population
Informed consent was waived due to the COVID-19 emergency.
In this retrospective case-control study were selected 150 COVID-19 patients. COVID-19 was confirmed by RT-PCR with oropharyngeal swab and chest CT performed at admission time. All chest CTs were performed because of severe clinical conditions, sudden clinical deterioration, or mismatch between clinical and chest X-ray findings. For each patient we recorded sex, age, Body Mass Index (BMI), troponin value, presence of chronic pulmonary obstructive disease, hypertension, chronic renal disease, diabetes, and oncologic conditions.
The case group (Group A) was composed by 50 in-patients admitted to ICU and under IV; the matching control group for sex, age and BMI (Group B) was composed by 100 in-patients in a non-ICU Unit and not under IV (e.g. Infective Disease or Pulmonology Units).
After, from these two Groups we originated another casecontrol selection: The case group (Group A1) composed by 30 patients selected from Group A, and the matching control group for age, sex, BMI and CT lung severity score (LSS) (Group B1) composed by 60 patients selected from Group B.
Chest CT protocol
All CT examinations were performed on a 64-row CT scanner. Chest CTs were carried out without contrast medium, in a single inspiratory breath-hold, from the lung apices to the costophrenic recesses, using the following parameters: tube voltage 120 kVp; tube current 90-130 mAs; collimation 64 X 0.625. The CT images were reconstructed with slice increment and thickness of 0.625 mm. MPRs and MIPs images were also generated.
Chest-CT evaluation
Two radiologists reviewed in consensus chest CT images for lung evaluation and for CACS and AC assessment.
Lungs were analyzed with pulmonary parenchyma window setting (width/level: 1300/-600 HU). A semiquantitate CT lung severity score (LSS) was used: for each patient, percentage of involvement in each lobe was recorded as follows: 0 – none (0%); 1 – minimal (1–25%); 2 – mild (26–50%); 3 – moderate (51–75%); 4 – severe (76–100%). Total LSS was obtained adding the five lobes scores (range 0-20) [21].
WS was employed for CACS evaluation; a mediastinal window setting (width/level 400/40 HU) was used. We assigned to each patient a score for any of the major coronary vessels (left main trunk, left anterior descending artery, left circumflex artery, right coronary artery), as follows: 0 – no visually detected calcium; 1 – only a single high-density pixel was detected; 3 – calcium was dense enough to create blooming artefact; and 2 – calcium density between 1 and 3. The WS was calculated by adding the score of each vessel (range 0-12) [19]. Finally, the absence or presence of AC was evaluated in each patient.
Statistical analysis
Statistical analysis was performed with a dedicated software (MedCalc v19.1.6, MedCalc Software, Ostend, Belgium). Continuous variables were displayed as mean ± Standard Deviation (SD) and categorical variables were reported as count and percentage. Sex, age, BMI, troponin value, LSS, WS and AC were compared between Groups A and B and, then, between Groups A1 and B1. Moreover, LSS and WS were correlated both in our study population and in Groups A and B. Student’s T-Test was used to compare continuous variables. Chi-square or Fisher’s exact Test were used to compare categorical variables. Pearson’s Test (r) was used to correlate WS with LSS. A statistical significant value (p) of 0.05 or less indicated a statistically significant difference.
Results
Study population
Among the 150 included patients, 105 (70.0%) were male and 45 (30.0%) female. Their age was 70.2 ± 13.3 years; BMI was 25.1 ± 3.8 kg/m2 . Troponin value was 0.27 ± 0.11 ng/dL. LSS was 7.5 ± 3.9, WS was 6.4 ± 3.0 and AC were present in 97/150 (64.7%) patients. LSS and WS had a linear positive correlation (r=0.2186, p=0.0139).
The patient’s characteristics are reported in Table 1.
Table 1:Patients’ characteristics
Characteristics |
Study Population (n=150) |
Sex – M/F |
105 (70%) / 45 (30%) |
Age (years) |
70.2 ± 13.3 |
Body Mass Index (kg/m2) |
25.1 ± 3.8 |
Troponin value (ng/dL) |
0.27 ± 0.11 |
Chronic pulmonary obstructive disease |
11 (7.3%) |
Hypertension |
59 (39.3%) |
Chronic renal disease |
12 (8.0%) |
Diabetes |
44 (29.3%) |
Cancer |
17 (11.3%) |
LLS |
7.5 ± 3.9 |
WS |
6.4 ± 3.0 |
AC – Y/N |
97 (64.7%) / 53 (35.3%) |
Abbreviations – N: Number; M: Male; F: Female; LLS: Lung Severity Score; WS: Weston Score; AC: Aortic Valve Calcification; Y: Yes; N: No.
Comparison between Groups A and B
LLS was 10.0 ± 5.3 in Group A and 6.4 ± 3.4 in Group B, with a statistically significant difference (p=0.0002). Troponin value was 0.37 ± 0.17 ng/mL in Group A and 0.19 ± 0.07 ng/mL in Group B with a statistically significant difference (p=0.0001). Statistically significant differences were also present in WS, 7.4 ± 4.0 in Group A and 5.7 ± 2.6 in Group B (p=0.0146), and in the presence of AC (p=0.0016). In Group B, LSS and WS had a linear positive correlation (r=0.2240, p=0.0405); instead, in Group A there was no correlation between these values (r=0.0238, p=0.8806).
The comparison between Group A and B is reported in Table 2.
Comparison between Groups A1 and B1
There was a statistically significant difference in WS between Groups A1 and B1, respectively, 7.9 ± 3.7 and 4.4 ± 2.6 (p=0.0007). Moreover, the presence of AC was significantly higher in Group A1 [27/30 (90.0%)] than in Group B1 [35/60 (58.3%)], p=0.0032.
The comparison between Groups A1 and B1 is reported in Table 3.
Table 2: Comparison between Groups A and B.
|
Group A |
Group B |
p |
Sex – M/F |
35 (70%) / 30 (30%) |
70 (70%) / 30 (30%) |
1.0000 |
Age (years) |
68.6 ± 13.5 |
71.0 ± 13.4 |
0.5740 |
Body Mass Index (kg/m2) |
25.4 ± 3.5 |
24.9 ± 4.0 |
0.3870 |
Troponin value (ng/dL) |
0.37 ± 0.17 |
0.19 ± 0.07 |
0.0001 |
LLS |
10.0 ± 5.3 |
6.4 ± 3.4 |
0.0002 |
WS |
7.4 ± 4.0 |
5.7 ± 2.6 |
0.0146 |
AC – Y/N |
41 (82%) / 9 (30%) |
56 (56.0%) / 44 (44.0%) |
0.0016 |
Abbreviations – N: Number; M: Male; F: Female; LLS: Lung Severity Score; WS: Weston Score; AC: Aortic Valve Calcification; Y: Yes; N: No.
Table 3: Comparison between Groups A1 and B1.
|
Group A1 |
Group B1 |
p |
Sex – M/F |
21 (70%) / 9 (30%) |
38 (63.3%) / 22 (36.7%) |
0.6401 |
Age (years) |
67.4 ± 13.7 |
70.6 ± 13.8 |
0.3014 |
Body Mass Index (kg/m2) |
25.2 ± 4.2 |
24.7 ± 4.1 |
0.5899 |
LLS |
5.5 ± 2.3 |
5.1 ± 2.6 |
0.4771 |
WS |
7.9 ± 3.7 |
4.4 ± 2.6 |
0.0007 |
AC – Y/N |
27 (90.0%) / 3 (10.0%) |
35 (58.3%) / 25 (41.7%) |
0.0032 |
Abbreviations – N: Number; M: Male; F: Female; LLS: Lung Severity Score; WS: Weston Score; AC: Aortic Valve Calcification; Y: Yes; N: No.
Discussion
Acute myocardial injury, detected by troponin increase in the early course of the disease, may be one of the clinical consequences of COVID-19 [7,8]. The onset and/or the presence of cardiac disease are related to a more severe prognosis in COVID-19 patients, often leading to ICU admission, IV or death [12-14]. CACS and AC are strong independent predictors of cardiac mortality and therefore they add incremental prognostic information to estimate cardiac disease [15-18]. The WS, a visual CACS system on standard non-gated chest-CT, strongly correlates with AS, which is considered the most traditional and reliable method to evaluate CACS [19].
The purpose of our study was to evaluate CACS by use of WS in non-gated chest CT, and the presence of AC in COVID-19 patients and eventually to investigate their prognostic role in this disease.
In our study, comparison between in-patients in ICU under IV (Group A) and in-patients with same age, sex and BMI but in non-ICU Units and not under IV (Group B) led to interesting results. First, WS was significantly higher in Group A than in Group B, respectively, 7.4 ± 4.0 and 5.7±2.6 (p=0.0146). This result suggests that CACS, measured by WS, could be an important prognostic factor for COVID-19 disease severity. Our hypothesis seems to be supported by previous works [17,22,23], in which the authors concluded that a WS > 7 (as it proved to be in Group A of our study), well correlates with an AS > 400, and this value indicates a high risk of cardiac disease, independent from underlying risk factors [24]. Conversely, a WS between 2 and 7 (as it proved to be in Group B of our study) well correlates with an AC between 100 and 400 [22,23], which is associated with moderate cardiovascular risk [24,25]. These results are in agreement with the assumption that COVID-19 patients who are also at increased risk for cardiac disease, carry a higher risk of worse COVID-19 evolution, related to ICU admission and IV support [12-14], and that CACS may be an indicator of this risk. Moreover, cardiovascular morbidity and mortality are higher in patients with AC [26]. In our study, the higher rate of AC in Group A (82.0%) compared to Group B (56.0%) well correlates with this assumption: AC could be a second indicator of higher risk of cardiac disease and worse evolution in COVID-19 patients. Then, to prove if CACS and AC could be independent factors of greater risk of ICU admission, we carried out a further selection of patients in two groups who had comparable LSS (Groups A1 and B1). It is noteworthy that both CACS and AC values are also different in these further groups. Especially, in ICU patients compared with patients in non-ICU Units, the WS and AC were significantly higher, respectively, 7.9 ± 3.7 vs 4.4 ± 2.6, (p=0.0007), and 27/30 (90.0%) vs 35/25 (58.3%) (p=0.0032). Based on these results we hypothesize that these factors may be independent risk factors for patient admission to ICU with need for IV. Finally, we correlated the LSS and WS, to verify if CACS could be related to lung involvement severity in COVID-19 patients. Interestingly, a statistically significant correlation was found both in all our study population and in patients in non-ICU Units (Group B). A possible explanation of these results could be that CACS is a surrogate marker of subclinical atherosclerosis, which is associated with inflammatory markers [27]. Therefore severity of the initial lung involvement, particularly when lung disease does not require IV, may be partly related to an underlying sub-clinical and pro-inflammatory state that may impair immune response and pulmonary function.
To our knowledge this study represents the first pilot study to understand how the radiologist can further contribute to improve risk stratification in COVID-19 patients, evaluating cardiovascular findings (mainly coronary arteries calcium using WS and presence of AC) in chest CT exams, in addition to pericardial effusion and cardiomegaly.
This study has some limitations. First, it is a retrospective mono-centric study. Second, the WS, AC and LSS were evaluated in consensus by two radiologists but the inter-observer agreement was not calculated.
Conclusion
In conclusion, the radiologist can contribute to improve risk stratification in COVID-19 patients, evaluating cardiovascular findings in chest CT exams. CACS (WS) and AC may have a predictive role in the evolution of COVID-19, since both correlate with a higher risk of cardiac disease and this can lead to a worse evolution, with ICU admission and IV support. Besides, since we found a statistically significant correlation between LSS and WS in all our population and in non-ICU patients, CACS could be a surrogate marker of subclinical atherosclerosis and pro-inflammatory state, which could worsen also lung involvement in COVID-19 patients. Further prospective and multi-centric studies are needed to confirm our hypothesis.
Conflict of interest: The author declared no potential conflicts of interests associated with this study.
Ethical standards: All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. Institutional review board approved the study.
Disclosure: The authors have no disclosure.
Funding: This research did not receive any specific grant from funding agencies in the public, commercial, or pnot-forprofit sectors.
References
- Situation report – 182. Coronavirus disease 2019 (COVID-19). World Health Organization. 2020.
- Sauter JL, Baine MK, Butnor KJ, et al. Buonocore DJ, Chang JC, Jungbluth AA, et al. Insights Into Pathogenesis of Fatal COVID-19 Pneumonia From Histopathology With Immunohistochemical and Viral RNA Studies. Histopathology. 2020.
- Mangalmurti NS, Reilly JP, Cines DB, et al. COVID-ARDS Clarified: A Vascular Endotype? Am J Respir Crit Care Med. 2020.
- Ragab D, Eldin HS, Taeimah M, et al. The COVID-19 Cytokine Storm; What We Know So Far. Front Immunol 2020; 11: 1446.
- Mishra AK, Sahu KK, George AA, et al. A review of cardiac manifestations and predictors of outcome in patients with COVID - 19. Heart Lung. 2020.
- Chen L, Li X, Chen M, et al. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS-CoV-2. Cardiovasc Res. 2020; 116: 1097–1100.
- Shi S, Qin M, Shen B, et al. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020; 395: 497–506.
- Wu C, Chen X, Cai Y. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020.
- Bikdeli B, Madhavan MV, Jimenez D, et al. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up. J Am Coll Cardiol. 2020; 75: 2950–73.
- Guo T, Fan Y, Chen M. Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019. JAMA Cardiol. 2020.
- Varga Z, Flammer AJ, Steiger P, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020; 395: 1417–1418.
- Wei J, Huang F, Xiong T, et al. Acute myocardial injury is common in patients with COVID-19 and impairs their prognosis. Heart. 2020; 106: 1154-1159.
- Montone RA, Iannaccone G, Meucci MC, et al. Myocardial and Microvascular Injury Due to Coronavirus Disease 2019. Eur Cardiol. 2020; 15: e52.
- Onder G, Rezza G, Brusaferro S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. JAMA. 2020; 323: 1775-1776.
- van der Aalst CM, Denissen S, Vonder M, et al. Screening for Cardiovascular Disease Risk Using Traditional Risk Factor Assessment or Coronary Artery Calcium Scoring: The ROBINSCA Trial. Eur Heart J Cardiovasc Imaging. 2020.
- Rozanski A, Arnson Y, Gransar H, et al. Associations Among Selfreported Physical Activity, Coronary Artery Calcium Scores, and Mortality Risk in Older Adults. Mayo Clin Proc Innov Qual Outcomes. 2020; 4: 229-237.
- Fletcher AJ, Singh T, Syed MBJ, et al. Imaging aortic valve calcification: significance, approach and implications. Clin Radiol. 2020.
- Krishnamoorthy P, Gupta S, Lu M, et al. Usefulness of the Echocardiographic Calcium Score to Refine Risk of Major Adverse Cardiovascular Events Beyond the Traditional Framingham Risk Score. Am J Cardiol. 2019; 123: 392-395.
- Kirsch J, Buitrago I, Mohammed T, et al. Detection of Coronary Calcium During Standard Chest Computed Tomography Correlates With Multi-Detector Computed Tomography Coronary Artery Calcium Score. Int J Cardiovasc Imaging. 2012; 28: 1249- 1256.
- Wu J., Wu X., Zeng W. Chest CT findings in patients with coronavirus disease 2019 and its relationship with clinical features. Invest Radiol. 2020.
- Li K, Fang Y, Li W, et al. CT image visual quantitative evaluation and clinical classification of coronavirus disease (COVID-19). Eur Radiol. 2020; 30: 4407-4416.
- Bhatt S, Kazerooni E, Newell Jr J, et al. Visual Estimate of Coronary Artery Calcium Predicts Cardiovascular Disease in COPD. Chest. 2018; 154: 579-587.
- Chandra D, Gupta A, Leader J, et al. Assessment of coronary artery calcium by chest CT compared with EKG-gated cardiac CT in the multicenter AIDS cohort study. PLoS One. 2017; 12: e0176557.
- Zimmermann G, Rüther T, Ziegler F, et al. Predictive value of coronary calcifications for future cardiac events in asymptomatic patients: underestimation of risk in asymptomatic smokers Int J Cardiovasc Imaging. 2019; 35: 1387-1393.
- Hong J, Blankstein R, Shaw L, et al. Implications of Coronary Artery Calcium Testing for Treatment Decisions Among Statin Candidates According to the ACC/AHA Cholesterol Management Guidelines: A Cost-Effectiveness Analysis JACC Cardiovasc Imaging. 2017; 10: 938-952.
- Thomassen HK, Cioffi G, Gerdts E, et al Echocardiographic aortic valve calcification and outcomes in women and men with aortic stenosis. Heart 2017; 103: 1619–1624.
- Mehta A, Patel J, Rifai M, et al. Inflammation and coronary artery calcification in South Asians: The Mediators of Atherosclerosis in South Asians Living in America (MASALA) study. Atherosclerosis. 2018; 270: 49-56.