Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Two cases of massive hemoptysis due to bronchial artery supply to previously embolized pulmonary arteriovenous malformation
Aoi Ootaka; Tomoki Tozawa; Masazumi Matsuda; Motoko Sasajima; Kento Hatakeyama; Emika Murasawa; Takahiro Ishinari; Manabu Hashimoto*
Department of Radiology, Akita University School of Medicine, Japan
*Corresponding Author: Manabu Hashimoto
Department of Radiology, Akita University School of
Medicine, 1-1-1 Hondo Akita City, Akita, 010-8543,
Japan.
Email: : hashimms@med.akita-u.ac.jp
Received : Sep 03, 2021
Accepted : Oct 05, 2021
Published : Oct 12, 2021
Archived : www.jcimcr.org
Copyright : © Hashimoto M (2021).
Abstract
We present two patients who experienced massive hemoptysis during follow up after pulmonary artery embolization with coils for Pulmonary Arteriovenous Malformations (PAVM). The treated PAVMs responsible for hemoptysis were supplied from the bronchial artery. Hemoptysis was controlled for a short time by bronchial artery embolization with n-butyl-2-cyanoacrylate in case 1 and gelatin sponge in Case 2. Thereafter, however, one patient (Case 2) died of recurrent massive hemoptysis. These cases may indicate that bronchial artery supply is the cause of both hemoptysis and reperfusion of treated PAVMs.
Keywords: pulmonary arteriovenous malformation; embolotherapy; hemoptysis.
Citation: Hashimoto M, Ootaka A, Tozawa T, Matsuda M, Sasajima M. Two cases of massive hemoptysis due to bronchial artery supply to previously embolized pulmonary arteriovenous malformation. J Clin Images Med Case Rep. 2021; 2(5): 1356.
Introduction
Since the first report on Pulmonary Arteriovenous Malformation (PAVM) embolization by Porstmann in 1977 [1], pulmonary artery embolization has become the first-line procedure for the treatment of PAVM. The most important purpose of embolization is to prevent paradoxical embolization and rupture [2].
The recurrence of treated PAVMs is not rare. During followup, flow through previously placed coils (recanalization) or flow through accessory arteries (reperfusion) of treated PAVMs is found in up to 25% of lesions [3,4]. Woodward et al reported that recanalization of the embolized vessels was the most frequent pattern, with a high success rate of retreatment (91%) in cases with recurrence [3]. Because many collateral vessels arising from an adjacent pulmonary artery and its capillary bed exist, reperfused PAVMs are difficult to treat [3]. Cusumano et al showed that treatment success rate of repeated embolization for recurrent PAVMs was 54.7% [4].
Although it may be a rare pattern of recurrence, bronchial artery supply can cause reperfusion of treated PAVMs [5-8]. In these reports, some of the patients with bronchial artery supply to previously treated PAVMs presented hemoptysis [5-8]. We herein report two cases of recurrent PAVMs that were caused by bronchial artery supply leading to massive hemoptysis (estimated bleeding >100 mL/d).
Case reports
Case I
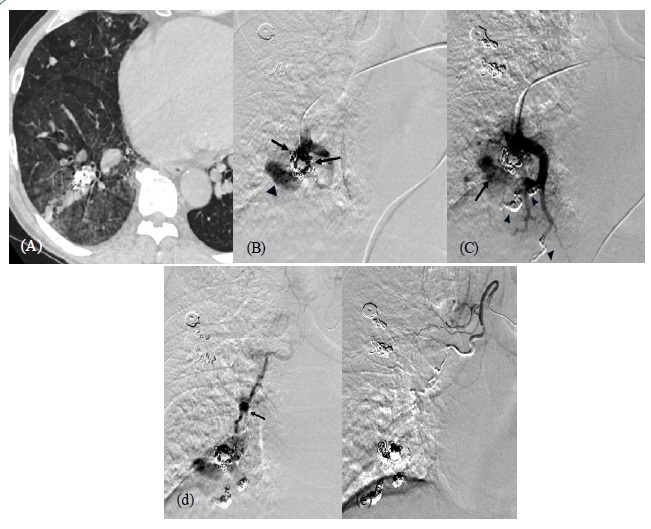
A 53-year-old man with Hereditary Hemorrhagic Telangiectasia (HHT) presented with hemoptysis. He had undergone coil embolization of multiple PAVMs six times over a 12-year period. The last session was performed 39 months ago. CT on admission showed an increased lung density suggestive of bleeding around the treated PAVM in the right lower lobe (Figure 1a). We performed embolization of the feeding artery of the PAVM with microcoils (Azur CX 18; Terumo Medical, Target XL; Stryker). Adjacent PAVMs were also embolized simultaneously (Figure 1b,c). However, we could not obtain complete stasis in the PAVM. Slow residual flow remained in the PAVM. Hemoptysis did not subside after pulmonary artery embolization. Two days later, we decided to perform an angiography of the bronchial artery (Figure 1d,e). It showed that the PAVM was supplied by the right bronchial artery. We advanced a microcatheter coaxially into the target branch of the right bronchial artery as distally as possible to obtain a secure catheter position and avoid the spinal branch. Then, an insignificant arterial dissection occurred and thereby, antegrade flow of the artery slowed. We decided to use n-butyl-2-cyanoacrylate (NBCA) mixed with iodized oil (Lipiodol Ultrafluid; Guerbet, Roisy, France) at a ratio of 1:6 as an embolic material. The bronchial artery was embolized by careful injected 0.4ml of the mixture under digital subtraction angiography, without causing any complication or migrating the embolic material to the pulmonary vein. After this treatment, hemoptysis disappeared during 16 months of follow-up.
Case II
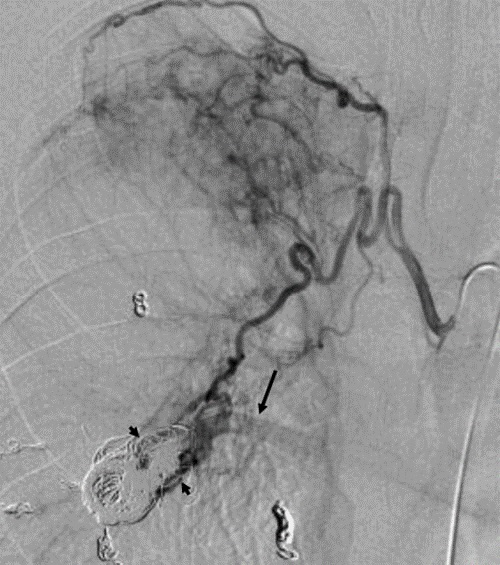
A 35-year-old man with HHT presented with recurrent hemoptysis. He had undergone two coil embolization sessions for multiple PAVMs in the past 5 years. Bronchoscopy on admission demonstrated that bleeding was from the right lower bronchus. Pulmonary angiography showed recanalization of the treated PAVM in the right S9. We embolized the feeding artery of the PAVM in the right S9 with additional coils (Tornade .035inch; Cook Medical). Thereafter, a small amount of hemoptysis remained. Ten months later, massive hemoptysis developed. Pulmonary angiography showed complete occlusion of the treated PAVM. Right bronchial angiography showed reperfusion of the treated PAVM (Figure 2). We advanced a microcatheter into the proximal portion of the target branch of the right bronchial artery and performed embolization with gelatin sponge of 2mm in size (Gelpart; Nihon Kayaku, Tokyo, Japan) without complications. Before use, we crushed the particles into even smaller particles by pumping several times using a 3-way stopcock. We injected gelatin sponge until flow reduced significantly. The patient’s condition improved after bronchial artery embolization. Massive hemoptysis recurred 4 months later. We attempted to perform right bronchial artery embolization with gelatin sponge in the same way. There was no complication. After that, he complained of several episodes of a small amount of hemoptysis. Ten months later, he died of massive hemoptysis in his house.
Discussion
Communication between the bronchial and pulmonary arteries exists at the respiratory bronchiole level in the normal lung. This communication can become enlarged and act as systemic-to-pulmonary reperfusion of treated PAVMs after pulmonary artery embolization [9]. The inflammatory process around treated PAVMs may accelerate reperfusion through the bronchial artery [7]. To reduce the existing of the reperfusion, we should occlude feeding pulmonary branches as close as possible to the PAVMs.
In our cases, massive hemoptysis developed after pulmonary artery embolization for PAVMs. Although we could not achieve complete stasis by pulmonary artery embolization in the treated PAVMs leading to bleeding in 1 of 2 cases, both were reperfused from the bronchial artery and hemoptysis was controlled for a short time after bronchial artery embolization. The results of two cases may reflect that bronchial artery supply is the cause of both hemoptysis and reperfusion of treated PAVMs. It is not clear when we should perform bronchial angiography for the evaluation of recurrent PAVMs. However, we consider hemoptysis to be a critical symptom related to the existence of systemic reperfusion of treated PAVMs.
The embolic agent used for bronchial artery embolization was NBCA in case 1 and gelatin sponge in case 2. The selection of embolus was made based on the operator’s decision at the time of procedure. Although bronchial artery embolization with NBCA may provide higher hemoptysis-free survival in comparison to polyvinyl alcohol particles [10], we think that gelatin sponge was effective for the short-term management of hemoptysis. Recurrent massive hemoptysis led to lethal hemoptysis in case 2. NBCA might be a preferable embolic agent for controlling hemoptysis in cases involving repeated bronchial artery embolization. Although procedure-related complication was not observed in our cases, optimal embolic agents remain unclear. It is important to avoid passage of embolus through PAVMs leading to paradoxical embolization. When it is feasible to advance a microcatheter into the distal portion of the bronchial artery close to the PAVMs, microcoils may be suitable embolus for embolization. If not, we think NBCA can be used as embolic agents, but careful injection is required.
Previous reports documented surgical resection as the treatment of choice for patients with hemoptysis due to systemic artery supply to the previously treated PAVMs [5,7]. We are not sure that surgery is the first-line therapy for the patients who tolerate surgery. When patients present with recurrent massive hemoptysis after bronchial artery embolization as in case 2, however, we think surgical resection is the treatment of choice.
We reported two cases with massive hemoptysis after pulmonary artery embolization for PAVMs. The PAVMs responsible for bleeding were reperfused by the bronchial artery. Bronchial artery embolization was effective for controlling hemoptysis, at least for a short time.
References
- Porstmann W. Therapeutic embolization of arterio-venous pulmonary fistula by catheter technique. In: Kelop O, editor. Current concepts in pediatric radiology. Springer. 1977; 23-31.
- White RI Jr. Pulmonary arteriovenous malformations: how do we diagnose them and why is it important to do so? Radiology. 1992; 182: 633-635.
- Woodward CS, Pyeritz RE, Chittams JL, Trerotola SO. Treated pulmonary arteriovenous malformations: Patterns of persistence and associated retreatment success. Radiology. 2013; 269: 919- 926.
- Cusumano LR, Duckwiler GR, Roberts DG, McWlliams JP. Treatment of recurrent pulmonary arteriovenous malformations: comparison of proximal versus distal embolization technique. Cardiovasc Interv Radiol. 2020; 43: 29-36.
- De Wispelaere JF, Trigaux JP, Weynants P, Delos M, De Coene B. Systemic supply to a pulmonary arteriovenous malformation: Potential explanation for recurrence. Cardiovasc Interv Radiol. 1996; 19: 285-287.
- Sagara K, Miyazono N, Inoue h, Ueno K, Nishida H, et al. Recanalization after coil embolotherapy of pulmonary arteriovenous malformations: study of long-term outcome and mechanism for recanalization. Am J Roentgenol. 1998; 170: 727-730.
- Kasai H, Sugiura T, Kobayashi T, Okamura R, Oota M, et al. Recurrent of pulmonary arteriovenous malformation with nontuberculous mycobacteria infection caused by perfusion from pulmonary artery and bronchial artery after coil embolization. Intern Med. 2019;58: 1593-1596.
- Pierucci P, Murphy J, Henderson KJ, Chyun DA, White RI Jr. New definition and natural history of patients with diffuse pulmonary arteriovenous maiformations. Chest. 2008; 133: 653-661.
- Sherrier RH, Chiles C, Newman GE. Chronic multiple pulmonary emboli: regional response of the bronchial circulation. Invest Radiol 1989; 24: 437-441.
- Woo S, Yoon CJ, Chung JW, Kang SG, Jae HJ, et al. Bronchial artery embolization to control hemoptysis: Comparison of NButyl-2-cyanoacrylate and polyvinyl alcohol particles. Radiology 2013; 269: 594-602.