Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Pulmonary thromboembolism as presenting feature of COVID-19: Report of four cases
Negar Omidi1; Seyyed Mojtaba Ghorashi1; Masoumeh Lotfi-Tokaldany1; Shaghayegh Ghasemi2; Shapour Shirani1*
1 Tehran Heart center, Tehran University of Medical Sciences, Tehran, Iran.
2 Tehran University of Medical Sciences, Tehran, Iran.
*Corresponding Author: Shapour Shirani
Head of Imaging Department, Tehran Heart Center,
Tehran University of Medical Sciences, 1411713138,
Tehran, Iran.
Email: mojtaba.ghorashi8@gmail.com
Received : Sep 01, 2021
Accepted : Oct 07, 2021
Published : Oct 14, 2021
Archived : www.jcimcr.org
Copyright : © Shirani S (2021).
Abstract
Coronavirus disease 2019 (COVID-19) infection is associated with a hypercoagulable state. The similarity in clinical presentation between Pulmonary Thromboembolism (PTE) and COVID-19 render the triage of these patients challenging. We present four cases of COVID-19 who were admitted with first impression of pulmonary thromboembolism in emergency department. The patients were male with age ranged between 46 and 60 years old. None of the patients had criteria of severe COVID-19. Pulmonary Computed Tomography (CT) angiography at admission was consistent with PTE. Incidental finding in pulmonary CT angiography was lung lesions in favor of COVID-19. Pulmonary thromboembolism might be a first presentation feature in COVID-19 patients. Overlaps between presenting symptoms and paraclinical findings such as chest CT and elevations in D-dimer levels in PTE and COVID-19 should be kept in mind.
Keywords: COVID-19; coronavirus; pulmonary thromboembolism; computed tomography.
Citation: Omidi N, Ghorashi SM, Lotfi-Tokaldany M, Ghasemi S, Shirani S. Pulmonary thromboembolism as presenting feature of COVID-19: Report of four cases. J Clin Images Med Case Rep. 2021; 2(5): 1363.
Introduction
A Coronavirus disease 2019 (COVID-19) infection is associated with a hypercoagulable state and increased levels of serum D-dimer and fibrinogen [1]. According to autopsy findings in 12 patients with COVID-19, the prevalence of venous thrombosis was 58% in those not suspected to have thrombosis, and the main cause of death was Pulmonary Thromboembolism (PTE) in about 30% of patients [2]. Llitjos et al. reported that around 70% of mechanically ventilated patients with COVID-19 that received anticoagulation had venous thromboembolism by screening [3]. The similarity in clinical presentation between PTE and COVID-19 and the increased D-dimer level in both render the triage of these patients challenging.
So far, in most of the cases reported on thromboembolic events in COVID-19, embolic events occurred during the period of hospital admission. In this report we introduce four patients who had their first clinical presentation of COVID-19 infection in the form of PTE. These patients were admitted in emergency department with impression of unprovoked PTE, while COVID-19 pneumonia found incidentally in pulmonary Computed Tomography Angiography (CTA).
Case presentation
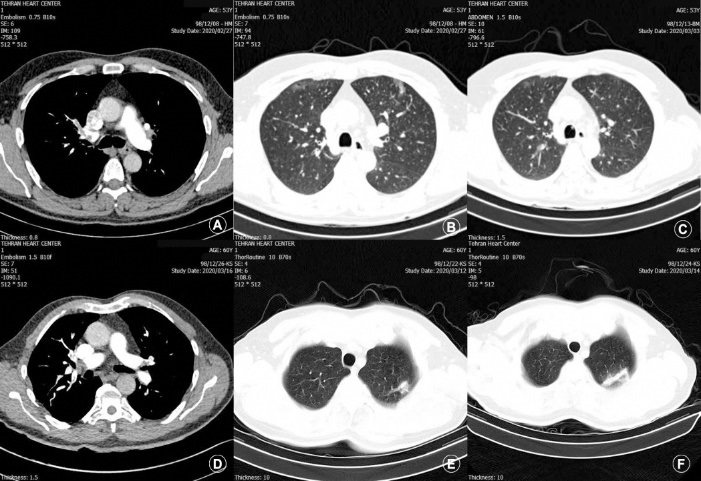
This study was approved by the Ethics Committee of Tehran University of Medical Sciences (ethical code: IR.TUMS.VCR.REC.1399.111) and was conducted in compliance with the Helsinki Declaration. This case series study includes four patients with first impression of PTE, admitted in emergency department of Tehran Heart Center, Tehran, Iran [4]; from 28 February 2020 until 19 April 2020. Thoracic 16-slice CT (SOMATOM Sensation 16, Siemens Healthcare, Forchheim, Germany) without contrast and pulmonary CT angiography with a dual-source 128-slice CT scanner (SOMATOM Flash, Siemens Healthcare, Forchheim, Germany) were performed for the patients at admission. Pulmonary CT angiography at admission was consistent with PTE (Figure 1). Incidental finding in pulmonary CT angiography was lung lesions in favor of COVID-19; however the result of realtime reverse-transcriptase-polymerase assay was negative for the patients. The bleeding risk was assessed, and the patients were kept under regular observation for early recognition of possible renal impairment, liver dysfunction, and respiratory distress.
The Table 1 demonstrates demographic and clinical characteristics, and laboratory findings of the patients. All four patients were male with age ranging between 46 and 60 years old. Patient number 1 had deep vein thrombosis in color Doppler ultrasonography. The patients received at least one approved antiviral treatment in addition to anticoagulant or Catheter Directed Thrombolysis (CDT). Given the negative real-time reverse-transcriptase-polymerase assay results, spiral chest CT was repeated to follow the pattern of lung involvement. All the patients were afebrile on admission and none had severe form of COVID-19 infection. Although all were admitted in ICU, none needed mechanical ventilation. None had bleeding episode during hospital stay. The level of D-dimer rose to at least 5 times the normal value (>0.5 µg/mL and range = 2.76 - 8.79 µg/mL). In case number 4, the location of lung injury was not concordant with thromboembolic segments. In case number 3, lung injury was misdiagnosed as pulmonary infarction, however the serial chest CT revealed progression of lung lesions compatible with COVID-19. The patients were recovered and discharged alive.
Table 1: Demographic, clinical, chest CT and laboratory findings of the cases
|
Case 1 |
Case 2 |
Case 3 |
Case4 |
Sex |
Male |
Male |
Male |
Male |
Age |
46 |
53 |
60 |
52 |
Chief complaint |
Right leg edema |
Dyspnea |
Chest pain |
Dyspnea |
Past medical history/risk factors |
Smoker |
Smoker |
- |
Dyslipidemia |
LV Ejection fraction (%) |
55 |
45-50 |
55 |
50 |
Pulmonary CT angiography |
Bilateral lobar PTE, cons. In LLL and RLL |
Bilateral lobar PTE, RV strain |
Bilateral lobar PTE |
LUL and LLL PTE |
Second CT |
GGO: LUL |
GGO: RUL and LUL |
GGO: LUL |
GGO: RUL and LUL |
Intervention/Medication |
UFH |
CDT/UFH/HCQ/ |
UFH/HCQ |
CDT/NOAC/UFH |
Length of hospital stay (days) |
8 |
10 |
8 |
7 |
Laboratory findings |
|
|
|
|
WBC (count/mm3) |
16610 |
14910 |
11410 |
8100 |
Lymphocyte (%) |
8.1 |
18.3 |
26.4 |
33.1 |
Platelet count/µL |
368000 |
181000 |
329000 |
105200 |
CRP (mg/dL) |
9.11 |
6.9 |
3.24 |
5.5 |
D-Dimer (µg/mL) |
- |
7.52 |
2.76 |
5.99 |
hs-TnI (ng/mL) |
60.1 |
1051.5 |
37 |
77.5 |
ALT (IU/L) |
29 |
47 |
- |
37 |
AST (IU/L) |
41 |
72 |
- |
34 |
Prothrombin time (sec) |
10.6 |
13.3 |
12.4 |
- |
INR |
1 |
1.31 |
1.2 |
- |
LV; Left Ventricular; CT: Computed Tomography; GGO: Ground Glass Opacity: LLL: Left Lower Lobe; RLL: Right Lower Lobe: LUL: Left Upper Lobe: RUL: Right Upper Lobe; UFH: Unfractionated Heparin; HCQ: Hydroxychloroquine; NOAC: Novel Oral Anticoagulant; CDT: Catheter Directed Thrombolysis; IU/L: International Units Per Liter; WBC: White Blood Cell; CRP; C-Reactive Protein; Hs-Tni: High Sensitive Troponin I; ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase; INR: International Normalized Ratio. |
||||
Discussion
As suggested in previous studies, a severe complication of COVID-19 is viral pneumonia, which may predispose patients to PTE by activating serious inflammation and rendering them prone to a thrombotic state [5,6]. The thrombotic state as a side effect of antiviral therapy was excluded in our study population as none of them was under the antiviral treatment prior to admission. Both scenarios should be considered: PTE as the first presentation or as a complication in the course of COVID-19 [17].
The concomitant presentation of PTE without common predisposing factors is an issue that could be explained by a change in the coagulation cascade in patients with COVID-19. Overlaps between presenting symptoms and paraclinical findings such as chest CT and elevations in D-dimer levels in PTE and COVID-19 should be kept in mind. Clinical symptoms such as fever should be interpreted with regard to C-reactive protein, the lymphocyte count, and chest CT findings. In our patients, we detected no severe involvement of lungs in CT, which is discordant with the findings of a study by Danzi et al, who reported extensive bilateral parenchymal involvement [8]. The guidelines have recommended non-contrast CT for the diagnosis and follow-up of COVID-19. It would be advisable to order pulmonary CT angiography in patients with COVID-19 that are nonresponsive to routine medications.
To differentiate pulmonary infarction due to PTE from COVID-19 induced lung lesions in chest CT, serial CT could be of great value. We performed follow-up CT scanning for the 4 patients, which makes an evaluation of changes in lesions over time and the prediction of outcomes more feasible [9]. However, further original research is needed to determine clinical presentation, laboratory tests, and chest CT findings of patients with concurrent PTE and COVID-19.
Conclusion
With respect to the possible coexistence of COVID-19 and PTE, the diagnosis of acute PTE based on clinical and paraclinical findings could be a challenging issue. Patients may have no criteria of severe COVID-19. We suggest contrast pulmonary CT angiography for patients with COVID-19 if no contraindication exists. A comprehensive interpretation of all findings should be considered in these patients.
Declarations
Acknowledgements: The authors would like to thank all the staffs of Tehran Heart center.
Funding: This study was supported by Tehran University of Medical Science [grant number: 99-1-138-47404].
References
- Tang N, Bai H, Chen X, Gong J, Li D, Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost 2020; 18:1094-9.
- Wichmann D, Sperhake J-P, Lütgehetmann M, et al. Autopsy Findings and Venous Thromboembolism in Patients With COVID-19: A Prospective Cohort Study. Ann Intern Med 2020.
- Llitjos JF, Leclerc M, Chochois C, et al. High incidence of venous thromboembolic events in anticoagulated severe COVID‐19 patients. J Thromb Haemost 2020; 00:1–4.
- Poorhosseini H, Abbasi SH. The Tehran Heart Center. Eru Heart J. 2018; 3: 2695–2703.
- Peng Y, Meng K, Guan H, et al. Clinical characteristics and outcomes of 112 cardiovascular disease patients infected by 2019- nCoV. Zhonghua xin xue guan bing za zhi 2020; 48:E004.
- Kwong JC, Schwartz KL, Campitelli MA, et al. Acute myocardial infarction after laboratory-confirmed influenza infection. N Engl J Med. 2018; 378: 345-53.
- Rotzinger D, Beigelman-Aubry C, von Garnier C, Qanadli S. Pulmonary embolism in patients with COVID-19: Time to change the paradigm of computed tomography. Thrombosis research. 2020; 190: 58-59.
- Danzi GB, Loffi M, Galeazzi G, Gherbesi E. Acute pulmonary embolism and COVID-19 pneumonia: a random association? Eur Heart J. 2020; 41: 1858.
- Pan F, Ye T, Sun P, et al. Time course of lung changes on chest CT during recovery from 2019 novel coronavirus (COVID-19) pneumonia. Radiology 2020; 295: 715–721.