Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Acute spinal cord ischemic stroke; A rare but potentially devastating stroke emergency in young healthy adults
Liaquat Ali1; Ahmad Muhammad1,3; Islam Ahmed2; Khawaja Hassan Haroon1,3*
1 Department of Neurology, Hamad General Hospital, HMC, Doha, Qatar
2 Department of Radiology, Hamad General Hospital, HMC, Doha, Qatar
3 Weill Cornell Medicine, Doha, Qatar.
*Corresponding Author: Khawaja Hassan
Haroon
Hamad General Hospital, Neurology Dept HGH, al
Rayyan road, 3050, Doha, Qatar
Email: drkhharoon@yahoo.com
Received : Sep 09, 2021
Accepted : Oct 14, 2021
Published : Oct 21, 2021
Archived : www.jcimcr.org
Copyright : © Haroon KH (2021).
Abstract
Introduction: Acute spinal cord infarction represents 1% of all strokes. The neurological presentation of spinal cord infarction is defined by vascular territory i.e. anterior or posterior spinal arteries. A broad spectrum of diseases can cause spinal cord infarction, most common cause is surgical repair or diseases of the thoracoabdominal aorta. MRI of the spine shows hyperintensities on T2WI, restricted diffusion on DWI and vertebral body infarction adjacent to a cord signal abnormality. Poor prognostic factors for recovery are severe impairment at presentation, female sex, advanced age, and lack of improvement in the first 24 hours. We describe clinical presentation, radiological findings and 3-month outcome of four patients who presented with ischemic spinal cord infarction.
Methods: We retrospectively analysed our prospectively collected data and found four cases of spinal cord infarction for the period of May 2020 to April 2021. Total of 1,326 stroke patients (1125 i.e. 85% ischemic and 201 i.e. 15% haemorrhagic) were admitted in Hamad General Hospital. At our centre, spinal cord infarction accounts 0.30% of total stroke and 0.35% of ischemic strokes. We describe four young patients, presented with abrupt onset of focal neurologic deficit. Their MRI of the spine revealed hyperintensity signals in T2WI, restricted diffusion on DWI of spinal cord. Two patients had vertebral artery occlusion or dissection. One patient was discharged home and three patients underwent multidisciplinary rehabilitation program.
Discussion: Spinal cord infarction may present with acute paraparesis or quadriparesis with devastating consequences. Blood flow to the spinal cord is influenced by perfusion pressure. The most common cause of spinal cord infarction in this case series was vertebral artery dissection or occlusion. Our case series suggests early recognition of spinal cord stroke, appropriate investigations and early treatment with further rehabilitation could improve patient outcome.
Conclusion: Spinal cord infarction, a rare but devastating condition, represents 1% of all strokes but 0.3% of total strokes at our centre. Early recognition of spinal cord ischemia especially when presenting with abrupt onset focal neurological deficit, focus on underlying pathology with appropriate imaging and further investigations, secondary stroke prevention and rehabilitation are vital factors to improve neurological recovery. Future randomized trails are needed to establish the efficacy and safety of drug therapy including rt-PA in spinal cord infarction.
Abbreviations: ASA: Anterior spinal artery; PSA: Posterior spinal artery; MRI: Magnetic resonance imaging; MRA: Magnetic resonance arteriogram; DWI: Diffusion weighted imaging; ICP: Intra-cranial pressure.
Citation: Ali L, Muhammad A, Ahmed I, Haroon KH. Acute spinal cord ischemic stroke; A rare but potentially devastating stroke emergency in young healthy adults. J Clin Images Med Case Rep. 2021; 2(5): 1375.
Introduction
Spinal cord infraction, a rare but devastating condition, accounts for 1% of all strokes and 4 to 10% caused by vertebral artery dissection. A broad spectrum of diseases can cause spinal cord infarction, most common cause is surgical repair or diseases of the thoracoabdominal aorta. MRI of the spine would help to establish the diagnosis, exact location and extent of cord infarction.
We describe clinical presentation, radiological findings and 3-month outcome of four patients who presented with spinal cord ischemia.
Case presentations
We retrospectively identified and analysed following four cases of spinal cord stroke from prospectively collected data from May 2020 to April 2021. Spinal cord infarction represents 0.3% of all strokes at our centre.
Case I
A 41-year-old gentleman with no prior comorbidities, woke up in the morning with right sided weakness, spinning sensations, recurrent vomiting and severe headache. There was no history of fever, cough or difficulty in breathing. Neurological examination revealed that he was drowsy but arousable, he had right upper limb power 4/5 with dysmetria, and had gazeevoked nystagmus. Initial non-contrast CT of the head showed acute infarct of right cerebellum and medulla with associated oedema, mild mass effect, and small bilateral thalami infarcts. Patient was admitted to MICU for close neuro vitals monitoring and possible neurosurgical intervention.
MRI head showed acute infarct in the right cerebellar hemisphere and medulla oblongata with minimum mass effect at the fourth ventricle, tiny acute ischemic infract in bilateral thalami, left occipital and posterior temporal area. MRA of the head and neck were normal. Screening COVID-19 PCR test turned positive. He became short of breath, desaturated and found to have his right lung collapsed on Chest x-ray. He was intubated and treated as per COVID-19 treatment protocol. He was also given antiplatelet, statin and prophylactic LMWH.
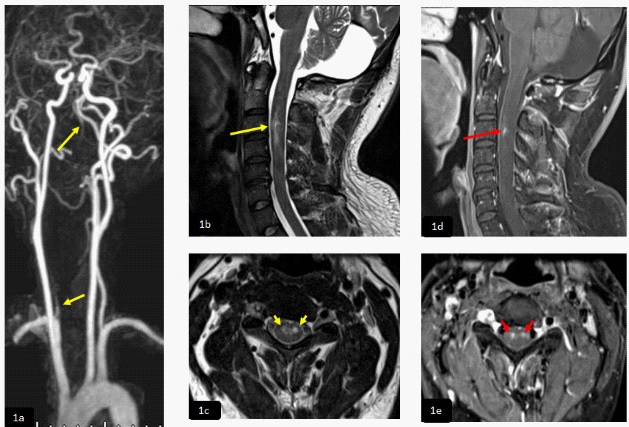
At day 9, while being considered for extubating, he developed both lower limbs weakness with power 0/5 with difficulty to ascertain exact sensory level and impaired pain and temperature sense. MRI of the cervicothoracic spine, sagittal DWI and ADC map (1a,1b) demonstrated long segment of diffusion restriction in the upper anterior dorsal cord extending from D1- D3. There was high signal intensity in sagittal and axial T2WI (1c,1d) proximal to infracted cord without diffusion restriction and with subtle patchy post contrast enhancement seen in axial T1W1 post contrast (1e) representing subacute infract (red arrow). Axial T1 (1f) revealed high signal intensity in the right vertebral artery denoting thrombosis (short red arrow). There was abnormal signal intensity and post contrast enhancement of the cerebellum due to subacute infraction seen in sagittal T1 post contrast (1g).
Later, patient was transferred to rehabilitation facility. After 3 months in rehabilitation, there were minimal improvement in his lower limbs leading to severe disability with modified Rankin Scale of 5 on discharge.
Case II
A 37-year-old gentleman was admitted to hospital with late presentation of sudden onset of right upper limb weakness which quickly progressed to his right lower limb over next few minutes. After few hours, he developed left sided weakness and numbness all over the body below his neck. A day before his presentation, he had neck message followed by mild neck pain. He did not have any medical conditions.
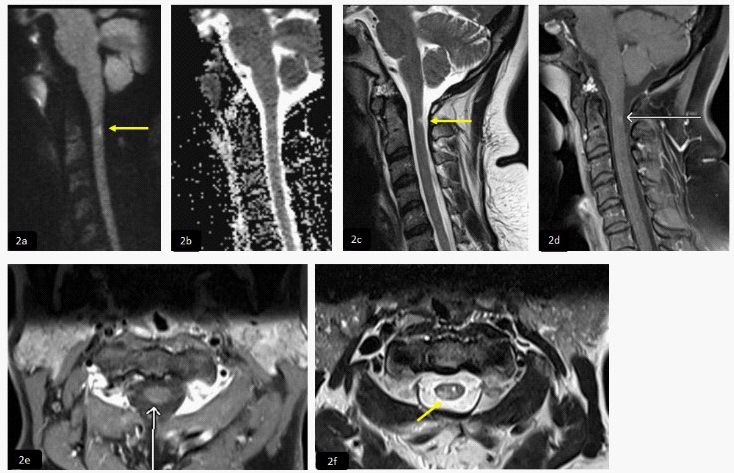
Neurological examination revealed that he was alert and oriented. Cranial nerves examination was normal, motor strength 1/5 on right side while 3/5 on left side and sensory level at C4 dermatome with loss of pain and temperature sense. MRI and MRA of the head were normal. 3D MRA of the neck (2a) showed dissection of the right vertebral artery just distal to its origin (yellow arrows). Sagittal and axial T2WI (2b,2c) and T1 post contrast (2d,2e) respectively, demonstrated intermedullary T2 bright signal intensity seen at the anterior aspect of the spinal cord involving the central grey matter predominantly ventral horns between C2-C4 levels (yellow arrows) and evidence of postcontrast enhancement (red arrows). These features are consistent with subacute cord infarction.
Patient underwent multidisciplinary rehabilitation program. After 3 months in rehabilitation centre, patient went back to home with residual deficit of right-side extremity strength of 3/5 while left side extremity of 4/5 and was able to walk using single crutch under supervision with mRS 4.
Case III
A 55-year-old right-handed gentleman with history of T2DM, essential HTN and heavy smoking, presented to Emergency Department in the evening with sudden onset of left lower limb weakness which progressed to right lower limb with difficulty in walking. He also had pain in both upper extremities since morning. On admission, neurological examination revealed bilateral lower limb weakness with left side weaker than right side distally with power of 4/5, sensory level at T4 dermatome with impaired pain and temperature sensations and equivocal planters bilaterally. Initial CT of the head, perfusion, angiogram, MRI and MRA of the head were all normal.
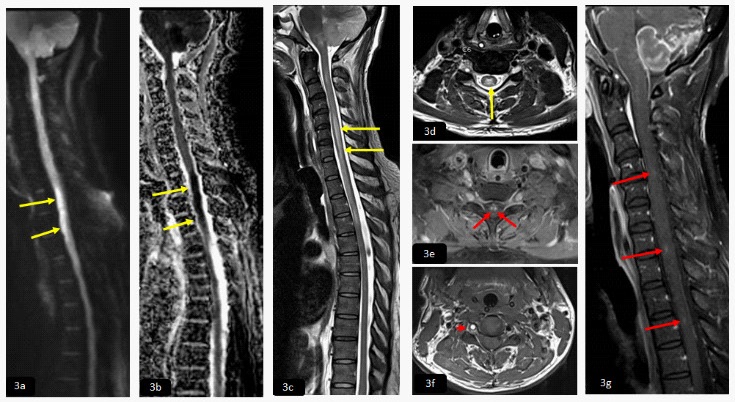
MRI of the cervicothoracic spine, sagittal DWI and ADC map (3a,3b) demonstrated focal area of diffusion restriction opposite to the level of C7. Sagittal and axial T2WI (3c,3d) showed abnormal bright signal intensity seen in the anterior aspect of the cervical cord, giving appearance of owl eye (short yellow arrows).
Patient was started on aspirin and statin for secondary prevention of stroke. His risk factors were addressed, and he underwent rehabilitation. At 3-month follow up, he showed significant improvement with mild weakness in both legs, walking independently with mRS 2.
Case IV
A 41-year-old right-handed lady with history of hypothyroidism went under varicose vein surgery under general anaesthesia. She was admitted to our unit as she noticed right-sided upper and lower extremity numbness and weakness, mainly in right hand while in recovery room.
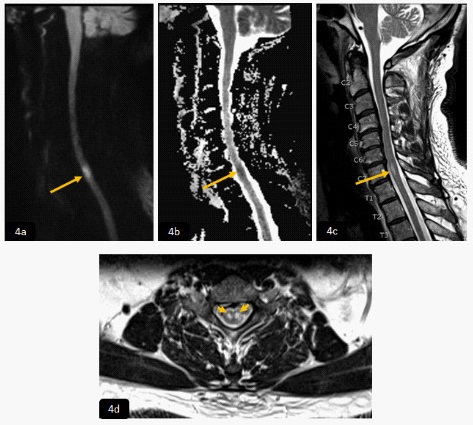
Examination showed right upper limb dysmetria with strength of 4+/5 upper extremity. Urgent CT of the head, perfusion and angiogram, MRI and MRA of the head performed, were all normal. MRI of the cervical spine showed focal intramedullary area of slightly high signal intensity in DWI (4a) and normal ADC map value (4b) in right aspect of the posterior column of the cervical cord at the level of C2 (yellow arrows), it demonstrated high signal intensity in T2WI and faint post contrast enhancement in sagittal and axial T2WI and T1WI post contrast (4c,4d) and (4e,4f) respectively, in keeping with subacute cord infarction.
During her stay in hospital, she made good recovery. On assessment at 3 months follow up visit, she still had subtle righthand grip weakness and tingling sensations with mRS 1. Follow up MRI of the head and spine after 3 months showed persistent T2WI cord hyperintensity at C2 level with focal area of established gliosis without enhancement.
All our four patients had Echocardiogram, 48-hour Holter, lupus anticoagulant and anti-cardiolipin antibodies which were normal. They were all treated with antiplatelet, statin and our 3rd patient had aggressive management of his conventional risk factors.
Discussion
Acute spinal cord stroke syndrome represents 5-8% of acute myelopathies [1,2]. It has been estimated that spinal cord infarction accounts for 1% of all strokes and 4% to 10% caused by vertebral dissection [3,4]. Though small but our series of spinal cord infarction accounts for 0.3% of all strokes at our centre. A spinal cord lesion may be suspected when there are bilateral motor and sensory signs or symptoms that do not involve the head or face [5] as seen in our first three patients. Patients typically present with acute paraparesis or quadriparesis, depending on the level of the spinal cord involved. The onset of spinal cord infarction may be abrupt as in all our cases, a significant proportion experience a decline over a few to several hours [6,7]. In one case series of 133 patients with spinal cord infarction, the time from symptom onset to nadir deficit was more than 4 hours in 44% of patients, and greater than 12 hours in 23%. Most patients (72%) had severe back or limb pain at symptom onset [8].
Three major vessels, one anterior spinal artery (ASA) and a pair of posterior spinal arteries (PSA), arising from the vertebral arteries in the neck supply the spinal cord. These are augmented along their course by greater number of radicular arteries [9,11,12]. The ASA supplies the anterior two-thirds of the spinal cord including anterior horns of the grey matter, spinothalamic and corticospinal tracts [10]. The thoracic spinal cord is particularly dependent on radicular contributions and may be the most vulnerable to infarction [11]. The PSAs supply the dorsal columns and the posterior horns. Two major spinal veins, one anterior and one posterior, drain into a series of radicular veins, then into intravertebral and paravertebral plexuses, and finally into the azygous and pelvic venous systems [13]. As spinal veins have no valves, Valsalva and increased intra-abdominal pressure can lead to increased venous pressure and decreased spinal cord perfusion [11].
Spinal blood flow is influenced by metabolic demand, which is highest in the grey matter and most abundant in the cervical and lumbar segments, responds to both hypoxemia and hypercapnia with increased blood flow [13,14]. Blood flow to the spinal cord is also influenced by perfusion pressure, which is the difference between mean arterial and intraspinal canal pressure. Through autoregulation, spinal blood flow is maintained at a constant level over a range of mean arterial pressures, however, there are upper and lower limits of systemic blood pressure beyond which autoregulation fails [15]. As a result, systemic hypotension or increased intraspinal canal pressure may decrease perfusion and put the cord at risk. Since the spinal cord exists in a fixed space, intraspinal canal pressure is sensitive to changes in the contents of the spinal canal and may rise significantly in pathological states.
A broad spectrum of diseases can cause spinal cord infarction [11]. The underlying mechanisms can be broadly categorized into diseases or procedures involving the thoracoabdominal aorta (aortic surgery, aortic dissection), Intrinsic arterial occlusion resulting from arteriosclerosis, vasculitis, infection, embolic occlusion, thrombosis, hypoperfusion and venous infarction. Our 1st case had severe COVID-19 disease with likely underlying hypercoagulable state which trigged the thrombosis and acute brain and spinal cord infarctions [26] complicated by severe sepsis and hypoxemia. 2nd case had neck message which triggered vertebral artery dissection leading to cervical cord infarction.
A large proportion of spontaneous spinal cord infarction does not have an identified aetiology, many of these are presumably secondary to atherothrombotic disease. Our 3rd case work-up was negative for cardioembolic disease. We presume, in view of his risk factors, that cord infarction could be secondary to atherosclerotic disease. The most common cause of spinal cord infarction is surgical repair of the thoracoabdominal aorta. Spinal cord infarction can also complicate other non-aortic surgeries, as well as episodes of profound systemic hypotension such as those occurring during cardiopulmonary arrest. Our 4th patient had varicose vein surgery under GA without evidence of hypotension during procedure. As her investigations including young stroke work up were normal, we presume the underlying mechanisms could be either kinking/spasm of spinal vessel leading to cervical cord infarction due to neck movements especially extension and flexion.
The neurological presentation of spinal cord infarction is largely defined by the vascular territory involved. The severity of neurological deficit may vary widely from minor weakness to paraplegia [16]. Majority of the spinal cord infarctions present with acute neurological deficits attributable to the Anterior Spinal Artery (ASA) territory. However, one-third of the patients may have less typical presentations including Posterior Spinal Artery (PSA), central cord infarct (often the result of severe hypotension), sulcal artery syndrome (resulting in a partial BrownSéquard syndrome) and complete cord transection syndromes [17].
An ASA infarct typically presents as bilateral loss of motor function, pain and temperature sensation as our 1st three patients had, with relative sparing of proprioception and vibratory sense below the level of the lesion [18]. Autonomic dysfunction such as hypotension, sexual and/or bowel and bladder dysfunction may be present [20]. Rostral cervical cord lesion will compromise the respiration and a very rostral ASA infarctions produce sensory loss in all modalities because of involvement of the medial lemniscus in the medulla. PSA infarction produces unilateral loss of proprioception and vibratory sense below the level of the injury and total anaesthesia at the level of the injury. Weakness is typically mild and transient as in our last case.
Venous infarction of the spinal cord has been association with spinal vascular malformations. These infarcts may be either haemorrhagic, presents with sudden onset of pain and flaccid paraparesis or non-haemorrhagic, presents with gradually progressive neurologic decline [19]. There are many other potential causes of acute myelopathy including compressive myelopathy (neoplasm, epidural or subdural hematoma, abscess), transverse myelitis, Guillain-Barré syndrome, acute dissection and rupture of descending aorta.
The diagnosis of spinal cord infarction is made in the setting of rapidly developing myelopathic deficits in the absence of another identifiable aetiology by history, examination and MRI spine [3]. MRI of the spine can be normal in the acute setting, thus repeating it few days later is recommended. MRI changes associated with spinal cord ischemia include hyperintensities on T2WI in published reports ranges from 45 to 73% depending in part on the scan's timing [19]. A restricted diffusion on DWI appears to be significantly more sensitive than hyperintensities on T2-WI [21], still DWI has shown incomplete sensitivity in spinal cord infraction given limitations of spatial resolution and susceptibility artefacts [8], compared to cerebral infarction. A vertebral body infarction adjacent to a cord signal abnormality on MRI is a specific indicator of ischemia and a useful confirmatory sign if present [22]. Our 1st and 3rd patients clearly had diffusion restriction lesions in the cervical cord. Our 4th patient had shown focal intramedullary area of slightly high signal intensity on DWI and normal ADC map value but hyperintensities on T2WI while our 2nd patient showed hyperintensities on T2WI.
Spinal cord infarction is a rare but often devastating condition. Patients with moderate to severe deficits resulting from a high thoracic or cervical cord infarct should be admitted to an intensive care unit with close monitoring of vital signs and neurological status. Autonomic dysreflexia characterized by episodic paroxysmal hypertension with headache, bradycardia, flushing, and sweating, is most often described after a traumatic spinal cord injury but may be a potential concern after spinal cord infarction involving the cervical and upper thoracic cord as well. The case fatality rate is reported between 10 and 20 % [23]. Patients presenting in the setting of cardiac arrest and acute aortic rupture or dissection, and those with high cervical lesions are at greatest risk of dying [24].
Among survivors, most make some improvement in functional deficits. Independent gait is achieved in 11 to 46 %, while 20 to 57% remain wheelchair bound [25]. Poor prognostic factors for recovery include severe impairment at presentation, female sex, advanced age, and lack of improvement in the first 24 hours [25]. Our 1st two patients had severe disabling deficit from onset and had poor functional outcome at 3-months. Our 3rd patient had disabling symptoms at onset due to paraparesis and his 3-month functional outcome was good. Our last patient had good outcome at 3-month as her initial deficit improved in the 1st 24 hours.
All our patients were investigated to identify underlying cause, treated with antiplatelets and stains, risk factors modification and rehabilitation at our dedicated centre. Treatment is generally supportive, though IV thrombolysis within 4.5 hour of onset is an option in appropriate clinical setting but its efficacy and safety are still unproven in spontaneous spinal cord infarction. Focus on maintenance of adequate spinal cord perfusion pressure, secondary stroke prevention and rehabilitation are vital to improve neurologic recovery.
Conclusion
Spinal cord infarction is a rare condition, represents 1% of all strokes, though it accounts 0.3% of total strokes at our centre. Early recognition of spinal cord ischemia especially when presenting with abrupt onset focal neurological deficit, immediate appropriate imaging and identifying the underlying cause will help to tailor the acute therapy. MRI of the spine helps in diagnosing spinal cord ischemia, its level as well as extent on DWI and T2WI, with further investigations focused to identify underlying pathology. Secondary stroke prevention with risk factors modification and rehabilitation are vital to improve neurological recovery. Future trials are needed to establish the efficacy and safety of drug therapy including rt-PA in spinal cord infarction.
Declarations
Acknowledgements: Protocol ID MRC-04-21-379 entitled “Acute Spinal Cord Ischemic Stroke; A Rare but devastating Stroke Emergency in young healthy adults” has been approved by Medical Research Council MRC.
Conflict of interest: There is no conflict of interest.
Funding disclosure: Nil
Author’s contribution: Dr Liaquat Ali: data collection, data analysis, manuscript writing & literature review. Dr Ahmad Muhammad: data collection and analysis, manuscript writing, editing, literature and critical review. Dr Islam Ahmed: Images review and selection. Dr Khawaja Hassan Haroon: data collection and analysis, manuscript writing, editing and critical review.
References
- Nedeltchev K, Loher TJ, Stepper F et-al. Long-term outcome of acute spinal cord ischemia syndrome. Stroke. 2004; 35: 560-5.
- Vargas MI, Gariani J, Sztajzel R et-al. Spinal cord ischemia: practical imaging tips, pearls, and pitfalls. AJNR Am J Neuroradiol. 2014; 36: 825 30.
- Novy J, Carruzzo A, Maeder P, Bogousslavsky J. Spinal cord ischemia: clinical and imaging patterns, pathogenesis, and outcomes in 27 patients. Arch Neurol 2006; 63: 1113.
- Cheng MY, Lyu RK, Chang YJ, et al. Spinal cord infarction in Chinese patients. Clinical features, risk factors, imaging and prognosis. Cerebrovasc Dis. 2008; 26: 502.
- baker Ab. Spinal Cord Localization. J Lancet 1965; 85: 269.
- Novy J, Carruzzo A, Maeder P, Bogousslavsky J. Spinal cord ischemia: clinical and imaging patterns, pathogenesis, and outcomes in 27 patients. Arch Neurol. 2006; 63: 1113.
- Cheng MY, Lyu RK, Chang YJ, et al. Spinal cord infarction in Chinese patients. Clinical features, risk factors, imaging and prognosis. Cerebrovasc Dis. 2008; 26: 502.
- Zalewski NL, Rabinstein AA, Krecke KN, et al. Characteristics of Spontaneous Spinal Cord Infarction and Proposed Diagnostic Criteria. JAMA Neurol. 2019; 76: 56.
- Prasad S, Price RS, Kranick SM, et al. Clinical reasoning: a 59-yearold woman with acute paraplegia. Neurology. 2007; 69: E41.
- Hurst RW. Spinal vascular disorders. In: Resonance Imaging of the Brain and Spine, 2nd ed, Atlas SW (Ed), Lippincott, Philadelphia. 2006. p.1387.
- Mohr JP, Benavente O, Barnett HJ. Spinal Cord Ischemia. In: Stroke Pathophysiology, Diagnosis, and Management, Barnett HJ, Mohr JP, Stein BM, Yatsu FM (Eds), Churchill Livingstone, Philadelphia 1998. p.423.
- Parent A. Blood Supply of the Central Nervous System. In: Carpenter’s Human Neuroanatomy, 9th ed, Williams and Wilkins. 1996; 93.
- Cheshire WP, Santos CC, Massey EW, Howard JF Jr. Spinal cord infarction: Etiology and outcome. Neurology. 1996; 47: 321.
- Marcus ML, Heistad DD, Ehrhardt JC, Abboud FM. Regulation of total and regional spinal cord blood flow. Circ Res. 1977; 41: 128.
- Hickey R, Albin MS, Bunegin L, Gelineau J. Autoregulation of spinal cord blood flow: Is the cord a microcosm of the brain? Stroke. 1986; 17: 1183.
- Hsu CY, Cheng CY, Lee JD, et al. Clinical features and outcomes of spinal cord infarction following vertebral artery dissection: a systematic review of the literature. Neurol Res. 2013; 35: 676.
- Weidauer S, Nichtweiss M, Lanfermann H, Zanella FE. Spinal cord infarction: MR imaging and clinical features in 16 cases. Neuroradiology. 2002; 44: 851.
- Foo D, Rossier AB. Anterior spinal artery syndrome and its natural history. Paraplegia. 1983; 21: 1.
- Mohr JP, Benavente O, Barnett HJ. Spinal Cord Ischemia. In: Stroke Pathophysiology, Diagnosis, and Management, Barnett HJ, Mohr JP, Stein BM, Yatsu FM (Eds), Churchill Livingstone, Philadelphia 1998; 423.
- Cheung AT, Weiss SJ, McGarvey ML, et al. Interventions for reversing delayed-onset postoperative paraplegia after thoracic aortic reconstruction. Ann Thorac Surg. 2002; 74: 413.
- Thurnher MM, Bammer R. Diffusion-weighted MR imaging (DWI) in spinal cord ischemia. Neuroradiology. 2006; 48: 795.
- Faig J, Busse O, Salbeck R. Vertebral body infarction as a confirmatory sign of spinal cord ischemic stroke: Report of three cases and review of the literature. Stroke. 1998; 29: 239.
- Cheshire WP, Santos CC, Massey EW, Howard JF Jr. Spinal cord infarction: etiology and outcome. Neurology. 1996; 47: 321.
- Latoś T, Markiewicz R, Gaszczyk G. [Spirographic studies and measurement of airflow resistance in evaluation of bronchial hyperreactivity in children with asthma]. Pneumonol Pol. 1990; 58: 107.
- Nedeltchev K, Loher TJ, Stepper F, et al. Long-term outcome of acute spinal cord ischemia syndrome. Stroke. 2004; 35: 560.
- Haroon K, H, Muhammad A, Hussain S, Patro S, N: COVID-19 Related Cerebrovascular Thromboembolic Complications in Three Young Patients. Case Rep Neurol. 2020; 12: 321-328.