Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Second primary nasopharyngeal carcinoma after high - dose chemotherapy and autologous bone marrow transplantation due to Hodgkin’s lymphoma: Сlinical case with literature overview
Vaska Vassileva; Viktor Petrov; Lena Marinova*
Medical Oncology Clinic, Radiation and Metabolic Brachytherapy Compartment, UMHAT “Queen Joanna” Sofia, Bulgaria.
*Corresponding Author: Lena Marinova
Medical Oncology Clinic, Radiation and Metabolic
Brachytherapy Compartment, UMHAT “Queen
Joanna” Sofia, Bulgaria.
Email: rad_marinova@abv.bg
Received : Sep 14, 2021
Accepted : Oct 18, 2021
Published : Oct 25, 2021
Archived : www.jcimcr.org
Copyright : © Marinova L (2021).
Abstract
A second primary nasopharyngeal carcinoma diagnosed after Hodgkin’s Lymphoma is a rare disease.
We present a 47-year old patient with nasopharyngeal carcinoma one year after high-dose chemotherapy and 8 months after autologous bone marrow transplantation due to Hodgkin’s Lymphoma with achieved clinical and hemopoetic remission. The diagnosis of the second primary neoplasm was made after nasopharyngoscopy with biopsy, MRI and PET/CT. The patient was positive for Epstein-Barr viral infection. Intensity-modulated radiotherapy combined with chemotherapy was performed using VMAT technique. In the primary nasopharyngeal tumor was achieved total dose of 70 Gy with 2.12 Gy per fraction while in the regional lymphatic basin (levels IB-V) with the retropharyngeal lymph nodes bilaterally is achieved a total dose of 59.4 Gy with 1.8 Gy per fraction. In the discussion the pathogenetic connection between the Epstein-Barr viral infection and the debut of nasopharyngeal carcinoma is focused on.
The follow-up of patients who have undergone chemo- and/or radiotherapy due to Hodgkin’s Lymphoma is importance for the early diagnosis of a second malignant tumor and its proper treatment.
Keywords: nasopharyngeal carcinoma; epstein-barr virus; hodgkin’s lymphoma; high-dose chemotherapy; radiotherapy.
Citation: Vassileva V, Petrov V, Marinova L. Second primary nasopharyngeal carcinoma after high - dose chemotherapy and autologous bone marrow transplantation due to Hodgkin’s lymphoma: Сlinical case with literature overview. J Clin Images Med Case Rep. 2021; 2(5): 1381.
Introduction
Nasopharyngeal Carcinoma (NFC) is a relatively rare disease with an annual incidence of less than 1 in every 100,000 people [1]. NFC is an endemic neoplasm in southern China, where the incidence reaches 21 per 100,000 population [2]. The rarest new morbidity has been reported in northern Europe and the United States [3]. There are two age peaks 15-25 years and 50-60 years [4]. About half of all patients in the United States are under the age of 55. In Bulgaria, the incidence of NFCs in 2017 was 0.9 and the mortality rate was 0.4 per 100 thousand people [5]. We present a rare clinical case of a second primary NFC, diagnosed after one year of high-dose chemotherapy (Ch) and Autologous Bone Marrow Transplantation (ABMT) due to Hodgkin’s lymphoma (HL).
Clinical case
We present a 47-year-old man, diagnosed with Hodgkin’s lymphoma (stage II, histological variant nodular sclerosis) in July 2019. Until the month. September 2020, the patient underwent high-dose Ch with a duration of 12 courses (8 courses under the BEACOPP scheme, followed by 4 courses under the BeGeV scheme. Autologous Bone Marrow Transplantation (ABMT) was performed in October 2019. On PET / CT in June 2020, a tumor formation of 26/28 mm in the posterior wall of the nasopharynx with high metabolic activity, SUVmax-24.1. / 23 mm was found. Which protrudes slightly towards the lumen of the nasopharynx and circularly engages the two Eustachian tubes, more pronounced on the left.
For the purpose of clinical and diagnostic clarification, the patient was hospitalized in a Head and Neck Clinic, where nasopharyngoscopy with biopsy was performed. Histological result (July 21, 2021) - non-keratinizing undifferentiated nasopharyngeal carcinoma, type 2B according to the WHO (SchminkeRegaud lymphoepithelioma). The staging is T3N2cM0, clinical stage III. From paraclinical studies: HGB-127 g / l; HCT-0.42 L / L; RBC-5.09 T / l; WBC-7.05 G / l; PLT-239.30 G / l; Glucose-5.69 mmol / l; Urea 5.5 mmol / l; Creatinine-74 umol / l; ASAT-14 U/l; ALAT- 16 U/l. The patient was positive for Epstein-Barr virus infection. On 09.08.2021 the patient was hospitalized in the Department of Radiation Therapy and Metabolic Brachytherapy of UMHAT “Queen Joanna”.
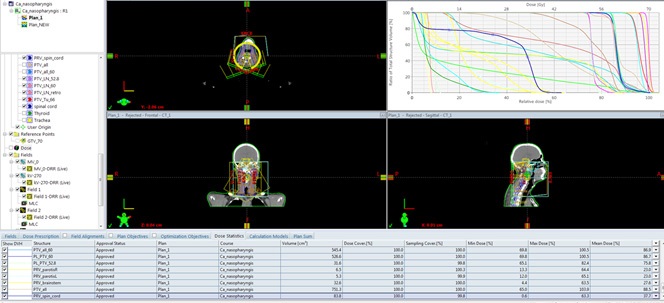
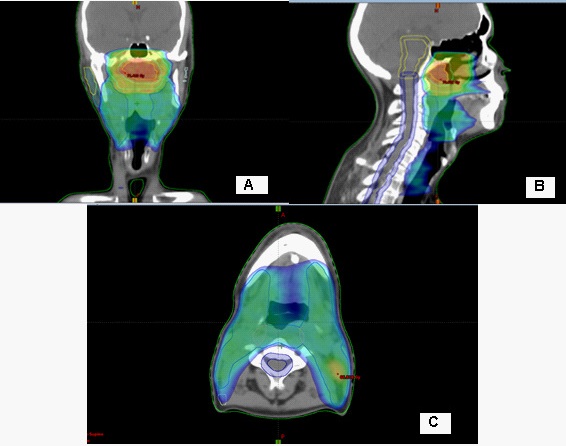
Intensively modulated radiotherapy (IML) with VMAT technique, combined with Ch on the 1st day of Cisplatin 80 mg i.v. and on the third-fourth day 750 mg 5-Fluorouracil i.v. was performed. In the primary tumor, total dose (TD) 70 Gy at Daily Dose (DD) 2.12 Gy, and in the regional cervical lymphatic nodes bilaterally to TD-59.4 Gy at DD-1.8 Gy, conducted in 33 fractions were realized (Figure 1, 2). The patient tolerated the combined radio-chemotherapy very well.
Discussion
NFC is diagnosed twice as often in men than in women. Early NFC has no specific symptoms. In advanced disease, symptoms include cervical lymphadenopathy, nasal breathing difficulties, shooting pains in the ears. The clinical stage of the disease is a significant prognostic factor [3]. There are mainly three types of nasopharyngeal carcinomas according to the WHO classification (type I - keratinizing squamous cell carcinoma; type II - non-keratinizing differentiated squamous cell carcinoma; type III - non-keratinizing undifferentiated squamous cell carcinoma [6-8].
The treatment approach for the three types of carcinomas is the same, with the difference that the non-keratinizing ones (type II and type III) are more radiation- and chemosensitive [9]. Type I has a better prognosis because it is less common with involved lymph nodes, and type III is less favorable due to the low degree of differentiation of tumor cells and the more frequent distant metastasis [10].
In some NFCs, microscopic pathohistological analysis reveals the presence of cells of the immune system - lymphocytes. The term 'lymphoepithelioma' is often used for undifferentiated nasopharyngeal carcinomas, in which a number of multiple lymphocytes between tumor cells in the microscopic field were detected [11].
The incidence of Epstein-Barr virus (EBV) positive patients is higher in non-keratinizing and / or poorly differentiated NFCs than in keratinizing carcinomas [12]. The causal relationship of EBV to epithelial cell tumors, more specifically to NFC and EBV-positive gastric cancer (EBV-GC), is not clear enough. It is currently thought to be due to latent EBV infection, which is the cause of precancerous genetic changes [13]. This genetic defects causes uncontrolled cell division and causes carcinogenesis. There are several well-described EBV latent models reported during the different stages of the viral life cycle, that are observed in different malignancies [14,15]. Numerous genetic changes have been found in NFCs, with frequent deletions of parts of chromosomes 3p, 9p, 11q, 13q and 14q and specific genetic changes in chromosomes 3p (Rassf1a, RaRβ2) and 9p (P16, P15, P14, DAP kinase) [16]. It is clear that deletion mapping, genomic sequencing, comparative genomic hybridization, or global gene expression profiling alone would not have easily identified candidate tumor suppressor genes (TSGs) from the huge array of lost regions or genes observed in nasopharyngeal carcinoma [17]. In squamous cell carcinomas of the head and neck, factors directly related to increased tumor radiation and chemosensitivity have been demonstrated, including positive human papilloma virus, high Ki-67 and no P53 mutation [18,19]. Self-radiotherapy (RT) is associated with disease-free 5-year survival of 95-100% in early NFC T1a-T2N0M0, and in locally advanced stage III-IV disease, combined chemo-radiotherapy (ChRT) is required [7]. For an objective and accurate assessment of local tumor control after combined ChRT of NFC, Moonkyoo Kong et al./2017 recommend waiting a longer period (>4 months) [20]. In the presented clinical case, due to locally advanced disease, we performed combined radiochemotherapy to high OOD 70 Gy in the tumor formation, which the patient tolerated very well, despite the previous 12 cycles of Ch (Figure 1,2).
Hodgkin's lymphoma (LH) or lymphogranulomatosis is a malignant disease of the lymph tissue. In the present clinical case, a differential diagnosis with primary nasopharyngeal LH is required, in which negative immunohistochemical expression to CD15, CD20 and CD3 is demonstrated, but positive to CD30 and epithelial membrane antigen (EMA) [21]. The reason for the late lethal outcome of Hodgkin's lymphoma after ABMT is recurrence of the disease in 56%, second malignancies in 25%, cardiac toxicity in 6%, pulmonary complications in 5%, and other causes - 13.5% [21].
From the study of Amit Sud et al / 2017, patients who survived Hodgkin's lymphoma were 2.4 times more likely to develop a second primary epithelial or mesenchymal neoplasm than the general population of the same age. The risk of secondary carcinogenesis was 1.3 times higher than those treated for early carcinoma (p <.001), 3.3 times higher for lung cancer, 2 times higher for colorectal cancer, and 1.8 times higher. times higher for breast cancer [23]. The presented clinical case does not concern secondary carcinogenesis after high-dose Ch, as the time period between the two neoplasms is extremely short. It is most likely carcinogenesis from latent EBV infection, which causes uncontrolled cell division of the nasopharyngeal epithelium after an extremely weakened immune response from the impending prolonged high-dose Ch.
Conclusion
Nasopharyngeal Carcinoma (NFC) is a relatively rare disease. It is extremely rare as a second primary neoplasm after longterm polychemotherapy and ABMT due to Hodgkin's lymphoma. In the presented clinical case, it is not a secondary carcinogenesis after high-dose Ch, as the time period between the two neoplasms is extremely short. Follow-up of patients who have undergone chemo and /or radiotherapy for Hodgkin's lymphoma is essential for the early diagnosis of a second malignancy and its adequate treatment. Following an extremely weakened immune response from impending Ch, the most likely cause of the second primary NFC is latent EBV infection, which causes uncontrolled cell division of the nasopharyngeal epithelium.
References
- Z Zahariev T. Hadjieva National standard for radiotherapy of nasopharyngeal carcinoma. Adopted by consensus of HLB, Sunny Beach. 2009.
- Neda Mahdavifar, Mahshid Ghoncheh, Abdollah Mohammadian-Hafshejani, Bahman Khosravi and Hamid Salehiniyae, Epidemiology and Inequality in the Incidence and Mortality of Nasopharynx Cancer in Asia. Osong Public Health Res Perspect. 2016; 7: 360–372
- Jiade J. Lu. Lin Kong and Nancy Lee.1 Decision Making in Radiation Oncology. 2011.
- Gautam Prasad, James Rambert, Erick K. Hansen and Sue S. Yom. Handbook of Evidence based Radiation Oncology. Chapter 5. Nasopharyngeal Cancer. 2010. Page 99.
- Zdravka Valerianova and Associates. Bulgarian National Rabban Register, Uniform, Tom XXVI. 2020.
- Caponigro F, Longo F, Ionna F, Perri F. Treatment approaches to nasopharyngeal carcinoma: A review. Anticancer Drugs. 2010; 21: 471–477.
- Perri F, Bosso D, Buonerba C, Lorenzo GD, Scarpati GD. Locally advanced nasopharyngeal carcinoma: Current and emerging treatment strategies. World J Clin Oncol. 2011; 2: 377–383.
- Yuting Zhan and Songqing Fan. Multiple Mechanisms Involving in Radioresistance of Nasopharyngeal Carcinoma. J Cancer. 2020; 11: 4193–4204.
- Qi Zeng, Yi-min Lu, Jun Liu. Inhibition of ZIP4 reverses epithelialto-mesenchymal transition and enhances the radiosensitivity in human nasopharyngeal carcinoma cells. Cell Death & Disease. 2019; 10.
- Gautam Prasad, James Rambert, Erick K. Hansen and Sue S. Yom. Handbook of Evidence based Radiation Oncology. Chapter 5. Nasopharyngeal Cancer. 2010. Page 100.
- Kwok Wai Lo, Ka Fai To and Dolly P. Huang. Focus on nasopharyngeal carcinoma. Cancer Cell. 2004; 5.
- Peter M. Som, Hugh D. Curtin. St Louis. Head and neck imaging. Combined chemo-radiotherapy in locally advanced nasopharyngeal carcinomas. World J Clin Oncol. 2013; 4: 47–51.
- Lawrence S. Young and Christopher W. Dawson. Epstein-Barr virus and nasopharyngeal carcinoma. Chin J Cancer. 2014; 33: 581–590.
- Young LS, Rickinson AB. Epstein-Barr virus: 40 years on. Nat Rev Cancer. 2004; 4: 757–768.
- Rickinson AB, Kieff E. Epstein-Barr Virus. In: Knipe DM, Howley PM, editors. Fields Virology. Philadelphia: Lippincott Williams and Wilkins; 2001; 2575–2627.
- Lo KW, Chung GTY, To KF. Deciphering the molecular genetic basis of NPC through molecular, cytogenetic, and epigenetic approaches. Sem Cancer Biol. 2012; 22: 79–86.
- Lung HL, Cheung AKL, Ko JMY, et al. Deciphering the molecular genetic basis of NPC through functional approaches. Sem Cancer Biol. 2012; 22: 87–95.
- Lavertu P, Adelstein DJ, Myles J, Secic M. P53 and Ki-67 as outcome predictors for advanced squamous cell cancers of the head and neck treated with chemoradiotherapy. Laryngoscope; 2001; 111: 1878–1892.
- Lassen P, Eriksen JG, Hamilton-Dutoit S, Tramm T, Alsner J, Overgaard J. Effect of HPV-associated p16INK4A expression on response to radiotherapy and survival in squamous cell carcinoma of the head and neck. J Clin Oncol. 2009; 27: 1992–1998.
- Moonkyoo Kong, Seong Eon Hong. Tumor regression patterns by follow-up duration in patients with nasopharyngeal carcinoma treated with concurrent chemoradiotherapy. Journal of Radiation Research. 2017; 58: 232–237,
- Youssef Bensouda, Kawtar El Hassani, Nabil Ismaili,et al. Primary nasopharyngeal Hodgkin’s disease: case report and literature review. Journal of Medical Case Reports. 2010; 4: 116 .
- Bhatia S, Robison LL, Francisco L, et al. Late mortality in survivors of autologous hematopoietic-cell transplantation: report from the Bone Marrow Transplant Survivor Study. Blood. 2005; 105: 4215-22.
- Amit Sud, Hauke Thomsen, Kristina Sundquist, Richard S. Houlston, and Kari Hemminki. Risk of Second Cancer in Hodgkin Lymphoma Survivors and Inflence of Family History. J Clin Oncol. 2017; 35: 1584-1590.