Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
ITP revealing a SMM: A case report and review of the literature
Rim Rakez1*; Areej Chefaii2; Rym Hadhri3; Mouna Bahrini Sassi4; Sarra Boukhriss1; Mohamed Adnene Laatiri5
1 Assistant in Hematology, Hematology Department, Fattouma Bourguiba Hospital of Monastir, Tunisia.
2 Hematology Resident, Hematology Department, Fattouma Bourguiba Hospital of Monastir, Tunisia
3 Associate Professor, Anatomopathology, Fattouma Bourguiba Hospital of Monastir, Tunisia.
4 Associate Professor, Biological Hematology, Fattouma Bourguiba Hospital of Monastir, Tunisia.
5 Head of the Department Of Hematology at Fattouma Bourguiba Hospital of Monastir, Tunisia.
*Corresponding Author: Rim Rakez
Hematology Department, Fattouma Bourguiba Hospital
of Monastir, Tunisia
Email: rimrakez@yahoo.fr
Received : Sep 16, 2021
Accepted : Oct 25, 2021
Published : Oct 29, 2021
Archived : www.jcimcr.org
Copyright : © Rakez R (2021).
Abstract
Immune thrombocytopenic purpura is an autoimmune disorder retained after elimination of other causes of low platelets’ rate. It is mostly seen with B cell lymphoproliferative disorders. Immune thrombocytopenic purpura’s association with plasma cell neoplasms is possible but extremely rare. Although several pathophysiological mechanisms have been proposed, the causal link between these two conditions is not yet clearly understood. Therapeutic management is not standardized and depends mainly on the type of gammopathy and the chronology of onset of immune thrombocytopenic purpura compared to multiple myeloma. Our case is about an 81-year-old male diagnosed with concurrent smoldering multiple myeloma and immune thrombocytopenic purpura who was started on steroids without anti-neoplastic therapy for multiple myeloma with partial platelet response. We also review the few reported cases of simultanious immune thrombocytopenic purpura and smoldering multiple myelom or multiple myeloma.
Keywords: ITP; thrombocytopenia; smoldering multiple myeloma; corticosteroids.
Abbreviations: SMM: Smoldering Multiple Myeloma; MDE: Myeloma Defining Events; MM: Multiple Myeloma; ITP: Immune Thrombocytopenic Purpura; Ivig: Intravenous Immunoglobulin.
Citation: Rakez R, Chefaii A, Hadhri R, Sassi MB, Boukhriss S, et al. ITP revealing a SMM: A case report and review of the literature. J Clin Images Med Case Rep. 2021; 2(5): 1392.
Introduction
Smoldering Multiple Myeloma (SMM) is a pre-malignant asymptomatic disease defined by an abnormal number of clonal bone marrow plasma cells (≥ 10 %) and/or a serum monoclonal protein (M-protein) ≥ 3 g/dl with absence of both CRAB criteria and myeloma defining events (MDE) [1]. This condition therefore involves a high risk of progression to Multiple Myeloma (MM).
Several studies linked MM to diverse autoimmune disorders. However, only few cases reported the association of Immune Thrombocytopenic Purpura (ITP) with MM even less with SMM since only 4 cases of this uncommon association were described up to now.
Here we present a case of an ITP leading to SMM diagnosis in an 81-year-old patient who responded to first-line treatment by steroids. We also review the cases of simultaneous diagnosis of ITP and SMM/MM reported in the literature and discuss the epidemiological and clinical characteristics of its clinical presentation in addition to the therapeutic implications of this unusual association.
Case presentation
An 81-year-old male, with no past history was referred to our outpatient department because of the incidental discovery of an isolated thrombocytopenia during a routine check-up.
History was negative for hematological or autoimmune symptoms. Besides, no reported fever, weight loss, anorexia or recent drug intake were mentioned by the patient.
The physical exam was unremarkable for any petechiae, bruises or bleeding. There was no palpable spleen or evidence of lymphadenopathy.
Blood investigations revealed a normal haemoglobin rate of 12.5 g/dl, normal white blood cells count of 5910 /mm3 and marked thrombocytopenia of 19 000/mm3 validated by the peripheral smear count showing no other abnormalities.
The coagulation status was within normal limits. Blood chemistries were normal: creatinine: 99 umol/l, albumin: 46.6 g/dl, total protein: 67g/l, calcium: 2.5 umol/l, lactate dehydrogenase: 219 UI/l, vitamin B12: 312 pg/ml (200 – 500 pg/dL) and folate: 8 ug/l (5-15 ug/l). Viral markers (anti-HCV antibody, antiHBV antibody and anti-HIV antibody), anti-thyroid antibody and anti-nuclear antibody were negative. HP serology was negative.
Further workup regarding the thrombocytopenia revealed, on serum protein electrophoresis, the presence of a monoclonal protein peak at a concentration of 1.7 g/l, identified as IgG Lambda by immunofixation electrophoresis. The urine protein electrophoresis was unremarkable. Beta 2 miroglobulin was within normal range (1.4 mg/L (0.85 – 1.62 mg/l).
Abdominal ultrasound confirmed the absence of splenomegaly
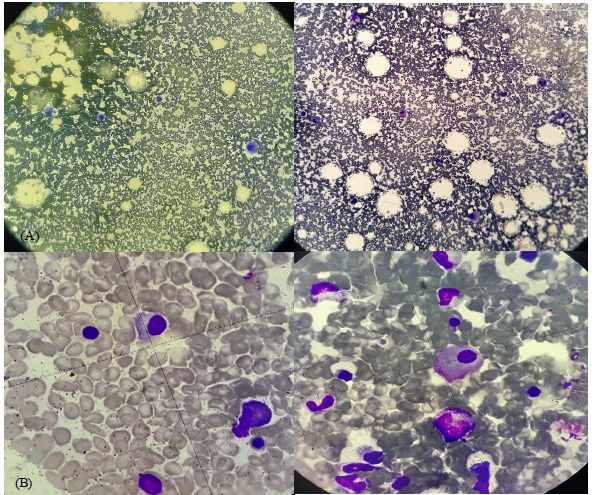
Bone marrow aspirate (Figure 1) showed 10% plasma cells of total marrow nucleated ones with increased megakaryocytes in all stages of maturation. Cytogenetic and Fish analysis for multiple myeloma was unrevealing.
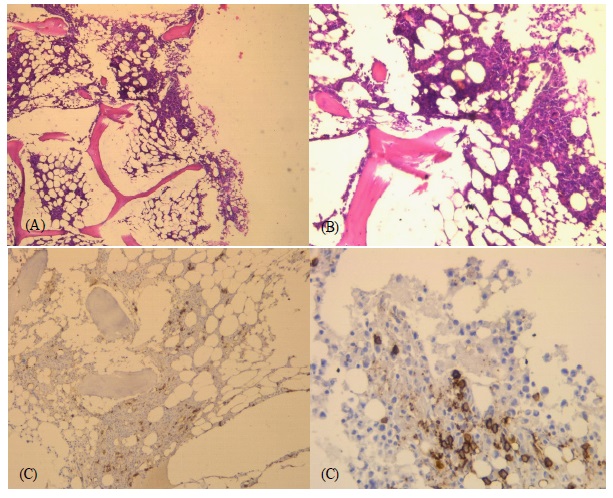
Bone marrow biopsy (Figure 2) showed normocellular marrow with increased plasma cells positive for CD138.
Medullar RMI was negative for any osteolytic bone lesion or medullar compression.
Hence, the diagnosis of concurrent SMM and ITP was made due to the association of isolated thrombocytopenia with no secondary causes, increased megakaryocytes on bone marrow aspiration meeting the definition of SMM described below.
The patient was started on oral steroid therapy (prednisolone 1 mg/kg/day) for 15 days, with a partial and transient increase in his platelet count (85 000/mm3 ). By steroids’ tapering the platelet count fell to 44 000/mm3 and remained stable one year after the diagnosis with no hemorrhagic syndrome.
Discussion
Thrombocytopenia is commonly associated with MM, either secondary to replacement of normal hematopoiesis by clonal plasma cells, or therapy-related due to marrow suppression [2,3]. In case of SMM, such thrombopenia is not observed since the tumor burden is not important and usually there is no need for treatment in this situation [4].
While ITP is more commonly seen with B cell lymphoproliferative disorders especially chronic lymphocytic leukemia followed by Hodgkin's and non-Hodgkin's lymphomas [5], it is rarely observed with plasma cell neoplasms [6]. In fact, some studies underligned the predisposition of MM/SMM patients to develop auto-immune disorders such as autoimmune hematologic, neurological and rheumatologic conditions [7]. But coexisting ITP and MM or SMM is an unusual association since only 13 cases were reported in the literature up to now: 4 patients with ITP preceeding MM diagnosis [3,8,9], 5 cases of MM preceeding ITP [10-12] whereas the simultaneous diagnosis of both conditions appears to be even more unexpected: 2 cases of ITP and SMM [6,13] and 2 cases of ITP and MM [9,14] (Table 1).
Although poorly understood, several mechanisms of ITP and SMM/MM association have been suggested. The most adopted ones include immune deregulation responsible on one hand, for chronic antigenic stimulation and therefore an increase in peripheral platelet destruction and on the other hand, for an impaired platelet production due to intrinsic platelet defects [3,4,15]. For some authors, this immne stimulation may introduce pro-oncogenic mutations in rapidly dividing cells such as plasma cells leading to SMM and MM [7]. The paraprotein associated with such monoclonal gammapathies was also reported to be implicated by acting as a reactive antibody against platelets [13]. This last hypothesis suggests a possible correlation between the paraprotein concentration and the depth of the thrombocytopenia, and therefore the severity of the haemorrhagic syndrome.
Only 4 cases of associated MM/SMM and ITP diagnosed concurrently have been reported in the literature so far (Table 1). Only the patient in case 4 didn’t present with hemorrhagic syndrome which may indicate that ITP associated with MM does not seem to be more predisposing to this clinical feature than when seen with SMM.
Table 1: Cases of SMM/MM with concurrent ITP.
Case |
Reference |
Age (Y.O) / Gender |
Gammapathy type |
Circumstances of discovery |
Platelet count (10⁹/L) |
Hemorrhagic syndrome |
Immuno- |
Paraprotein concentration (g/dL) |
Plasma cells (bone marrow) |
CRAB signs |
ITP management : 1st line treatment |
Evolution of platelet count |
ITP management : 2nd line treatment |
SMM/ MM management |
ITP response to chemotherapy |
1 |
Siniscalchi et al (8) |
67 / M |
SMM |
Gastrointestinal bleeding |
3 |
Yes Gastrointestinal |
IgG Kappa |
2.6 |
30% |
None |
Steroids then IVIg |
Transient increase |
Rituximab |
None |
- |
2 |
Ankur Jain (6) |
57 / F |
SMM |
Generalized bluish spots over the body |
2 |
Yes Cutaneous |
IgA Lambda |
Unspecified |
20% |
None |
Steroids |
Rapid increase (3 |
- |
None |
- |
3 |
Gupta et al (9) |
49 / M |
MM |
Severe epistaxis |
21 |
Yes |
IgG Lambda |
4.0 |
20% |
Anemia |
IVIg |
Increase to 100 G/L |
- |
VAD |
Thrombocytopenia on several occasions |
4 |
Humaira Sarfraz et al |
60 / M |
MM |
Generalised weakness and weight loss |
1 |
No |
IgG Lambda |
0.8 |
30% |
Anemia |
IVIg + steroids |
No improve-ment |
- |
Cyclophosphamid + bortezomib + Dexamethasone |
Platelet count increesed to 171 k/μL |
5 |
Our case |
81 / M |
SMM |
Incidental discovery, |
19 |
No |
IgG Lambda |
1.7 |
10% |
None |
Steroids |
Increase to 85G/L |
- |
None |
- |
In this case, and so it is for the reported ones, the diagnosis of ITP was established based on the isolated character of the thrombocytopenia, the normal blood smear and the exclusion of other conditions known to cause secondary thrombocytopenia [16,17]. Two of the patients reported (cases 3 and 4) fullfiled criteria for active MM at the time of ITP diagnosis by having 10 % or more plasma cells in the bone marrow with at least a CRAB sign. Whereas, our patient, same for the patients reported in cases 1 and 2, was asymptomatic and did not have any of the CRAB signs nor the MM defining events. They both only fulfill criteria for peripheral blood (serum M-protein peak) and bone marrow-based diagnosis of MM. This condition defines SMM fortuitously discovered as part of the etiological investigation of thrombocytopenia.
Although the bone marrow biopsy does not appear in the initial etiological assessment of MM or SMM, it finds all its interest here given the association of this gammapathy with ITP in order to rule out a central cause of thrombocytopenia.
Thereby, immune destruction of platelets should be considered whenever unexplained thrombocytopenia occurs in patients with MM or SMM.
All patients except case 2 were shown to have IgG paraprotein type with concentrations ranging from 0.8 to 4.18 g/dl. Due to the rarety of this association, we could not significantly relate the nature of the paraprotein to an ITP predisposition.
Regarding the lastest ASH guidelines [18], the initial conventional treatment options (steroids and intravenous immunoglobulin (IVIg)) should be opted in asymptomatic patients with ITP if platelet’s rate < 20000/mm3.
This strategy seems to be sufficient to manage such immune dysregulation in SMM and MM cases. It should be noted that for second-line treatment, the non-surgical options (Rituximab and TPO-r mimetics) are preferable to splenectomy, firstly given the risk of immunosuppression and sepsis in SMM and MM patients and secondly considering their safety profile and their high response rates in these cases [13].
In addition to that, for some other authors, the therapeutic decision problem arises in the cases of associated ITP and SMM. The question, in this situation, is whether to establish an early intervention for better clinical results to avoid evolution to an active MM or to opt for a delayed intervention to reduce treatment-related complications [4,19]. Regarding the classifications proposed to predict the risk of progression to an agressive MM [1], ITP doesn’t appear to be part of MM treatment decisions. Based on that, the patient in our case had a low-risk of progression to MM and was then only given ITP treatment by prednisolone therapy allowing the increase in platelet count which is one more argument for the co-existence of ITP at SMM diagnosis.
Conclusion
Although the association of SMM and ITP is rare, it is necessary to evoke ITP in front of an isolated thrombocytopenia associated with this type of gammopathy. The diagnosis remains simple. When needed, the first-line treatment is based on corticosteroid therapy.
Ethics declarations
Conflicts of interest: None.
Consent for publication: The authors state that an informed and written consent was obtained from the patient prior to the publication.
Ethical approval: The authors declare that the article follows the Helsinki declaration guidelines and the manuscript has not been submitted to any other journal.
References
- Rajkumar SC, Dimopoulos MA, Palumbo A, et al. The IMWG updated criteria for the diagnosis of multiple myeloma. The lancet. 2014; 15: 538-548.
- Joseph D. Verdirame, John R. Feagler, et al. Multiple myeloma associated with immune thrombocytopenic purpura. Cancer. 1985; 56: 1199-1200.
- Tabata R, Tabata C, Ayako T, et al. Low dose cepharanthine ameliorates immune thrombocytopenic purpura associated with multiple myeloma. International Immunopharmacologie. 2012; 13: 242-244.
- Shaji K. Kumar. Timing of treatment of smoldering myeloma: Delay until progression. blood advances. 2018; 2: 21.
- Liebman HA, Stasi R. Secondary immune thrombocytopenic purpura. CurrOpinHematol. 2007; 14: 557-73.
- Jain A, Khunger JM, Siddharth Kapahtia,et al. Smoldering Multiple Myeloma Presenting as Immune Thrombocytopenia. Indian J Hematol Blood Transfus. 2019; 35: 565–566.
- Alexei Shimanovsky, Argote J. Alvarez, Shruti Murali, et al. Autoimmune manifestations in patients with multiple myeloma and monoclonal gammopathy of undetermined significance. BBA Clinical. 2016; 6: 12-18.
- Yao H, Zhang X, Liu J, et al. Multiple myeloma developing during long-term clinical course of refractory immune thrombocytopenic purpura: A case report and review of literature. Int J Clin Exp Pathol. 2015; 8: 15429–15432.
- Gupta V, Hegde U.M, R. Parameswaran, et al. Multiple myeloma and immune thrombocytopenia. Clin.Lab. Haem. 2009; 22: 239– 242.
- Verdirame JD, Feagler JR, James RC. Multiple myeloma associated with immune thrombocytopenic purpura. Cancer. 1985; 56: 1199–1200.
- Falco P, Bertola A, Bringhen S, et al. Successful management of immune thrombocytopenic purpura with thalidomide in a patient with multiple myeloma. Hematol. 2004; 5: 456–457.
- Faller E, Chapman L, Enright H, et al. Immune thrombocytopenia purpura associated with multiple myeloma. Ann Hematol. 2016; 95: 1371–1372.
- Siniscalchi A, Stasi R, Stefano F, et al. Management of immune thrombocytopoenia in a patient with newly-diagnosed smouldering myeloma and colorectal cancer. BMJ Case Rep. 2009; bcr03: 1715.
- Humaira Sarfraz, Kartik Anand, Shujuan Liu,et al. Multiple myeloma with concurrent immune thrombocytopenic purpura. ecancer.2020; 14: 1012.
- Douglas B. Cines, Robert McMillan.Pathogenesis of chronic immune thrombocytopenic purpura. Curr Opin Hematol. 2007; 14: 511-514.
- Douglas B. Cines, Howard Liebman, Roberto Stasi.Pathobiology of secondary immune thrombocytopenia. Seminars in Hematology. 2009; 46, No 1, S2-S14.
- Philip R Greipp, Jesus San Miguel, Brian G M Durie,et al.International staging system for multiple myeloma. J Clin Oncol. 2005; 2325. 6281.
- Cindy Neunert, Deirdra R. Terrell, Donald M Arnold, et al,American Society of Hematology 2019 guidelines of immune thrombocytopenia.Blood Adv. 2019; 3: 3829–3866.
- Ola Landgren.Shall we treat smoldering multiple myeloma in the near future. American Society of Hematology. 2017; 1: 194.