Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Diplopia and skin rash in a patient treated with immunotherapy nivolumab regimen
Theodora-Maria Papadimitriou1; Alexandros-Nikolaos Liatsos2; David Symeonidis3*; Spyridon Xynogalos4
1 Resident of Internal Medicine, Metaxa Cancer Institute, Piraeus, Greece.
2 Resident of Nephrology, Evaggelismos General Hospital, Athens, Greece.
3 Resident of Medical- Oncology, Metaxa Cancer Institute, Piraeus, Greece.
4 Head of Medical- Oncology Department, Metaxa Cancer Institute, Piraeus, Greece.
*Corresponding Author: David Symeonidis
Resident of Medical- Oncology, Metaxa Cancer Institute, Piraeus, Greece.
Email: davidsymeonidis@yahoo.com
Received : Oct 12, 2021
Accepted : Nov 04, 2021
Published : Nov 11, 2021
Archived : www.jcimcr.org
Copyright : © Symeonidis D (2021).
Citation: Papadimitriou TM, Liatsos AN, Symeonidis D, Xynogalos S. Diplopia and skin rash in a patient treated with immunotherapy nivolumab regimen. J Clin Images Med Case Rep. 2021; 2(6): 1401.
Background
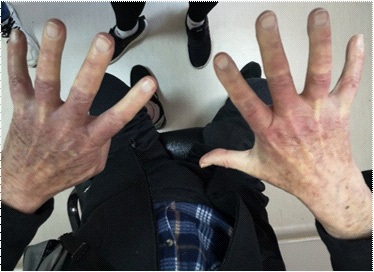
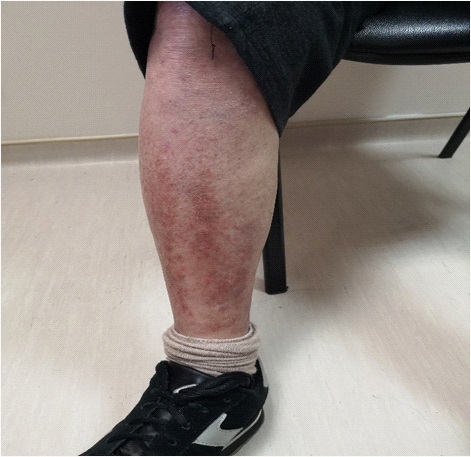
This is a case report of an 80-year-old caucasian male patient with metastatic urothelial bladder carcinoma and bone/liver metastases, who was being presented with diplopia, raynaud’s syndrome and skin rash on both legs, in response to immunotherapy with anti-PDL-1 Nivolumab regimen.
Case report
An 80-years old patient is being diagnosed with metastatic urothelial bladder carcinoma with liver, bone metastases and para-aortic lymph nodes. The diagnosis was based form liver biopsy. We initiated 1st line chemotherapy with carboplatin plus gemcitabine in March 2019 with a partial response after 3 cycles of therapy in both liver and lymph nodes. In September 2019 was presented with worsening upper abdomen pain and an ultra-sound (US) examination pointed out worsening disease confined to the liver. A new computed tomography (CT) of the abdomen confirmed the results. So, we proceed to a new line of therapy with vinflunine without adverse events. After 6 months in this regimen, a new CT scan showed progressive disease confined to liver and new perihilar lymph nodes in both lungs.
At that time, we decided to initiate 3rd line therapy with the anti-PDL1 Nivolumab 240 mg every 2 weeks. During the first 2 months the patient showed no symptoms, but at the beginning of 5th dose in the 3rd month of treatment he visited the emergency department in our hospital with acute onset binocular horizontal diplopia, on right lateral gaze, started 3 days ago and an irritating, eczematous skin rash on the anterior prominent of both legs, with numb and cold fingers in response to low temperatures (raynaud’s syndrome) [1].
Clinical examination on right lateral rectus showed esotropia in primary gaze, no findings in all other cranial nerves and no other neurological abnormalities [2]. A new CT scan of the brain did not provide us with any new data, it was a clear tomography, so we procced to a new Magnetic Resonance (MRI) of the brain, but again without any findings. Of course, we withdrew immunotherapy and started immediately dexamethasone at a dosage of 1 mg/kg with later suitable tapering. A neurologist examined the patient but could not find any other symptoms related to nervous system disorder based on negative tests for LambertEaton syndrome and myasthenia gravis, so we requested for an ophthalmological examination [3]. The ophthalmologist confirmed the diplopia symptomatology with no other findings.
After 7 days in dexamethasone treatment, we noted a gradually remission of all symptoms, both diplopia and the skin rash. The patient continued dexamethasone for a total duration of 4 weeks. His disease remained stable for 6 more months before a new progression. As for his diplopia is being examined every 3 months from an ophthalmologist without until now immune related findings.
Discussion
In this case, the patient suffered of horizontal diplopia caused by isolated cranial nerve VI palsy and Raynaud-like syndrome. After treating patient with high dose dexamethasone and withdrawing nivolumab the symptoms were fully improved suggesting an autoimmune profile [4]. In addition, without metastases in head and neck and without findings in CT of the skull base it is unlikely to be an isolated metastases to cranial nerve VI. In literature this isolated cranial nerve palsy is described only after administration of pembrolizumab and, to our knowledge, only 1 case of Raynaud-like syndrome has been published [5].
PD-1 is a co-receptor that is found on the surface of T cells. When PD-1 binds to one of its ligand, PD-L1 or PD-L2 , it regulates the autoimmune system by inhibiting the activation of T cell protecting against autoimmune diseases. Cancer cells can produce PD-L1 on their surface hiding themselves from immune system. PD-1 inhibitors (etc Nivolumab, Pembrolizumab) block this binding allowing autoimmune system to start an immune attack against both cancer cells or normal body cells [6] Τhere are assumptions that the perineural oedema and inflammation might be a causal factor of cranial nerve palsy [7,8]. The most common adverse effects of nivolumab are fatigue (46%), musculoskeletal pain (31%), diarrhea (26%), coughing (24%), rash (24%), shortness of breath (18%), arthralgia (18%) [2]. Inflammatory response as adverse effect can be found to any part of the body for example endocrine system, lung , gastrointestinal system and rarely to heart and nervous system (<1%) [1].
Neuromuscular complications of ICI-therapy are the most frequent neurological manifestations with myasthenia gravis characterized as most frequent (25% had a previous diagnosis of myasthenia gravis). Guillain-Barré syndrome is another severe adverse effect of the peripheral nervous system. symmetric painful paraesthesia and symmetric polyneuropathy were less common adverse effects. Neuromuscular side effects like myopathies, dermatomyositis , diplopia are not ordinary and more often related to administration of pembrolizumab [9].
The emergence of diplopia, Raynaud’s, and skin rash, induced by Nivolumab, demonstrates a new potential toxicity and highlights the need for careful monitoring and review of symptoms to avoid and minimize the impact of immune-related adverse events that can disrupt treatment or worsen patients’ conditions.
Conclusion
Diagnosis of mononeuritis and skin rash are conditions related to immunotherapy. Early identification and treatment are important elements for a patient’s course. Possible such side effects also highlight the effectiveness of immunotherapy whose action remains even after discontinuation.
References
- Kumar V, Chaudhary N, Garg M, Floudas CS, Soni P, Chandra AB. Current diagnosis and management of immune related adverse events (irAEs) induced by immune checkpoint inhibitor therapy. Frontiers in Pharmacology. 2017; 8: p. 49.
- Tanaka R, Maruyama H, Tomidokoro Y, et al. Nivolumab-induced chronic inflammatory demyelinating polyradiculoneuropathy mimicking rapid-onset Guillain-Barré syndrome: a case report. Japanese Journal of Clinical Oncology. 2016; 46(9): 875–878.
- Shirai T., Sano T., Kamijo F., et al. Acetylcholine receptor binding antibody-associated myasthenia gravis and rhabdomyolysis induced by nivolumab in a patient with melanoma. Japanese Journal of Clinical Oncology. 2016; 46(1): 86–88.
- Sanders S, Kawasaki A, Purvin V. Long-term prognosis in patients with vasculopathic sixth nerve palsy. Am J Ophthalmol. 2002;134:81–84.
- Khan S, von Itzstein MS, Lu R, Bermas BL, Karp DR, Khan SA, Fattah FJ, Park JY, Saltarski JM, Gloria-McCutchen Y, Xie Y, Li QZ, Wakeland EK, Gerber DE. Late-Onset Immunotherapy Toxicity and Delayed Autoantibody Changes: Checkpoint InhibitorInduced Raynaud’s-Like Phenomenon. Oncologist. 2020; 25(5): e753-e757.
- Villadolid J., Amin A. Immune checkpoint inhibitors in clinical practice: update on management of immune-related toxicities. Translational Lung Cancer Research. 2015; 4(5): 560–575.
- King A, Stacey E, Stephenson G, Trimble R. Spontaneous recovery rates for unilateral sixth nerve palsies. Eye. 1995; 9: 476– 478.
- Boutros C., Tarhini A., Routier E., et al. Safety profiles of anti-CTLA-4 and anti-PD-1 antibodies alone and in combination. Nature Reviews Clinical oncology. 2016; 13(8): 473–486.
- Korey A. Jaben, Jasmine H. Francis, Alexander N. Shoushtari & David H. Abramson (2019): Isolated Abducens Nerve Palsy Following Pembrolizumab, NeuroOphthalmology