Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Intrabiliary papillary myxoma: A case report
Lu Hexiao1; Zhang Shenglong2; Bhushan Sandeep3; Yin Sineng4*
1 Zunyi Medical University, Zunyi, Guizhou 563003, China.
2 Department of General Surgery, Chengdu Second People’s Hospital, Chengdu, Sichuan 600017, China.
3 Department of Cardiothoracic surgery, Chengdu second People’s Hospital, Chengdu, Sichuan 610017, China
4 Department of General Surgery, Zunyi Medical University, Chengdu Second People’s Hospital, Chengdu, Sichuan 600017,China
*Corresponding Author: Yin Sineng
Department of General Surgery, Zunyi Medical University, Chengdu Second People’s Hospital, Chengdu,
Sichuan 600017, China.
Email: doctoryinsn@sina.com
Received : Oct 10, 2021
Accepted : Nov 10, 2021
Published : Nov 17, 2021
Archived : www.jcimcr.org
Copyright : © Sineng Y (2021).
Abstract
In 2010, the World Health Organization (WHO) classified intraductal papillary mucinous tumor (ipmn-b) as a solid tumor of the liver producing mucinous bile, which is rare and difficult to diagnose. We reported a 71 year old female patient with ipmn-b due to abdominal pain. Through the description of the causes, diagnosis, treatment and prognosis, we can get a better understanding of the disease. Ipmn-b is considered as a kind of benign precancerous lesion of cholangiocarcinoma with good prognosis.
Keywords: papillary mucinous tumor; cholangiocarcinoma; neoplasm.
Citation: Hexiao L, Shenglong Z, Sandeep B, Sineng Y. Intrabiliary papillary myxoma: A case report. J Clin Images Med Case Rep. 2021; 2(6): 1410.
Introduction
Intraductal papillary mucinous neoplasm of the bile duct (ipmn-b) is a kind of intraductal papillary neoplasm of the bile duct (ipnb). It is a rare benign lesion with high malignant transformation rate. It is characterized by intraluminal papillary mass with bile duct obstruction and dilatation. Some of these lowgrade malignant tumors secrete large amounts of mucin which can change the normal flow of bile and lead to the expansion and spread of intrahepatic and extrahepatic bile ducts. Here we are reporting a case of ipmn-b in intrahepatic bile duct which was recently treated in Chengdu Second People’s hospital.
Case report
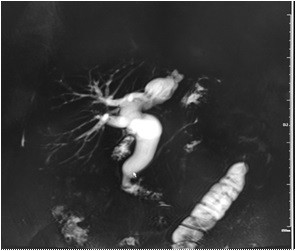
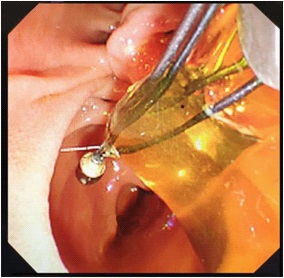
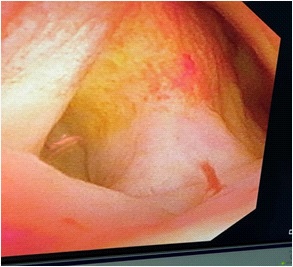
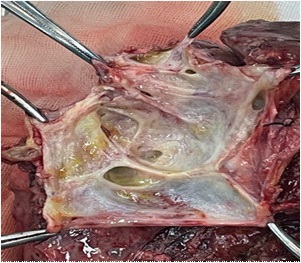
A 71 years old female patient was admitted with “upper abdominal pain for 5 days”. 10 years before admission, he underwent partial resection of the left lateral hepatic lobe, cholecystectomy, common bile duct exploration and T tube drainage for intrahepatic bile duct stones and gallbladder stones Surgery. 5 years before admission, due to common bile duct stones, abdominal cavity exploration, common bile duct exploration and T tube drainage was performed. The patient had repeated upper abdominal pain for 5 days, accompanied by chills and high fever, with a maximum body temperature of 39.0 degrees Celsius, accompanied by nausea and vomiting. An emergency CT scan in our hospital showed that the intrahepatic bile ducts were dilated and the left bile duct and common bile duct were significantly dilated with punctate calculi. She was admitted to our department for further treatment. Physical examination on admission shows her general condition was good, the skin and sclera are mild yellowish, and multiple scars from previous surgery are seen. Her other examination shows tumor indicators: CA19-9, CEA and AFP had no obvious abnormalities, her blood and biochemical examination after admission is presented in Table 1. Her abdominal ultrasound shows intra and extra hepatic bile ducts and main pancreatic duct was dilated. There was a sedimentary stone in the lower part of the common bile duct. Her MRI+MRCP shows intrahepatic and extrahepatic bile ducts were dilated (the common bile duct of the left lobe of the hepatic bile duct was significant with a transverse diameter of about 2.4 cm; the signal in the lumen of the lower part of the common bile duct is locally reduced, which was a silt-like stone. ERCP subsequently shows the patient underwent open lower left third lobe hepatectomy, hepatobiliary lesion and Ttube drainage. During the operation, obvious expansion of common bile duct >3 cm. After puncture, golden jelly like liquid was extracted, and the upper segment was longitudinally cut for 1 cm. After choledochoscopy the opening of the large duodenal papilla was fishmouth like, and the left hepatic duct was not developed, and the common bile duct could not be filled. The contrast agent flowed into the duodenum along the wall of the duct, and the jelly like mucus was removed from the stone mesh (Figure 2). Intrabiliary papillary mucinous tumor (ipnb-m) was considered by endoscopy. The patients recovered half a month after the operation and were followed up in the first month, the third month and the sixth month after the operation. The general condition was good and no tumor recurrence was found.
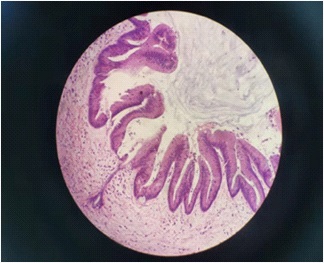
Postoperative pathology: Figure 5 medium grade intraductal papillary mucinous tumor of bile duct, dilatation of peripheral bile duct with medium grade intraductal neoplasia, and fatty degeneration of surrounding liver tissue. Immunohistochemistry: CK19 (+), CK7 (+), PCK (+), MUC5AC (+), MUC2 (+), ER (+), PR (+), Ki-67 (index 15%). It is consistent with intraductal papillary mucinous tumor (intermediate type).
Table 1: Changes in blood report and liver function of patients after admission
Date |
WBC(*109/L) |
Total bilirubi(mmol/L) |
Direct bilirubin mmol/L) |
Alanine transamination Transferase (U/L) |
Glutamy transferase (U/L) |
Alkaline phosphatse (U/L) |
2020-06-18 |
11.65 |
23.9 |
14.3 |
223 |
488 |
176 |
2020-06-25 |
3.73 |
35 |
24.2 |
282 |
273 |
129 |
2020-07-02 |
11.60 |
29.5 |
17.5 |
239 |
89 |
71 |
2020-07-05 |
10.48 |
19.0 |
11.9 |
213 |
78 |
67 |
2020-07-09 |
16.30 |
37.4 |
27.1 |
47 |
54 |
78 |
Discussion
Intrabiliary papillary myxoma is derived from bile duct epithelial cells and is supported by connective tissue from the lamina propria. It is a rare benign tumor characterized by papillary and villous spreading and growth into the duct. It is histologically described as mucinous and papillary like tumor [1]. In 2010, the World Health Organization (WHO) classified mucinous cystic tumors and intraductal papillary mucinous tumors into two different solid tumors with rare onset [2]. Rate and mucin production are their common features, and the presence of ovarian-like stroma (OLS) has been identified as the diagnosis of MCN-L.
In related studies, IPMN-B is considered to be the biliary counterpart of pancreatic papillary mucinous tumor (IPMN-P), which is similar to IPMN-P, and IPNM-B has the typical characteristics of bile ducts, which can produce a large amount of mucin, leading to bile ducts extensive expansion [3]. IPNM-B histology can be divided into four types of pancreaticobiliary duct type, intestinal type, gastric type and eosinophil type. The most common type is pancreaticobiliary duct type, which accounts for about 47%. The expression of MUC1 and MUC2 can be distinguished by immunohistochemical staining [4]. Sakamoto et al. divided IPMN-B patients into 3 types, type 1 bile duct dilatation, type 2 cyst type, and type 3 intermediate type, which are a mixture of the above two types [5]. The cases reported in this article belong to the typical intermediate type lesions. The incidence of IPMN-B is extremely low. The incidence in eastern countries such as Japan and South Korea is much higher than that in western countries. This is because endemic hepatolithiasis, cholangitis and biliary infections are more common in Asian countries. This is the occurrence of IPMN-B. The main risk factors of IPMN-B are considered to be a precancerous lesion with adenoma cancer sequence, with a cancerous rate of 64%-89% [6]. The main clinical manifestations are intermittent abdominal pain, obstructive jaundice, and fever. Intermittent jaundice is helpful for the diagnosis of IPMN-B, which may be due to its secretion of more mucin and intermittent tumor separation caused by intermittent obstruction of the bile duct. Moreover, 30% of IPNM-B patients may be accompanied by bile duct stones, which are easily misdiagnosed as simple bile duct stones and repeated visits . There is no specific laboratory test for IPMN-B, and fluctuating bilirubin can be of certain value for its diagnosis. The levels of CEA and CA242 can help distinguish between benign and malignant IPNM-B. Imaging methods such as MRCP, CT, and ultrasound are also of limited diagnostic value. The imaging manifestations of IPMN-B are tumors in the bile ducts, diffusely dilated bile ducts and mucus in the ducts, which are the unique characteristics of IPMN-B. Approximately 68% of patients may have simultaneous expansion of the proximal and distal bile ducts [7]. This is because the upstream expansion of the bile duct may be due to the tumor’s own obstruction or the stenosis caused by the tumor, and the downstream expansion is due to excessive mucin secretion. When the hepatopancreas ampulla is obstructed, it causes diffuse dilation of the intrahepatic and extrahepatic bile duct system.Therefore; we should be highly alert to this disease when we find diffuse dilatation of the common bile duct. There are also some patients who have not found tumors or nodules in the bile ducts, because MRCP and CT are difficult to detect small or flat tumors, and it is also difficult to detect mucin. The CT and MRCP signal intensity of mucin is almost the same as that of bile [8].
At this time, it needs to be combined with ERCP. The defect of the bile duct filled with contrast agent in ERCP does not match the smooth tube wall filled with contrast agent on CT, and the duodenal papilla or bile duct has jelly-like mucus in the choledochoscopy, which is of diagnostic significance, CT+MRCP examination in this case report showed obvious bile duct dilation and special signs of ERCP, which were consistent with the description in the literature. IPMN-B has a low incidence, atypical clinical symptoms and signs, and its preoperative diagnosis is very difficult. It needs to be related to hepatic mucinous mucinous cystic tumor (MCN-L), cholangiocarcinoma, congenital biliary dilatation, and recurrent bile duct Identification of stones.
There are various treatment methods for IPMN-B, including surgical resection of lesions, liver transplantation, and palliative treatment. Due to the characteristics of invasive precancerous lesions, radical surgical resection is the preferred treatment method. The surgical effect is good, and its five-year survival rate can reach 84% [9]. For patients who have no chance of surgery or who cannot tolerate surgery, laser cauterization or ablation, chemotherapeutic perfusion, argon ion beam coagulation, combined with endoscopic placement of bile duct stents, nasobiliary ducts or PTCD can be selected. It is also useful for IPMNB patients certain effect. In general, the prognosis of IPMN-B is better than that of cholangiocarcinoma, but the recurrence rate is higher. According to statistics, the recurrence after radical surgical resection is as high as 29.3% [9], so IPNM-B requires long-term follow-up even if the margin is negative.
Conclusion
IPMN-B is a type of benign biliary tract tumors that are clinically rare, difficult to diagnose, and have a high malignant rate. During the clinical treatment process, we need to be highly vigilant about this type of disease, reduce misdiagnosis and missed diagnosis, and early detection and early treatment are better for such patients. Improve the quality of life and improve the survival rate.
Declarations
Ethics approval and consent to participate: The protocol was approved by the Chengdu Second People’s Hospital Clinical Research Ethics Committee, and parents of all subjects provided informed consent.
Consent for publication: Not applicable.
Availability of data and materials: Contact author if needed.
Competing interests: The authors’ declare that there are no conflicts of interests.
Funding: Not Applicable.
Authors’ contributions: All the authors conceptualized this case, designed the contents and developed the search strategy. Author Lu Hexiao and Sandeep Bhushan conducted the literature search, summarized the report, and prepared the first draft of the article. All authors contributed to synthesis of the results of the final manuscript. All authors contribute equally to this manuscript.
Acknowledgements: Not applicable
References
- Ritchie David J, Okamoto Kanenori, White Stacey L. Intraductal papillary mucinous neoplasm of the biliary tract: A precursor lesion to cholangiocarcinoma. Radiol Case Rep. 2019; 14(4): 495- 500.
- DeLellis. WHO Classification of Tumours of the Digestive System. Fourth Edition[J]. 2010.
- Kunovsky Lumir, Kala Zdenek, Svaton Roman, Moravcik Petr, Mazanec Jan, Husty Jakub, Prochazka Vladimir. Mucinous Cystic Neoplasm of the Liver or Intraductal Papillary Mucinous Neoplasm of the Bile Duct? A Case Report and a Review of Literature. Ann Hepatol. 2018; 17(3): 519-524.
- Sclabas Guido M, Barton Joshua G, Smyrk Thomas C, Barrett David A, Khan Saboor, Kendrick Michael L, Reid-Lombardo Kaye M, Donohue John H, Nagorney David M, Que Florencia G. Frequency of subtypes of biliary intraductal papillary mucinous neoplasm and their MUC1, MUC2, and DPC4 expression patterns differ from pancreatic intraductal papillary mucinous neoplasm. J Am Coll Surg. 2012; 214(1): 27-32.
- Sakamoto E, Nimura Y, Hayakawa N, et al. Clinicopathological studies of mucin-producing cholangiocarcinoma. J Hepatobiliary Pancreat Surg. 1997; 4: 157-62.
- Wan XS, Xu YY, Qian JY, Yang XB, Wang AQ, He LH, et al. Intraductal papillary neoplasm of the bile duct. World J Gastroenterol. 2013; 19(46): 8595-604.
- Takanami Kentaro, Yamada Takayuki, Tsuda Masashi, Takase Kei, Ishida Kazuyuki, Nakamura Yasuhiro, Kanno Atsushi, Shimosegawa Tooru, Unno Michiaki, Takahashi Shoki. Intraductal papillary mucininous neoplasm of the bile ducts: multimodality assessment with pathologic correlation. Abdom Imaging. 2011; 36(4): 447-56.
- Sohn Won-Joon, Jo Sungho. A huge intraductal papillary mucinous carcinoma of the bile duct treated by right trisectionectomy with caudate lobectomy. World J Surg Oncol. 2009; 93.
- ROCHA F G, LEE H, KATABI N, et al. Intraductal papillary neoplasm of the bile duct: a biliary equivalent to intraductal papillary mucinous neoplasm of the pancreas? [J]. Hepatology. 2012; 56(4): 1352-1360.