Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Severe obesity in a patient on long-term treatment of chronic myeloid leukemia with imatinib: Body composition, associated disorders and outcome
Jorge Illarramendi1*; María Angeles Goñi1; Montserrat Alvarellos1; Ana Zugasti2; Jose Juan Illarramendi3
1 Services of Hematology, Hospital Universitario de Navarra, Pamplona, Spain.
2 Services of Endocrinology & Nutrition, Hospital Universitario de Navarra, Pamplona, Spain.
3 Services of Medical Oncology, Hospital Universitario de Navarra, Pamplona, Spain.
*Corresponding Author: Jose Juan Illarramendi
Service of Hematology, Hospital Universitario de Navarra, Irunlarrea, 3. 31008. Pamplona, Spain.
Email: illarrajorge@gmail.com
Received : Sep 20, 2021
Accepted : Nov 12, 2021
Published : Nov 19, 2021
Archived : www.jcimcr.org
Copyright : © Illarramendi J (2021).
Abstract
Chronic Myeloid Leukemia (CML) is a highly curable malignancy with tyrosine kinase inhibitors (TKI) as target therapy. As treatment is usually prolonged, there is a need to improve our knowledge about the impact of comorbidities in this context. Severe obesity is among the common comorbidities in these patients. We present the case of a patient with morbid obesity and associated sleep apnea hypopnea syndrome who was succesfully treated during 14 years with imatinib, a cornerstone TKI for this disease. Visceral fat area was extremely elevated in this patient.
Keywords: chronic myeloid leukemia; obesity; imatinib; sleep apnea hypopnea syndrome.
Abbreviations: CML: Chronic Myeloid Leukemia; BMI: Body Mass Index; TKI: Tyrosine Kinase Inhibitors.
Citation: Illarramendi J, Goñi MA, Alvarellos M, Zugasti A, Illarramendi JJ. Severe obesity in a patient on long-term treatment of chronic myeloid leukemia with imatinib: Body composition, associated disorders and outcome. J Clin Images Med Case Rep. 2021; 2(6): 1413.
Introduction
Modern treatment of chronic myeloid leukemia (CML) with tyrosine kinase inhibitors (TKI) is regarded among the most succesful developments in cancer medicine [1]. Overweight and obesity are common in many world areas, but there is a very limited knowledge on the impact of obesity in patients treated with TKI for CML [2]. We present the case of a patient who developed severe (morbid) obesity during the treatment with imatinib, with data on body composition, associated disorders and clinical outcome.
Case presentation
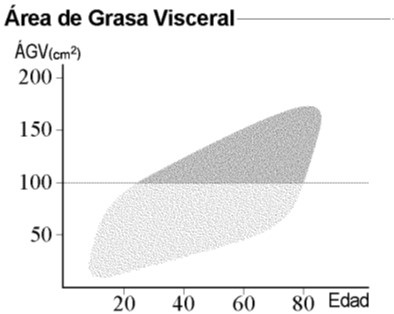
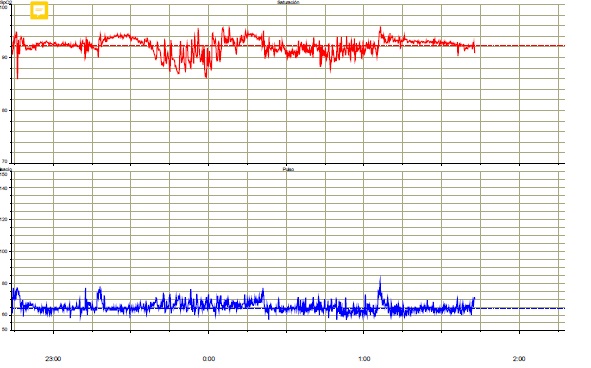
A Caucasian woman, born in February 1958, was diagnosed with chronic-phase CML in June 2007. She smoked tobacco until 1985 and had a previous diagnosis of breast cancer in 1993. Sokal and Hasford indexes were 0.72 and 4.13, respectively. Imatinib was started in June 2007 and mantained until June 2021, with a good clinical and molecular response (RM5). Complete cytogenetic response was obtained at 6 months and major molecular response at 17 months. Maintenance dose of imatinib was 400 mg/day. She was overweight during the adolescence but started to have a marked increase of weight after the diagnosis of CML. Weight in June 2021 was 121 kg, with a BMI of 48. Brachial perimeter: 40 cms. Abdominal perimeter: 129 cms. Hip perimeter: 125 cms. Body composition was studied with the InBody 770 analyzer, showing a normal percentage of fat body mass. The visceral fat area was extremely elevated to 246 cm2 (normal <100 cm2 ) (Figure 1). Study was completed with plethysmography and nocturnal pulsioximetry (Figure 2), suggesting a sleep apnea hypopnea syndrome. Some other events during the period of imatinib treatment with a potential association to obesity included leg deep vein thrombosis that was treated with acenocoumarol, peripheral edema and episodes of leg cellulitis. She needed conventional hospitalization for COVID-19 in November 2020, without complications and with ongoing antibody response until March 2021. Relevant drug therapies included long-term omeprazole for reflux esophagitis and torasemide for peripheral edema. She remains in complete clinical, cytogenetic and molecular response and in good condition after discontinuation of imatinib, with follow-up in the departments of Hematology, Medical Oncology and Nutrition.
Discussion
Several meta-analyses have shown an increase of incidence and mortality of CML in obese persons [3-6].
Significant weight gain has been claimed in one review of patients with CML after imatinib therapy, with more of 40% categorized as obese after 24 months of treatment [7], and similar data are available in other studies with wide samples of this group of patients [8,9]. This effect was also present in a series of 1142 surveyed Chinese CML patients treated with TKI. 42% of them referred weight gain, although the incidence was lower in patients treated with imatinib alone [10]. Some authors consider women as more prone to weight gain [11].
In relation to the hematologic outcomes, there have been a few reports in the literature on CML patients with severe obesity and/or bariatric surgery treated with TKI. Interestingly, one case report described a woman with a BMI of 50 and a weight of 122 kg with obstructive sleep apnea syndrome and gastroesophageal reflux who needed a higher dose of imatinib (800 mg/d) to obtain a complete cytogenetic response and was treated later with bariatric surgery [12]. Another report of mantained major molecular response with a dose of 800 mg/d has been described, but the patient was an extreme case of a woman with a BMI of 113 and a weight of 275 kg, different from other case in the same report of a man with a BMI of 51 and a weight of 166 kg who obtained the molecular target with an imatinib dose of 400 mg/d [13]. One further woman with a previous bariatric surgery for weight loss achieved a complete response with 400 mg/d of imatinib, but the authors did not explain if severe obesity was still present [14]. Another case of complete molecular response during the treatment with 400 mg/d of imatinib has been described in a CML woman with morbid obesity [15]. The authors found that bariatric surgery influenced the pharmacokinetics of imatinib, but the patient mantained the complete response with the dose of 400 mg/d. In fact, some authors recommend imatinib over second generation TKI like dasatinib or nilotinib in patients with gastric surgery for morbid obesity [16]. A delay in cytogenetic and molecular responses is another point to consider because it has been reported for patients with high BMI [17]. Nevertheless, that study was directed to patients with BMI >25 and not to patients with severe obesity as a group. The time to the achievement of major molecular response in our case report is closer to their group of patients with low BMI. A Japanese series has reported that patients with lower weight were less likely to obtain a complete cytogenetic response with imatinib [18].
Besides the case report cited previously, sleep apnea hypopnea syndrome has been ocasionally reported in patients with CML [19]. Regarding sleep apnea hypopnea syndrome in this context, we must regard that disturbed sleep is described among the most severe symptoms affecting quality of life in CML, and is included in specific questionnaires to evaluate the quality of life in CML patients, as the EORTC QLQ-CML24 [20] and the MD Anderson Symtom Inventory (MDASI) for patients with CML known as MDSAI [21]. Furthermore, one study has found that a high BMI and disturbed sleep are the most significant factors in relation to fatigue in these patients [22]. Discontinuation of TKI in patients in complete response may improve their quality of life because of a better sleep pattern. More than 20% of patients with CML in complete response participating in The Life After Stopping Tyrosine Kinase Inhibitors (LAST) had a meaningful improvement in sleep disturbances after stopping this treatment [23].
Prevention of cardiovascular toxicity is another important point to consider in CML patients with severe obesity. In fact, diet and weight management have been described as the letter D in the ABCDE steps to prevent cardiovascular disease in patients with CML treated with a TKI [24].
Conclusion
Long-term therapy with a conventional dose of imatinib was succesful for inducing and mantaining an adequate response in this patient. No increased doses were needed. Severe obesity developed during the treatment, with a very high area of visceral fat, and was associated to sleep apnea hypopnea syndrome, with a potentially impairing effect on her quality of life.
References
- Claudiani S, Apperley JF. The argument of using imatinib in CML. Hematology Am Asoc Hematol Educ Program. 2018: 161-167.
- Gougis P, Palmieri LJ, Funck-Brentano C, Paci A, Flippot R, et al. Major pitfalls of protein kinase inhibitors prescription: A review of their clinical pharmacology for daily use. Crit. Rev. Oncol. Hematol. 2019; 141: 112-124.
- Larsson SC, Wolk A. Overweight and obesity and incidence of leukemia: a meta-analysis of cohort studies. Int J Cancer. 2008; 122: 1418-1421.
- Castillo JJ, Reagan JL, Ingham RR, Furman M, Dalia S, Merhi B, et al. Obesity but not overweight increases the incidence and mortality of leukemia in adults: A meta-analysis of prospective cohort studies. Leuk Res. 2012; 36: 868-875.
- Psaltopoulou T, Sergentanis TN, Ntanasis-Stathopoulos I, Tzanninis IG, Riza E, Dimopoulos MA. Anthropometric characteristics, physical activity and risk of hematological malignancies: A systematic review and meta-analysis of cohort studies. Int J Cancer. 2019; 145: 347-359.
- Teras LR, Patel AV, Carter BD, Rees-Punia E, McCullough MI, Gapstur SM. Anthropometric factors and risk of myeloid leukaemias and myelodysplastic syndromes: a prospective study and meta-analysis. Br. J Haematol. 2019; 186: 243-254.
- Aduwa E, Szydlo R, Marin D, Foroni L, Reid A, Goldman J, et al. Significant weight gain in patients with chronic myeloid leukemia after imatinib. Blood. 2012; 120: 5087-5088.
- Jain P, Kantarjian H, Boddu P, et al. Analysis of cardiovascular and arteriothrombotic adverse events in chronic-phase CML patients after frontline TKIs. Blood Adv. 2019; 3: 851-861.
- Coutinho AD, Makenbaeva D, Farrelly E, Landsman-Blumberg PB, Lenihan D. Elevated cardiovascular disease risk in patients with chronci myelogenous leukemia seen in community-based oncology practices in the United States. Clin Lymphoma Myeloma Leuk. 2017; 17: 676-683.
- Yu L, Huang X, Gale RP, Wang H, Jiang Q. Variables associated with patient-reported symptoms in persons with chronic phase chronic myeloid leukemia receiving tyrosine kinase inhibitor therapy. Medicine (Baltimore). 2019; 98: e18079.
- Breccia M, Salaroli A, Serrao A, Zacheo I, Alimena G. Imatinib induced body mass changes in women with chronic myeloid leukemia. Ann Hematol. 2013; 92: 1581-1582.
- Liu H, Artz A. Reduction of imatinib absorption after gastric bypass surgery. Leuk. Lymphoma. 2011; 52: 310-313.
- Yassin MA, Kassem N, Ghassoub R. How I treat obesity and obesity related surgery in patients with chronic myeloid leukemia: An outcome of an ELN project. Clin Case Rep. 2021; 9: 1228- 1234.
- Centrone R, Bellesso M, Bonito D, Dias D, Santucci R, Aranha M, et al. Experience using imatinib and then nilotinib, as second line, in patient with chronic myeloid leukemia and previous bariatric surgery. A case report. Hematol Transfus Cell Ther. 2020; 42: 373-375.
- Pavlosky C, Egorin MJ, Shah DD, Beumer JH, Rogel S, Pavlosky S. Imatinib mesylate pharmacokinetics before and after sleeve gastrectomy in a morbidly obese patient with chronic myeloid leukemia. Pharmacotherapy. 2009; 29: 1152-1156.
- Yassin MA, Nashwan A, Kassem N. Second generation tyrosine kinase inhibitors as upfront therapy in the era of sleeve gastrectomy. Does it work?. Blood. 2017; 130.
- Breccia M, Loglisci G, Salaroli A, Serrao A, Mancini M, Diverio D, et al Delayed cytogenetic and major molecular responses associated to increased BMI at baseline in chronic myeloid leukemia patients treated with imatinib. Cancer Lett. 2013; 333: 32-35.
- Kanda Y, Okamoto S, Tuachi T, Kizaki M, Inokuchi K, et al. Multicenter prospective trial evaluating the tolerability of imatinib for japanese patients with chronic myelogenous leukemia in the chronic phase: Does body weight matter?. Am. J Hematol. 2008; 83: 835-839.
- Cornea MIP, Levrat E, Pugin P, Betticher DC. BCR-ABL1-positive chronic myeloid leukemia with erythrocytosis presenting as polycythemia vera: a case report. J Med Case Rep. 2015; 9: 30.
- Efficace F, Iurlo A, Patriarca A, Stagno F, Bee PC, et al. Validation and reference values of the EORTC QLQ-CML24 questionnaire to assess health-related quality of life in patients with chronic myeloid leukemia. Leuk Lymphoma. 2021. 2021: 62: 669-678.
- Williams LA, Garcia Gonzalez AG, Ault P, Mendoza TR, Sailors MLWilliams JL, et al. Measuring the symptom burden associated with the treatment of chronic myeloid leukemia. Blood. 2013; 122: 641- 647.
- Oswald LB, Hyland KA, Eisel SL, Hoogland AI, Knoop H, Nelson AM, et al. Correlates of fatigue severity in patients with chronic leukemia treated with targeted therapy. Support Care Cancer. 2021.
- Atallah E, Schiffer CA, Radich JP, Weinfurt KP, Zhang MJ, et al. Assessment of outcomes after stopping tyrosine kinase inhibitors among patients with chronic myeloid leukemia: A nonrandomized clinical trial. JAMA Oncol. 2021; 7: 42-50.
- Cortes J. How to manage CML patients with comorbidities. Blood. 2020; 136: 2507-2512.