Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Variations in treatment of an anastomotic leakage after Ivor Lewis Esophagectomy
Merel Lubbers1*; Frans van Workum2; Gijs Berkelmans3; Camiel Rosman2; Misha D Luyer3; Grard AP Nieuwenhuijzen3; Marc J van Det1; Ewout A Kouwenhoven1
1 Hospital Group Twente (ZGT), Department of Surgery, Zilvermeeuw 1, 7609 PP, Almelo, The Netherlands.
2 Radboud University Medical Centre, Department of Surgery, Geert Grooteplein Zuid 10, 6525 GA, Nijmegen, The Netherlands.
3 Catharina Hospital, Department of Surgery, Michelangelolaan 2, 5623 EJ, Eindhoven, The Netherlands.
*Corresponding Author: Merel Lubbers
Hospital Group Twente (ZGT), Departement of Surgery,
Zilvermeeuw 1, 7609PP Almelo, the Netherlands.
Email: m.lubbers@zgt.nl
Received : Sep 22, 2021
Accepted : Nov 15, 2021
Published : Nov 22, 2021
Archived : www.jcimcr.org
Copyright : © Lubbers M (2021).
Abstract
Background: Anastomotic Leakage (AL) after Ivor Lewis Esophagectomy (ILE) is a severe complication that often needs immediate treatment. However, there is no consensus on the optimal treatment. The aim of this study was to describe the outcomes of the different treatment options in patients with either contained or uncontained AL after ILE.
Methods: A retrospective analysis was performed on patients that developed AL after ILE in three high volume hospitals. Treatment was based on local preference. Endoscopic and surgical treatment were compared for patients with either contained (leakage confined to the mediastinum) or uncontained AL (leakage with intrapleural manifestations).
Results: In total, 73 patients with an AL were included. A contained leak was observed in 39 patients. Twenty-five patients (64%) underwent an endoscopic approach that was successful in 19 patients (76%); fourteen patients (36%) underwent a surgical approach that was successful in 11 patients (79%). Significantly more patients were (re)admitted to the ICU in the surgical group; other outcomes were similar.
An uncontained leak was observed in 34 patients. Endoscopic treatment was chosen in 14 patients (41%) and was successful in 10 patients (71%). A surgical approach was performed in 20 patients (59%) and was successful in 12 patients (60%). (Re) admission rate to the ICU was significantly higher in the surgical group, other outcomes were similar.
Conclusions: This study demonstrates that there is high variability in the treatment of AL after esophagectomy. Surgical and endoscopic techniques are both successfully used for patients with either contained or uncontained leakages. However, more research is necessary before a treatment algorithm can be developed.
Keywords: esophageal cancer; esophageal surgery; minimally invasive surgery; anastomotic leakage; endoscopic procedures.
Citation: Lubbers M, Workum FV, Berkelmans G, Rosman C, Luyer MD, et al. Variations in treatment of an anastomotic leakage after Ivor Lewis Esophagectomy . J Clin Images Med Case Rep. 2021; 2(6): 1417.
Introduction
Anastomotic Leakage (AL) is still a feared and life-threatening complication after Ivor Lewis Esophagectomy (ILE) with reported incidence rates of 5-25% and mortality rates up to 20-40 [1-5]. The classification of the Esophagectomy Complications Consensus Group (ECCG) has been introduced to differentiate between different types of AL [6]. This system classifies patients based on the type of therapy used during the treatment process. It is a helpful system to provide insight into which therapies have been used and its related outcomes. However, this classification does not take the location or the extent of the AL into account to provide a tool for an optimal treatment strategy.
Modalities to treat AL are diverse, varying from non-invasive strategies - antibiotics and nil per mouth - to invasive - endoscopic techniques such as clipping, stenting, placement of suction-drainage tubes, endovacuum therapy and surgery- with varying outcomes. There are multiple descriptive, retrospective, studies on the feasibility and effectiveness of a single modality treatment and some non-randomised studies comparing different techniques. However, there are no randomised studies on the safety and efficacy of these treatment modalities [3,7-10]. Consequently, it is still unknown which treatment is optimal with regard to safety and recovery in specific types of leakage. A recently published review showed similar results advising customized treatment depending on sequelae of the leak and the clinical condition of the patients [11].
The dynamic nature of AL makes comparisons between treatment modalities challenging. For objective comparison of different treatment strategies, patient’s inflammatory-response to AL, the severity and type of AL should be clearly categorised. Crestanello et all and Guo et all developed a classification to differentiate between contained and uncontained AL. This classification is based on the radiological extent of AL, and might be helpful in finding the most appropriate treatment for the different types of leakage [12,13].
The aim of this multicentre cohort study was to describe the outcomes of the different treatment options for patients with a contained and an uncontained anastomotic leakage after esophagectomy.
Materials and methods
Study design
This multicentre retrospective cohort analysis was performed in three high volume referral hospitals for esophageal cancer in the Netherlands, performing at least 40 esophagectomies annually. All patients were treated with curative intent for cT1-4aN0-3M0 esophageal cancer and received neoadjuvant chemoradiotherapy according to the CROSS-regimen unless contra-indicated [14]. All patients underwent totally minimally invasive esophagectomy with intrathoracic anastomosis as described by Ivor Lewis. Perioperative protocols (e.g. the positioning of the patients, the anastomotic procedure and the placements of drains and nasogastric tubes at the end of the surgery) differed between hospitals since this was a retrospective analysis.
Patients who developed AL in the period of January 2011 to September 2016 were included in this analysis [6]. Treatment strategies for AL were chosen based on local preferences of the upper-GI surgical teams of the three hospitals.
This retrospective study was part of an Institutional Review Board-approved protocol for ongoing assessment of esophagectomy outcomes and patient’s consent was waived (approval number ZGT17-30).
Diagnosis
AL was defined according to the ECCG definition as a full thickness defect involving the esophagus, anastomosis, staple line or conduit [6]. Routine post-operative diagnostics for AL were not performed. A contrast-enhanced CT-scan was performed in case of a suspicion of AL in order to confirm and define the extent of the leakage. If there was any doubt on the diagnosis AL after the CT-scan, patients underwent an endoscopy to confirm the diagnosis within 48 hours.
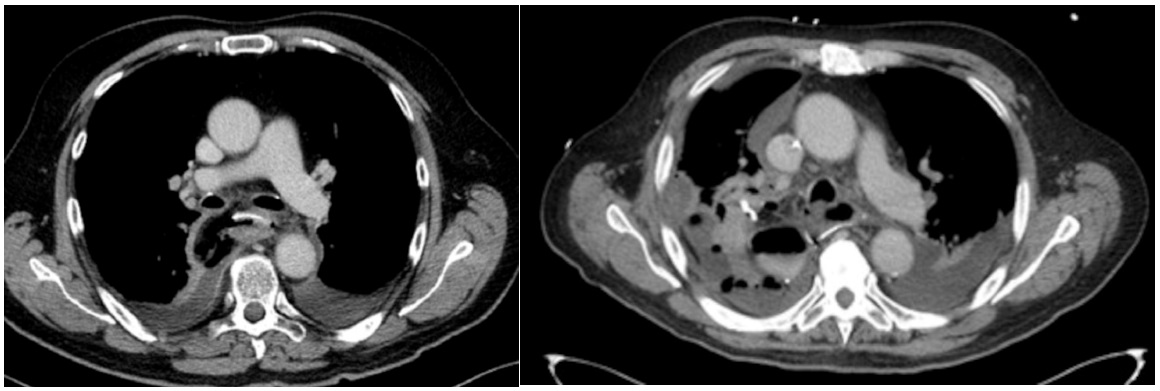
All CT-scans were evaluated by local radiologists and the leakage was stratified into contained leakage (cAL) or uncontained leakage (uAL). uAL was diagnosed in case of empyema or direct leakage of oral contrast into the thoracic cavity. cAL was diagnosed in patients with a limited AL, with the leakage of oral contrast limited to the mediastinum and no signs of empyema or intrapleural expansion of the leakage. [12,13]. Figure 1 shows an example of both a contained and an uncontained anastomotic leakage on CT-scan.
Treatment
After diagnosing AL, the local upper-GI surgeons made a therapeutic plan, tailored to each patient. Endoscopic, radiological and surgical strategies were described. Endoscopic treatment included suction-drainage, stenting and/or clipping of the defect. Radiological treatment included ultrasound - or CT-guided drainage; surgical approaches chosen were Video-Assisted Thoracoscopic Surgery (VATS) or thoracotomy. The type of surgical treatment performed was diverse, varying from drainage of the thorax, suturing of the defect and/or covering of the defect with muscle flaps.
The initial treatment-strategy chosen after diagnosis of AL was recorded and used to allocate patients to the surgical or endoscopic group. Patients who initially underwent a combined endoscopic and surgical approach were allocated to the surgical-group. Patients who were treated without surgical of endoscopic reinterventions (ECCG score 1) were excluded from the analysis.
Outcome parameters
Endpoints were the number of reinterventions (endoscopic, radiologic and surgical) needed during treatment, readmission to the ICU (no fixed protocol for readmission to the ICU), ICUand hospital stay, time to restart oral feeding, defined as thick liquids or more (yoghurt, soup etc), mortality and success/failure of treatment modality. Success of treatment was defined as clinical and biochemical improvement with downsizing to less invasive treatment according to the Clavien Dindo score. Failure of the treatment was defined as the necessity of additional reinterventions due to no clinical improvement, radiological deterioration or death. Upgrading of the invasiveness of therapy according to the Clavien Dindo score, was scored as failure as well.
For example a patient who underwent endoscopic treatment for AL, but developed empyema during the following days requiring surgery, was considered as a failed endoscopic treatment. A patient who developed repeated empyema for which repeated surgery was required, was considered as a failed surgical treatment. Additional interventions to accelerate healing of the leakage (e.g. the endoscopic placement of a clip or a stent in the final phase with the intention to accelerate closing of the defect after previous primary endoscopic therapy) or to replace a dislocated nasogastric tube were not considered as failure of the primary treatment.
Statistical analyses
Analyses were performed using Statistical Package for the Social Sciences version 22 (SPSS Inc., Chicago, US) and a difference was regarded significant at p<0.05. Descriptive statistics were applied for the patient characteristics and peri - and postoperative data: categorical variables were described as a number with corresponding percentage, continuous variables were described as median with interquartile range.
Analyses were performed for both the patients with cAL as with uAL; both groups were divided based on the primary treatment, which was either endoscopic or surgical. To compare these groups, the chi-square test and Fisher’s exact test were used for categorical variables when appropriate and the MannWhitney U test was used for continuous data.
Results
Within the indicated period of 5.5 years and among 680 esophagectomies, in total 73 patients were identified with an AL after ILE in the 3 participating hospitals. The number of patients that developed cAL and uAL were evenly distributed among the different hospitals (Table 1). The treatment strategy differed significantly. Two hospitals (A and B) had a less invasive strategy with the use of radiologic and endoscopic treatment, whereas hospital C performed more reoperations in case of AL.
Table 1:
|
Hospital A |
Hospital B |
Hospital C |
p-value |
Leakage |
18 (69) |
10 (40) |
11 (50) |
0.104 |
Treatment |
14 (54) |
20 (80) |
5 (23) |
<0.001 |
Baseline characteristics regarding tumour stage, co-morbidities and neo-adjuvant treatment of these patients and the procedures performed were similar in both groups, except the procedure time, which was significantly shorter in patients with uAL (supplementary table 1).
Contained leakage
Thirty-nine patients developed cAL after ILE. An endoscopic approach was chosen in 25 patients (64%) as primary treatment and was successful in 19 patients (76%). In 6 patients (24%) the endoscopic treatment failed; five patients underwent additional surgery of whom 2 patients deceased, one patient deceased without reoperation.
In total, 14 patients (36%) underwent a primary surgical approach that was successful in 11 patients (79%). This approach failed in 3 patients (21%) and they underwent repeated surgery, two of these patients deceased. In the endoscopic group, 11 patients (44%) were still admitted on the ICU at the time of diagnosis of the leakage and 2 patients (8%) were readmitted after diagnosis. In the surgical group, 12 patients (86%) were still admitted and 2 patients (14%) were readmitted (p=0.003).
The ICU stay, hospital stay and time to restart oral intake were similar in both groups.
In total 5 patients (12.8%) deceased as a result of the AL in the contained group.
Three of these patients underwent an endoscopic approach for the AL. Despite treatment, these three patients (12%) deteriorated fast and the leak progressed to an uncontained leak for which surgery was performed in two patients. Despite surgery, one of these patients developed an esophagobronchial fistula for which a stent was placed. Unfortunately, both patients deceased shortly after the surgery. The third patient developed a severe septic shock and died despite maximal support on the ICU.
Two patients in the surgical treatment group deceased as well (14.2%). The first patient was morbidly obese with severe pulmonary comorbidity. The patient developed clear pleural effusion that could not be drained radiologically due to obesity, and therefore underwent VATS with drainage. Despite this, the patient developed a severe septic shock as a result of an uncontained leak for which multiple surgical procedures were performed. The patient deceased four months postoperatively on the ICU.
The other patient underwent suturing of the defect and hereafter developed deep sepsis as a result of an uncontained leak. Despite several surgical interventions, the patient deceased one month postoperatively.
Uncontained leakage
Thirty-four patients developed uAL after IL. In 4 patients, clinical deterioration in combination with digestive tract excretion from the thoracic drains led to direct reoperation without diagnostic imaging. These patients were classified as uAL as well.
A primary endoscopic approach was chosen in 14 patients (41%) and was successful in 10 patients (71%). In 4 patients (29%) the endoscopic treatment failed and they had to undergo reoperation.
In total, 20 patients (59%) underwent a primary surgical approach and this was successful in 12 patients (60%). Eight patients (40%) failed the treatment and underwent repeated surgery.
In the endoscopic group, 4 patients (29%) were still admitted on the ICU at the time of diagnosis of the leakage and 4 patients (29%) were readmitted after diagnosis. In the surgical group, 13 patients (65%) were still admitted and 6 patients (30%) were readmitted (p=0.012).
The total amount of reinterventions, ICU stay, hospital stay and time to restart oral intake were similar as well. There was no mortality in this cohort.
Table 2:Characteristics of contained anastomotic leakages, treatment and its outcomes
|
Endoscopic N=25 |
Surgical |
p-value |
Total amount of interventions |
4 (3-8) |
4 (2-9) |
0.613 |
Additional interventions |
5 (20) |
3 (21) |
1.000 |
Type of endoscopic intervention~ |
22 (88) |
7 (50) |
0.019 |
Type of surgical intervention* |
4 (16) |
10 (72) |
0.640 |
Success of the treatment |
19 (76) |
11 (79) |
0.855 |
(Re)admission ICU |
13 (52) |
14 (100) |
0.003 |
ICU stay in days |
7 (2-15) |
12 (10-31) |
0.058 |
Hospital stay in days |
35 (24-56) |
32 (25-77) |
0.460 |
Time to oral intake in days |
33 (22-62) |
32 (19-47) |
0.645 |
Mortality |
3 (12) |
2 (14) |
1.000 |
Data are presented as number (percentage) or median (Interquartile Rate).
ICU: Intensive Care Unit
~ All endoscopic interventions used are scored.
*In patients who received several surgical reinterventions, the last surgical intervention was scored.
Table 3: Basic characteristics of the patients and procedures
|
Contained leakage |
Uncontained leakage |
p-value |
Age, in years |
66 (59-75) |
65 (60-67) |
0.273 |
Gender |
33 (85) |
30 (88) |
0.742 |
ASA classification |
4 (10) |
1 (3) |
0.577 |
cTNM Stage |
7 (18) |
6 (18) |
0.952 |
Tumor type |
33 (85) |
31 (91) |
0.616 |
Neoadjuvant treatment |
36 (92) |
29 (85) |
0.556 |
Procedure time, in minutes |
315 (260-360) |
272 (235-310) |
0.021 |
Blood loss, in milliliters |
100 (80-200) |
200 (100-300) |
0.097 |
Discussion
This retrospective multicentre cohort study analysed 73 patients with AL after ILE. Standard-of-care strategies were analysed for the treatment of AL in three large referral hospitals. Upon diagnosis, AL was classified as either contained or uncontained and the treatment strategy was divided into endoscopic vs. surgical. In accordance with literature, this cohort of patients with AL suffered from significant morbidity and mortality and adequate treatment often required reinterventions with prolonged ICU- and hospital stay.
The majority of the 39 patients with a cAL underwent a primary endoscopic treatment (N=25, 64%) that was successful in 19 patients (76%). This complies with the results by Crestanello en Guo showing that cAL can often be treated nonoperatively [12,13]. The remaining 14 patients (36%) underwent a primary surgical approach for a cAL, which was successful in 11 patients (79%). Since this was not a randomised study, treatment of AL was based on the preference of the upper GI teams in the three hospitals. Besides local preference, the condition of the patient could have influenced the decision for an endoscopic or surgical approach. More patients were admitted in the ICU at the time of diagnosis when a surgical approach was chosen, which suggests that patients who underwent reoperation might have been more seriously ill than patients who underwent an endoscopic treatment. This is also supported by a trend to a longer ICU-stay in the surgical-group. However this could also be a sign of the pathophysiological impact of the surgical approach per se, necessitating ICU treatment. It is unlikely that progression from a cAL to a uAL between diagnosis and surgery has increased the amount of admission in the ICU, since most patients (72%) underwent surgery within 24 hours after diagnosis of the leak.
A different pattern of care was observed in the patients with an uAL; in the majority of these patients (N=20, 59%) the primary treatment strategy was surgery, with a success rate of 60%. In the remaining 14 patients (41%) a primary endoscopic approach was chosen, combined with (radiologically guided) drainage of the intrathoracic collections. Ten out of 14 leaks (71%) could be managed successfully this way. In our surgery group, similar outcomes were observed as seen in patients who underwent surgery for a cAL; significantly more patients were still admitted on the ICU at the day of diagnosis and there was a trend towards a longer ICU- and hospital stay, which probably implies that patients in the surgical-group were more ill than those in the endoscopic-group.
There are several endoscopic techniques for treating AL after esophagectomy, like suction-drainage, stenting and/or clipping of the defect. The most important advantage of endoscopic treatment is that it is less invasive than surgery with a lower post procedural morbidity and mortality [8,10,15]. Furthermore, as opposed to an endoscopic approach, a transthoracic surgical approach by definition means spread of a contained leak into the thoracic cavity. However, nasogastric tubes for suction drainage and stents tend to dislocate easily and a stent must be removed after successful treatment, leading to repeated endoscopies as shown in our cohort. Although endoscopy is less invasive than surgery, repeated endoscopies can be a burden for patients as well. Moreover, intrathoracic manifestations of an AL cannot be treated with an endoscopic procedure alone, but should be combined with external drainage of the thoracic empyema or abscesses. These thoracic collections can be treated with percutaneous radiological techniques with increasing success rates and with lower morbidity and mortality than surgery. [16,17]
On the other hand, there are several surgical techniques in the treatment of AL as well. In case of a small, very early leak due to technical failure, the defect can be sutured. In case of a larger defect, a new anastomosis can be made or a muscle flap can be used to cover the defect. In most cases, however, the primary goal of a surgical approach is drainage of the thoracic empyema and containment of the leak.
It is striking that a total of 5 patients from the contained group died as a result of the AL, compared to no mortality in the uncontained group. In all of these 5 patients, the contained leak progressed to an uncontained leak despite surgical or endoscopic treatment. This indicates that an AL is a dynamic process, in which a ‘simple’ leak may progress into a ’complicated’ leak despite treatment. Therefore it remains essential to monitor the patient closely and regularly evaluate the result of the initiated treatment. It is important to immediately reassess a patient with the slightest deterioration, using endoscopy and/ or CT-scan, to assess the changes and act accordingly.
The classification of leakages into contained and uncontained might assist in the choice for a treatment-strategy. Ideally, we would have constructed a treatment algorithm in which we combined the type of leakage with the size of the leakage, the vitality of the gastric conduit and the clinical condition of the patient. One could imagine that a small, contained leak in a relatively fit patient can be treated endoscopically, and an aggressive surgical approach might be the first choice in case of a large, uncontained leak in a severely ill patient. Unfortunately however, we were not able to make such an algorithm due to lack of information about the size of the leakage and the condition of the patients in combination with a relatively small cohort. International data are now gathered in order to perform analyses in a very large cohort of over 1000 patients in order to develop a clinically relevant gradation and a treatment algorithm for AL. (www.Tentaclestudy.com).
Strength of the current study is that it reflects the daily practice of treatment of AL in three high-volume hospitals. It is the largest series in a single study to date and provides insights into inter-hospital variation since there was no uniform multicentre fixed treatment protocol.
An important limitation of the study is the heterogeneity between the patients and the institutions.
Local preferences of the hospital may also have contributed to the choice of treatment type, which is reflected in the significant differences in treatment strategy between the three hospitals. The expertise of endoscopic diagnosis and available treatment modalities of AL might differ between endoscopists in the hospitals, which might have influenced the choice of treatment. In addition, the surgeons’ personal preferences as well as the lack of evidence for the superiority of either endoscopic or surgical treatment could have influenced the decision for the treatment strategy as well.
The patient’s pathophysiological response to the leak might also have influenced the choice of the primary treatment. In this retrospective study it was not possible to determine the severity of the illness at time of diagnosis, and therefore the ICU-(re)-admission rate was used as a surrogate marker for the degree of illness. Ideally, we would have used the Modified Early Warning Score (MEWS), which includes fever, respiratory failure, hypotension and tachycardia into an illness score, to make an inventory of the degree of illness, since this quantitative score represent the level of sickness much better [18]. However, these data were not consistently registered and could therefore not be used for analysis.
The choice of surgical or endoscopic treatment is probably influenced by endoscopic findings. The endoscopic features of the AL, such as the vitality of the anastomosis and the gastric tube and the extensiveness of the anastomotic breakdown might have strongly influenced the decision making as well. Unfortunately, many endoscopy reports were found to be inadequate and lacked relevant information. Therefore, we were unable to further classify the leaks endoscopically in addition to the CT-images.
Conclusion
In conclusion, this retrospective cohort gives an insight in multiple approaches towards treating AL that complicated ILE. By classifying a leak as either contained or uncontained, treatment strategies and their successes can be compared in larger randomized cohort studies in the future. In our multicenter cohort, the endoscopic approach appears to be successful for the majority of patients with a contained leak and it could be considered in a selection of patients with an uncontained leak. However, it appears that more parameters influence the decision for a treatment strategy than just the type of leakage, necessitating more research before a treatment algorithm can be developed.
Declarations
Acknowledgements: We thank dr. Frits van den Wildenberg, Canisius Wilhelmina Hospital Nijmegen, the Netherlands, for the collected data.
Conflict of interest statement: Drs. F van Workum reports a metronic grant outside the submitted work.
Dr. M Luyer reports grants from Medtronic and Galvani outside the submitted work.
Dr. Nieuwenhuijzen reports personal fees from Medtronic and grants from the Dutch Cancer Foundation outside the submitted work.
Drs. M Lubbers, Dr G Berkelmans, Dr. C Rosman, Dr. MJ van Det and Dr. EA Kouwenhoven declare that they have no conflict of interest
References
- Rutegard M, Lagergren P, Rouvelas I, Lagergren Jl. Intrathoracic anastomotic leakage and mortality after esophageal cancer resection: A population-based study. Ann Surg Oncol. 2012; 19: 99-103.
- Junemann-Ramirez M, Awan MY, Khan ZM, Rahamim JS. Anastomotic leakage post-esophagogastrectomy for esophageal carcinoma: Retrospective analysis of predictive factors, management and influence on longterm survival in a high volume centre. Eur J Cardiothorac Surg. 2005; 27: 3-7.
- Schweigert M, Solymosi N, Dubecz A, González MP, Stein HJ, Ofner D. One decade of experience with endoscopic stenting for intrathoracic anastomotic leakage after esophagectomy: Brilliant breakthrough or flash in the pan? Am Sur. 2014; 80: 736- 745.
- Alldinger I, Sisic L, Hochreiter M, Weichert W, Blank S, Burian M et al. Outcome, complications, and mortality of an intrathoracic anastomosis in esophageal cancer in patients without a preoperative selection with a risk score. Langenbecks Arch Surg. 2015; 400: 9-18.
- Larburu Etxaniz S, Gonzales Reyna J, Elorza Orue JL, Gallego JIA, del Val IS, Letamendia EE et al. Cervical anastomotic leak after esophagectomy: Diagnosis and management. Cir Esp. 2013; 91: 31-37.
- Low DE, Alderson D, Cecconello I, Chang AC, Darling GE, D’Journo XB et al. International consensus on standardization of data collection for complications associated with esophagectomy: Esophagectomy complications consensus group (ECCG). Ann Surg. 2015; 262: 286-294.
- Freeman RK, Vyverberg A, Ascioti AJ. Esophageal stent placement for the treatment of acute intrathoracic anastomotic leak after esophagectomy. Ann Thorac Surg. 2011; 92: 204-8.
- Schniewind B, Schafmayer C, Voehrs G, Egberts J, von Schoenfels W, Rose T et al. Endoscopic endoluminal vacuum therapy is superior to other regimens in managing anastomotic leakage after esophagectomy: A comparative retrospective study. Surg Endosc. 2013; 27: 3883-3890.
- Mennigen R, Harting C, Lindner K, Vowinkel T, Rijcken E, Palmes D et al. Comparison of endoscopic vacuum therapy versus stent for anastomotic leak after esophagectomy. J Gastrointest Surg. 2015; 19: 1229-1235.
- Shuto K, Kono T, Akutsu Y, Uesato M, Mori M, Matsuo K et al. Naso-esophageal extraluminal drainage for postoperative anastomotic leak after thoracic esophagectomy for patients with esophageal cancer. Dis Esophagus. 2017; 30: 1-9.
- Verstegen MHP, Bouwense SAW, van Workum F, ten Broek R, Siersema P, Rovers M et al. Management of intrathoracic and cervical anastomotic leakage after esophagectomy for esophageal cancer: A systematic review. World J Emerg Surg. 2019; 14: 17-019-0235-4.
- Crestanello JA, Deschamps C, Cassivi SD, Nichols FC, Allen MS, Schleck C et al. Selective management of intrathoracic anastomotic leak after esophagectomy. J Thorac Cardiovasc Surg. 2005; 129: 254-260.
- Guo J, Chu X, Liu Y, Zhou N, Ma Y, Liang C. Choice of therapeutic strategies in intrathoracic anastomotic leak following esophagectomy. World J Surg Oncol. 2014; 12: 402-7819-12-402.
- van Hagen P, Hulshof MC, van Lanschot JJ, Steyerberg EW, van Berge Henegouwen MI, Wijnhoven BPL et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012; 366: 2074-2084.
- Nguyen NT, Rudersdorf PD, Smith BR, Reavis K, Nguyen XM, Stamos MJ. Management of gastrointestinal leaks after minimally invasive esophagectomy: Conventional treatments vs. endoscopic stenting. J Gastrointest Surg. 2011; 15: 1952-1960.
- Yu H. Management of pleural effusion, empyema, and lung abscess. Semin Intervent Radiol. 2011; 28: 75-86.
- Vaziri M, Abed O. Management of thoracic empyema: Review of 112 cases. Acta Med Iran. 2012; 50: 203-207.
- Gardner-Thorpe J, Love N, Wrightson J, Walsh S, Keeling N. The value of modified early warning score (MEWS) in surgical in-patients: A prospective observational study. Ann R Coll Surg Engl. 2006; 88: 571-575.