Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
En bloc vertebrectomy of a thoracic Ewing sarcoma including posterior arch: 3 years later
Mouadh Nefiss1*, Mohamed Amine Gharbi1; Saker Ayari1; Ramzi Bouzidi1; Sana Ben Slama2; Khelil Moncef Ezzaouia¹; Anis Tebourbi1
1 Faculty of Medicine of Tunis, Department of Orthopedic Surgery, Mongi Slim Marsa University Hospital, University of Tunis El Manar, Tunis, Tunisia
2Department of Anatomical Pathology, Mongi Slim Marsa University Hospital, Tunis, Tunisia
*Corresponding Author: Anthony Djaba Sackey
Ant Marine W A Consulting Services, Koforidua,
Ghana.
Email: deckcadetsackey09@gmail.com
Received : Sep 28, 2021
Accepted : Nov 19, 2021
Published : Nov 26, 2021
Archived : www.jcimcr.org
Copyright : © Nefiss M (2021).
Abstract
Ewing’s sarcoma of the mobile spine is a very rare pathology, especially after 30 years of age. The clinical manifestations and radiological aspects can be confusing, which delays the diagnosis. Approximately 25% of patients present with metastatic disease at diagnosis which worsen the prognosis. In localized forms the therapeutic goal is a surgical en bloc wide resection of the tumor, a restoration of spinal column stability and avoidance of recurrence through multimodal therapy including chemotherapy and radiotherapy. We report a case of primary Ewing´s sarcoma of 12th thoracic vertebra in 30 years old man treated with chemotherapy and a double approach en bloc vertebrectomy. The reconstruction was performed with anterior bone grafting and posterior stabilization. No tumor recurrence was observed at the 3 year follow-up assessment. Imaging and clinical aspects were analyzed as well as the management modalities and outcome.
Keywords: Ewing sarcoma; thoracic spine; surgery; En bloc vertebrectomy.
Abbreviations: EWS: Ewing’s Sarcoma; T12: 12th Thoracic Vertebra; WBB: Weinstein Boriani Biagini; MRI: Magnetic Resonance Imaging; CT: Computerized Tomography
Citation: Sackey AD, Sackey AD, John B, Lee-Duah RO. A look into “Outlining the challenges of Covid-19 health crises in Africa’s maritime industry: The case of maritime operations in marine warranty surveying practice”: Discussing ‘three pillar challenge’. J Clin Images Med Case Rep. 2021; 2(6): 1429.
Introduction
Primary Ewing’s Sarcoma (EWS) of the spine is extremely rare. It accounts for only 3.5% to 14.9% of all primary bone sarcomas [1,2]. It is characterized by a high proliferative and invasive potential and a confusing variety of imaging manifestations in adult patients [3]. Non-specific signs are often in the first place and the delay from the onset of symptoms to the diagnosis can take months which can worsen the prognosis [4]. Because of the rarity of the spinal localization of primary EWS in adults there is not a well-coded management protocol and a multitude of therapeutic strategies that mimic the management of EWS of the appendicular skeleton have been employed. The therapeutic goal is a complete tumor removal and spinal column stability restoration [1]. The publications of Weinstein, Boriani and Biagini and their surgical staging system (WBB) have been of great interest in this field [5,6]. We report a rare localization of solitary thoracic EWS in an adult patient that was treated with en bloc vertebrectomy.
Case report
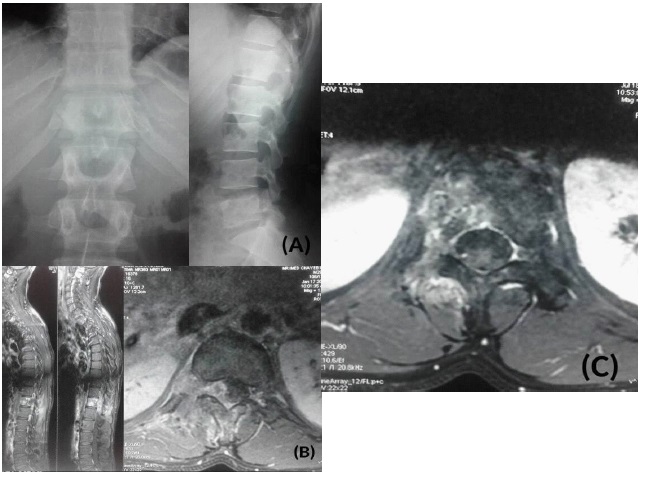
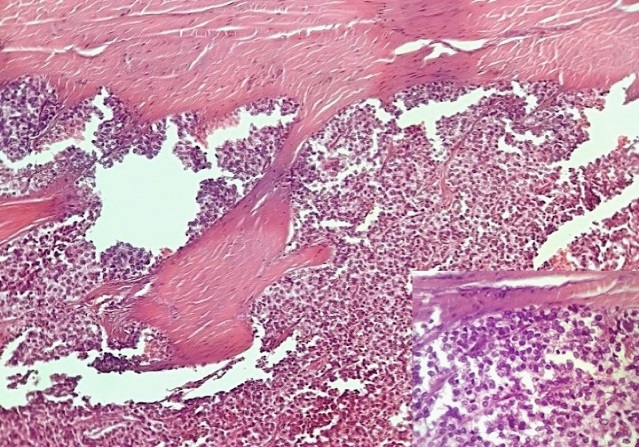
A 30-year-old man presented with a one-year history of a progressive, worsening back pain associated to a right intercostal neuralgia, no motor weakness or sphincter disturbance were found. There was no history of fever or trauma. Blood tests were normal and the X-ray rechecks after making the diagnosis showed an unobvious suspect image at the level of the right pedicle of the 12th thoracic vertebra (T12) with disappearance of its normal contours (Figure 1A). Magnetic resonance imaging (MRI) revealed a vascularized mass affecting the right hemivertebral body and posterior arch of T12 with paravertebral soft tissue extension. The spinal cord was mildly compressed (Figure 1B). An extension assessment concluded that it is a primary localization. We performed a Computerized Tomography (CT) guided biopsy and the diagnosis of EWS was confirmed by histopathology and immunohistochemistry showing uniformly small round cells which were strongly positive to CD99 (Figure 2).
After receiving neo-adjuvant chemotherapy, a new MRI showed vertebral and paravertebral soft tissue mass regression (Figure 1C). According to the WBB surgical staging system, sectors included in the axial T12 imaging were 1 to 6 (part of the body and part of the left posterior arch) and layers of tissue penetration were A to D.
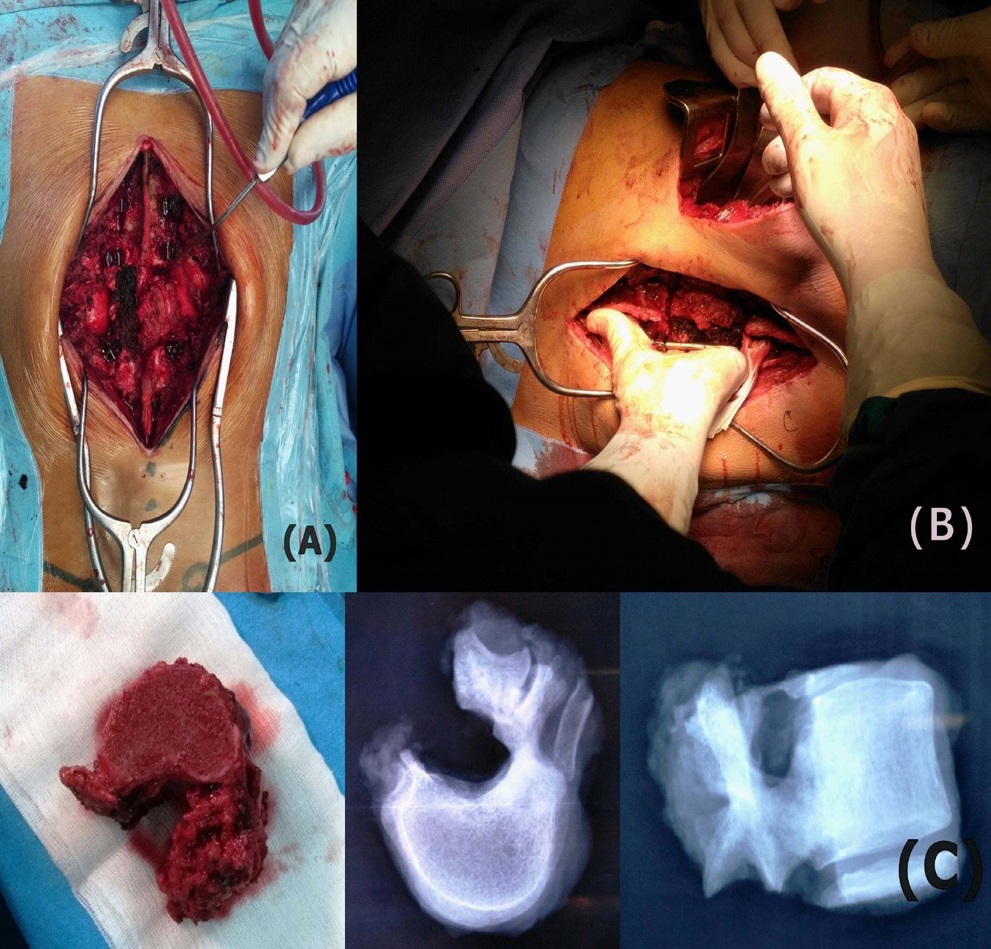
We performed an en bloc vertebrectomy through a double approach surgery: firstly a posterior approach with pedicle screw insertion (2 levels above and 2 levels below), remove of the healthy part of the posterior arch which allow to release the spinal cord from the tumor pseudocapsule , ligation and section of T12 roots (Figure 3A). Secondly a thoraco-phreno-lombotomy to separate the anterior part of the tumor and control the segmental artery. The separation was made by a Gigli saw and the extraction of the vertebra released from all its attachments was possible through a simple rotation of the specimen (Figure 3B and 3C). Reconstruction was performed with interbody free non-vascularized fibular bone grafting stabilized by the posterior instrumentation.
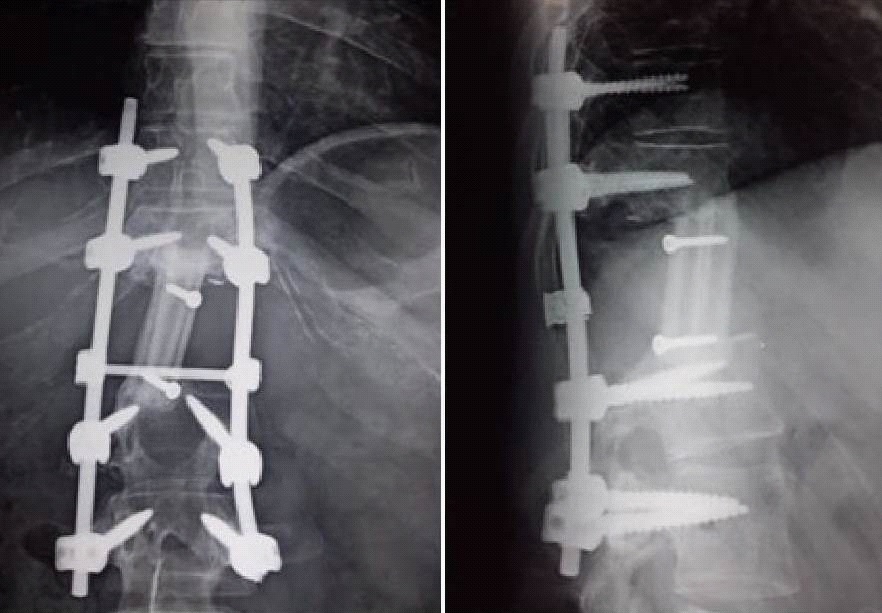
Postoperative course was uneventful. After six weeks of the surgery, the patient started adjuvant chemotherapy and completed treatment with no evidence of disease on re-evaluation at the end of therapy. Three years after surgery, the patient is surviving without neurologic deficit, X-rays revealed consolidation (Figure 4) and with no evidence of recurrence on MRI.
Discussion
Ewing's sarcoma remains an enigmatic and particular malignant tumor 100 years after its discovery. Spinal involvement most commonly results from metastasis in advanced stages of the disease, while EWS originating from the spine is rare and extremely rare if the sacrum is excluded [2,3]. Diagnosis could be quite difficult due to an insidious onset, non-specific symptoms and misinterpreted images. Thus, symptoms may not be present until neurological deficits occur and diagnosis suspicion may be after several consultations which increase doubts [7,8]. The average delay from the onset of symptoms to the diagnosis has been reported to be 34 weeks all locations combined [9]. In our case the delay was 48 weeks. That’s why approximately 25% of patients present with metastatic disease at diagnosis; fortunately this was not the case of our patient [3].
Definitive diagnosis requires cytological, immunohistochemical (CD99) and cytogenetic analysis of a pathologic specimen.The translocation involving chromosome 22 is identified in more than 90% of cases, and it is the landmark to differentiate EWS from other small, round blue cell tumors [3,10].
Currently, early diagnosis and multimodal treatment combining surgery, chemotherapy, and local radiation therapy increases the chance of a successful outcome [11]. Indelicato et al [12], in a review have reported a five-year overall survival rate of 71% and local control rate of 89% for non-metastatic spinal and paraspinal EWS. However, when compared to other sites of occurrence, prognosis of EWS of the spine remains worse [13].
Initial chemotherapy and local radiotherapy might be administered before surgery with the aim to shrink bulky and unresectable tumors, to eradicate micrometastases and for acute relief of epidural compression, but it should be noted that there is a variable sensitivity to radiation and chemotherapy due to biological heterogenecity [1].
During the last two decades, the outcome in patients with localized disease has improved through an aggressive surgery known as en bloc vertebrectomy by combining anterior and posterior approach or from a single posterior approach as described by Tomita and al [14].
Boriani et al [6] described three major methods of performing en bloc excisions in the thoracolumbar spine: vertebrectomy if the tumor is confined to zones 4 to 8 or 5 to 9; sagittal resection when the tumor occupies zones 3 to 5 or 8 to 10 and resection of the posterior arch when it is located between the zones 10 and 3 according to their surgical staging system.
However, en bloc resection is a highly demanding procedure that must be carefully planned and the greater surgical risk can be accepted only if it offers a safer result and is performed by specialized surgical and anesthesiology teams [5].
Among the difficulties to be considered is the release of the spinal cord if the tumor is expanding to layer D according to WBB staging system. In this case theoretical safe margin should include the dura in the resection specimen but the cost-to-benefit ratio of such procedure should be carefully evaluated. Boriani and his collaborators have shown that a simple release of the dura without resection can be accepted and without proven consequence on the risk of recurrence [6,13].
Compared to cases where only decompression or lesionectomy was done, patients who underwent en bloc spondylectomy had a lower recurrence rate [8,15]. This is why, whenever possible, surgical en bloc wide resection with an anterior column reconstruction is preferable in order to obtain a better oncological control and a better preservation of the spine biomechanics [3].
Conclusion
Primary Ewing’s sarcoma of the thoracic spine is an extremely rare tumor. It is a challenging disease not only to treat but also to diagnosis. A high index of suspicion is needed in patients who present with few or non-specific symptoms. Early diagnosis is essential to obtain better results and improve prognosis. In localized forms of the tumor multi modal treatment involving neo-adjuvant chemotherapy, en bloc vertebrectomy and adjuvant radiotherapy and chemotherapy is required for patient outcome and satisfactory quality of life.
Declarations
Acknowledgements: The authors declare no conflict of interest.
References
- Dini LI, Mendonça R, Gallo P. Primary Ewings sarcoma of the spine: case report. Arq Neuropsiquiatr. 2006; 64: 654‐659.
- Goktepe AS, Alaca R, Mohur H, Coskun U. Paraplegia: an unusual presentation of Ewing’s sarcoma. Spinal Cord. 2002; 40: 367-369.
- Iacoangeli M, Dobran M, Di Rienzo A, Di Somma LGM, Moriconi LAE, Nocchi N et al. Nonmetastatic Ewing’s Sarcoma of the Lumbar Spine in an Adult Patient. Case Rep Oncol Med. 2012; 2012: 165289
- Nair M, Sukumaran Nair R, Raghavan R, Parukkutty K, Sukumaran R. Primary Ewing’s Sarcoma of the Spine in Pediatric Patients: A Case Series Analysis and Literature Review. Middle East Journal of Cancer. 2015; 6: 115-20.
- Boriani S, Biagini R, De Iure F, Andreoli I, Campanacci L, De Fiore M et al. Primary bone tumors of the spine: a survey of the evaluation and treatment at the Istituto Ortopedico Rizzoli. Orthopedics. 1995; 18: 993‐1000
- Boriani S, Weinstein JN, Biagini R. Primary bone tumors of the spine. Terminology and surgical staging. Spine (Phila Pa 1976). 1997; 22: 1036‐1044.
- Ilaslan H, Sundaram M, Unni KK, Dekutoski MB. Primary Ewing’s sarcoma of the vertebral column. Skeletal Radiol. 2004; 33:506-513.
- Weinstein JB, Siegel MJ, Griffith RC. Spinal Ewing sarcoma: misleading appearances. Skeletal Radiol. 1984; 11: 262‐265.
- Widhe B, Widhe T. Initial symptoms and clinical features in osteosarcoma and Ewing sarcoma. J Bone Joint Surg Am. 2000; 82: 667-674.
- Turc-Carel C, Philip I, Berger MP, Philip T, Lenoir GM. Chromosome study of Ewing’s sarcoma (ES) cell lines. Consistency of a reciprocal translocation t(11;22)(q24;q12). Cancer Genet Cytogenet. 1984; 12: 1‐19
- Sharafuddin MJ, Haddad FS, Hitchon PW, Haddad SF, el-Khoury GY. Treatment options in primary Ewing’s sarcoma of the spine: report of seven cases and review of the literature. Neurosurgery. 1992; 30: 610‐619.
- Indelicato DJ, Keole SR, Shahlaee AH, Morris CG, Gibbs CP, Scarborough MT et al. Spinal and paraspinal Ewing tumors. Int J Radiat Oncol Biol Phys. 2010; 76: 1463‐1471.
- Boriani S, Amendola L, Corghi A, Cappuccio M, Bandiera S, Ferrari S et al. Ewing’s sarcoma of the mobile spine. Eur Rev Med Pharmacol Sci. 2011; 15: 831‐839.
- Tomita K, Kawahara N, Baba H, Tsuchiya H, Fujita T, Toribatake Y. Total en bloc spondylectomy. A new surgical technique for primary malignant vertebral tumors. Spine (Phila Pa 1976). 1997; 22: 324-333.
- Samartzis D, Marco RA, Benjamin R, Vaporciyan A, Rhines LD. Multilevel en bloc spondylectomy and chest wall excision via a simultaneous anterior and posterior approach for Ewing sarcoma. Spine (Phila Pa 1976). 2005; 30: 831‐837.