Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Research Article - Open Access, Volume 2
Geographic variation of antibiotic resistance in E. coli isolated from drinking water and foodstuffs in Sylhet division of Bangladesh
Belal Ahmad; Arifur Rahaman; Sadia Sinty Disha; Ashik Imran; Rashid Taqui; Nishat Tasnim Nousin; Shamim Mahmud; SM Kamruzzaman; Shaikh Mirja Nurunnabi*
Department of Biochemistry and Molecular Biology, Shahjalal University of Science and Technology, Sylhet-3114, Bangladesh
*Corresponding Author: Shaikh Mirja Nurunnabi
Department of Biochemistry and Molecular Biology,
Shahjalal University of Science and Technology,
Sylhet-3114, Bangladesh.
Email: shaikhmir-bmb@sust.edu
Received : Oct 06, 2021
Accepted : Nov 23, 2021
Published : Nov 30, 2021
Archived : www.jcimcr.org
Copyright : © Nurunnabi SM (2021).
Abstract
Geographical variation of antibiotic resistance (ABR) is a great difficulty for the empirical antibiotic’s prescription for infectious diseases. The development of ABR in bacteria is believed in associated with some risk factors. In addition, ABR against antibiotic drugs due to its associated risk factors has been linked with various negative health outcomes. However, almost no information is available in relation to such issues of bacteria isolated from environmental samples in Bangladesh. This study first time describes that information’s of E. coli isolated from foodstuffs and drinking water in urban and rural areas in Bangladesh. We isolated E. coli from 72 samples (urban 36 and rural 36) from foodstuffs (rice, carrot and tomato) and drinking water (filtered water, tea stall water and tube well water) in Sylhet division, and tested the antibiogram by disc diffusion and spread plate methods against 14 commonly used antibiotics. Sociodemographic data by using a structured questionnaire were collected from 577 participant’s child, adolescent and old in urban and rural areas. In this study, we found the occurrence of E. coli isolated from foodstuffs and drinking water is 33.33% and 11.11% in urban and rural areas respectively. This study represents E. coli is resistant (100%) to AMP, AMX, E and P in both the urban and rural areas. However, CTX (83%), C (100%), COT (33%), GEN (83%), DO (17%), AK (83%) and NA (50%) are resistant only in the urban but not rural area. On the other hand, AZM (100%), CIP (100%) and TE (100%) are completely sensitive to E.coli in both the urban and rural areas whereas CTX (100%) is only to rural E.coli. Importantly, multiple antibiotic resistance (MAR) of E.coli is significantly higher in urban than rural samples. Moreover, our data also indicates that ABR associated risk factors link with negative health outcomes in Sylhet division.
Keywords: antibiotic resistance; geographical variation; E.coli; drinking water; foodstuffs; risk factors; health outcomes.
Abbreviations: ABR: Antibiotic resistance; E. coli: Escherichia coli; ABRB: Antibiotic resistance bacteria; MAR: Multiple antibiotic resistance; MDR: Multi drug resistance; MIC: Minimum inhibitory concentration; LA: Luria agar; ZOI: Zone of inhibition; CLSI: Clinical laboratory standard institute; BOD: Biological oxygen demand; DO: Dissolved oxygen; CIP: Ciprofloxacin; AMP: Ampicillin; AMX: Amoxycillin; E: Erythromycin; AK: Amikacin; GEN: Gentamicin; C: Chloramphenicol; COT: Co-trimoxazole; NA: Nalidixic acid; AZM: Azithromycin; P: Penicillin; CTX: Cefotaxime; TE: Tetracycline; DO: Doxycycline.
Citation: Ahmad B, Rahaman A, Disha SS, Imran A, Taqui R. Geographic variation of antibiotic resistance in E. coli isolated from drinking water and foodstuffs in Sylhet division of Bangladesh. J Clin Images Med Case Rep. 2021; 2(6): 1439.
Introduction
Antibiotic resistance (ABR) has turned into a severe global health crisis and yet people are not completely aware of the threat that poses at the individual level as well as the community level [1-3]. Remarkly, the ABR costs are comparatively high in the developing countries where the healthcare system is not as advanced. As a result, ABR causes significant morbidity worldwide, and some estimates indicate attributable mortality could reach up to 10 million by 2050 [1-6]. Accordingly, bacterial ABR is now becoming main hindrance to use antibiotic in treating infectious disease in middle income country Bangladesh. Direct or indirect contact with drinking water and foodstuffs contaminated by antibiotic resistance bacteria (ABRB) could harm and infect the human population [7-10]. However, the presence and distribution of ABRB in a drinking water source, is always neglected due to strict legal protection and less anthropogenic activities. According to literatures, the context of ABR in drinking water sources is of serious grave concern [9,11-14]. Thus, considering the source of water and foodstuffs are directly related with human activity and health, understanding the prevalence of ABR in these sources is of great importance.
It is well known that ABR occurs as a natural process at low rates in bacteria [15]. In addition, ABR occurs via molecular mechanisms antibiotics efflux, enzymatic modifications of antibiotics, enzymatic breakdown of antibiotics and modification in target sites [1,2,6,16-18]. Thus, bacteria that cause infections in humans can develop or acquire resistance to antibiotics commonly used against them [5,19]. In Bacteria, the gradual development of ABR is mediated by diverse resistance genes that evolve as a result of antibiotic selection pressure exerted by the irrational overuse of antibiotics, misuse of antibiotics, selfmedication, ignorance of the common people to complete the course of antibiotics prescribed by the healthcare professionals and excessive use antibiotics in livestock’s, agriculture and food industry [2,3,5,15,17,19-24]. Additionally, ABR against antibiotic drugs due to its associated factors have been linked with various negative health outcomes infectious disease, chronic disease, respiratory disease, skin problem, severe allergies, hypersensitivity and risk of obesity, brain stroke, heart attack and cancer [25-35].
Escherichia coli (E. coli) a member of the faecal coliform group, has been used as an indicator of human enteric pathogens, infectious diseases and antimicrobial resistance [36-39]. Accordingly, E. coli is frequently associated with ABR that can survive well in aquatic environments, and is highly adept at horizontal gene transfer who is deemed as the vector for ABR dissemination [40, 41]. In many countries E. coli has shown that the variation of ABR, the associating factors of ABR and the negative health outcomes of ABR [5,20,21,23,28,34,42]. However, almost no information is available in relation to such issues of E.coli isolated from environmental samples in Bangladesh. This study first time describes that information’s of E. coli isolated from foodstuffs and drinking water in urban and rural areas of Sylhet division of Bangladesh. We determined the prevalence of E. coli and water quality in urban and rural areas. We investigated the antibiogram of E. coli isolated from foodstuffs and drinking water samples against 14 antibiotics commonly used in medical practices. Moreover, existences of regional differences in multiple antibiotic resistance (MAR) profiles will be estimated. We further examined the effects of water quality on MAR. Finally, we also studied the presence of possible link between factors associated with ABR and its negative health outcomes.
Material and methods
Sampling sites, samples collection, data collection and water quality data
From March 2018-February 2019, foodstuffs and drinking water were collected from urban and rural areas in Sylhet division. Foodstuffs (rice, tomatoes, and carrot) and drinking water (filter water, tap water and tea-stall water) were taken in this study. A structured questionnaire was used to collect data on socio-demographics (age, sex, education etc.), ABR associated factors, health diseases etc. from children, adolescent and old people in urban and rural areas. Data were entered in Excel file and generated graph by student’s t-test method. Water quality parameters were measured according to Bangladesh National Standard Methods. Briefly, water temperature (Thermometer method), pH (Glass electrode method), and dissolved oxygen (Electrochemical probe method) were measured in using a digital display thermometer (Ruiming, Changzhou, China), a pH meter (METTLER, Switzerland) and an YSI-58 instrument (Yellow Spring, USA) respectively in the Department of Geography and Environment, Shahjalal University of Science and Technology, Sylhet, Bangladesh.
Isolation of E. coli from foodstuffs and water samples and their antibiogram test
Membrane filtration method [43] was used to isolate E. coli from food stuffs and drinking water from urban and rural areas in Sylhet division. Briefly, a 100 ml aliquot of water from sample was filtered through a 0.22 μm membrane filter (Millipore, Carrightwohill, IRL) using a filter funnel and vacuum system (model, NC-10023). The membranes were placed on E. coli-Coliforms Chromogenic Mediums (Nissui, Qingdao, China) and incubated at 37◦C for 24 h. After incubation, blue colonies represented E. coli. Typical E. coli were picked and purified on the MacConkey Agar (Nissui, Qingdao, China).
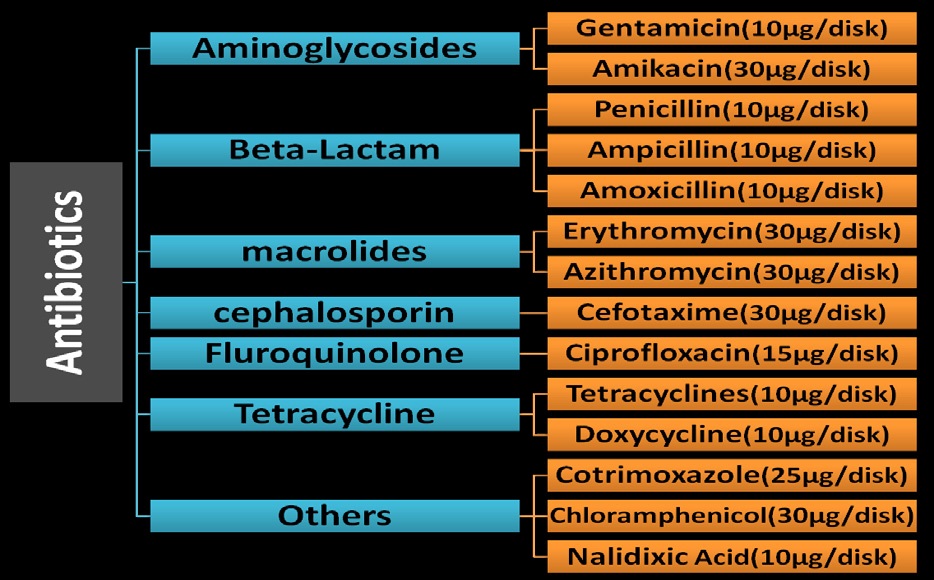
Antibiotic susceptibility testing for confirmed E. coli was conducted by the Kirby-Bauer disk diffusion and spread plate methods on Luria agar medium (Oxoid, Basingstoke, England) as recommended by Clinical Laboratory Standard Institute [44]. 14 (fourteen) different antibiotics (Figure 2) were used in this study ciprofloxacin (CIP), Ampicillin (AMP), Amoxicillin (AMX), Erythromycin (E), Amikacin (AK), Gentamicin (GEN), Chloramphenicol (C), Co-trimoxazole (COT), Nalidixic acid (NA), Azithromycin (AZM), PenicillinG (P), Cefotaxime (CTX), Tetracycline (TE) and Doxycycline (DO) (Oxoid, England). “Sensitive” “intermediate resistant” or “fully resistant” patterns of E. coli were judged using antibiotic minimum inhibitory concentration provided by the guidelines of the CLSI.
Disc diffusion method
This method was performed by using the antibiotic discs, which contain antibiotics at a particular concentration [45]. Ciprofloxacin, Ampicillin, Amoxicillin, Erythromycin, Amikacin, Gentamicin, Chloramphenicol, Co-trimoxazole, Nalidixic acid, Azithromycin, PenicillinG, Cefotaxime, Tetracycline and Doxycycline were used for this method. When the antibiotics are placed on the LA media with bacteria, it diffuses from the disc and spread outside the surrounding of the disc and produce circular zone. If the microorganism in the plate is sensitive, it will create a clear zone of circle around the disc on the other hand it will not create a clear zone which means the bacteria is resistant. Slide caliper was used to measure the zone of inhibition (ZOI) and categorized as Sensitive, Intermediate resistance and fully resistance.
Spread plate method
In spread plate method antibiotic solutions at specific concentration are mixed with the LA media. 13 different antibiotics were used in this method. In spread plate method bacterial colony was countered by colony counting device.
Results
Water quality of rural and urban drinking water
We measured water quality temperature, PH, BOD and DO in water from urban and rural samples (Table 1). We found that there are no remarkable changes of PH and temperature between urban and rural water. In addition, BOD value of rural water is considerably higher than unban water indicating polluted water in urban. Moderately polluted water has BOD value range of 2-8 mg/L. DO value was good in rural water than urban water. Healthy water should generally have DO concentrations above 6.5-8 mg
The prevalence and antibiogram of E. coli in urban and rural samples in Sylhet
We collected total 72 samples (n=36 from urban and n=36 from rural areas) from food stuffs (Tomato n=6, carrot n=6 and rice n=6 from refrigerator) and drinking water (tap water n=6, filtered water n=6 and tea stall water n=6) in Sylhet division. We found E. coli 12 (33.33%) and 4 (11.11%) in urban and rural samples respectively (Table 2). We tested antibiogram of 14 commonly used antibiotics by disc diffusion and spread plate (data not shown) methods in E.coli isolated from Carrot, tomato (data not shown), rice (data not shown), tea stall water, tap water (data not shown) and filtered water (data not shown) in urban and rural areas.
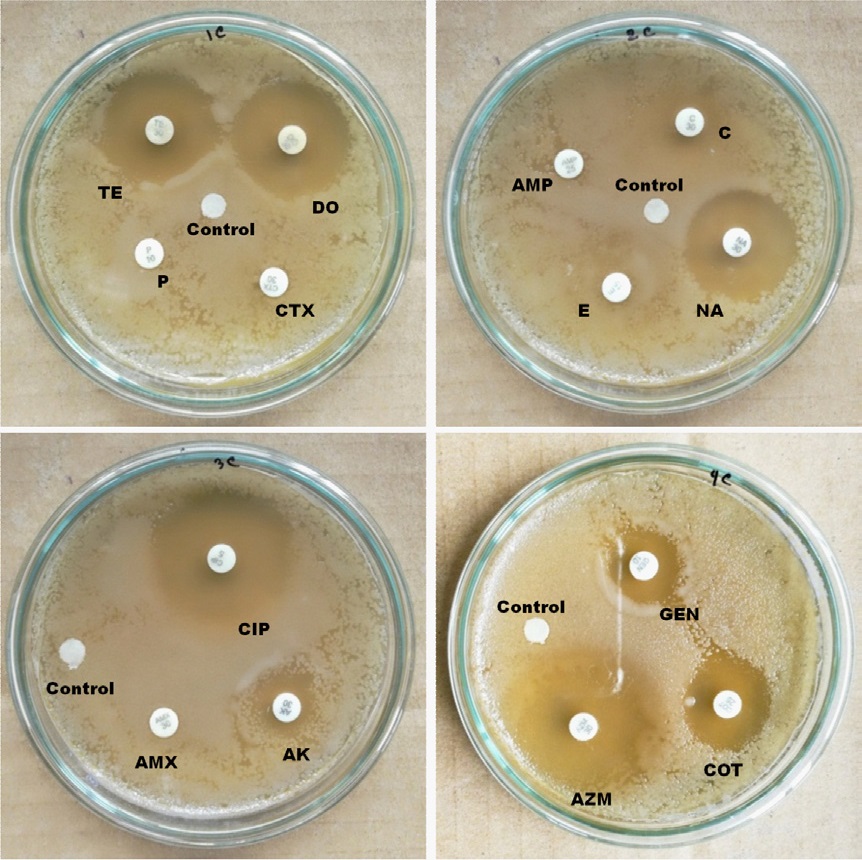
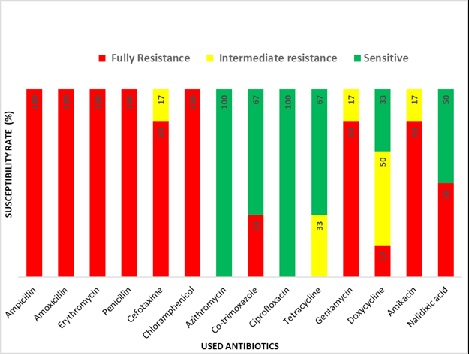
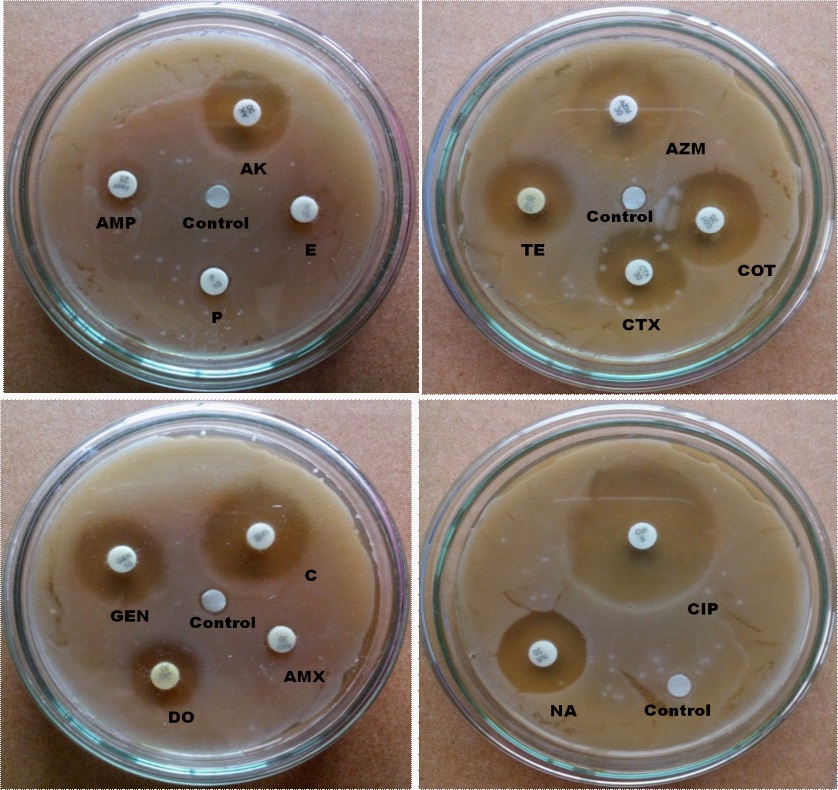
In the case of urban samples, our disc diffusion results show that P, CTX, AMP, E and AMX are completely, and C, AK and GEN are intermediate resistant whereas TE, DO, NA, CIP, AZM and COT are sensitive to E. coli isolated from carrot sample (Figure 3). E. coli from urban tea stall water is completely resistant to AMP, CTX, AMX, NA, E, P and COT, and intermediate resistant to DO, C and TE whereas sensitive to AK, GEN AZM and CIP (Figure 4). Our statistical analysis of urban samples show that E. coli is resistant to AMP (100%), AMX (100%), E (100%), P (100%), C (100%), CTX (83%), COT (33%), GEN (83%), DO (17%), AK (83%) and NA (50%), and intermediate resistant to CTX (17%), TE (33%), GEN (17%), DO (50%) and AK (17%) whereas sensitive to AZM (83%), COT (67%), CIP (100%), TE (67%), DO and NA (50%) (Figure 5).
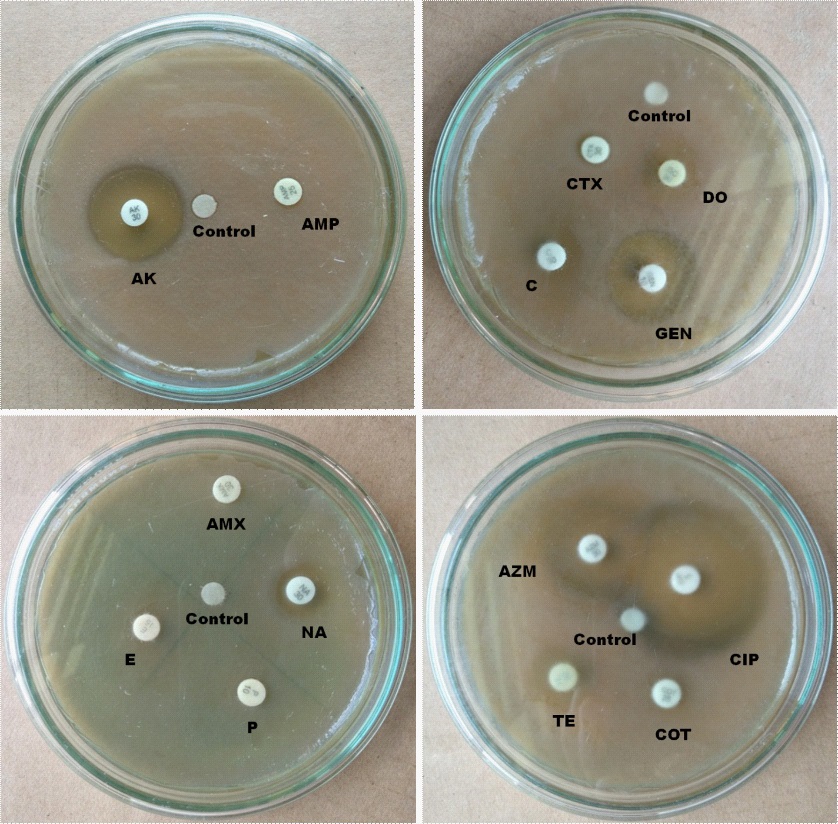
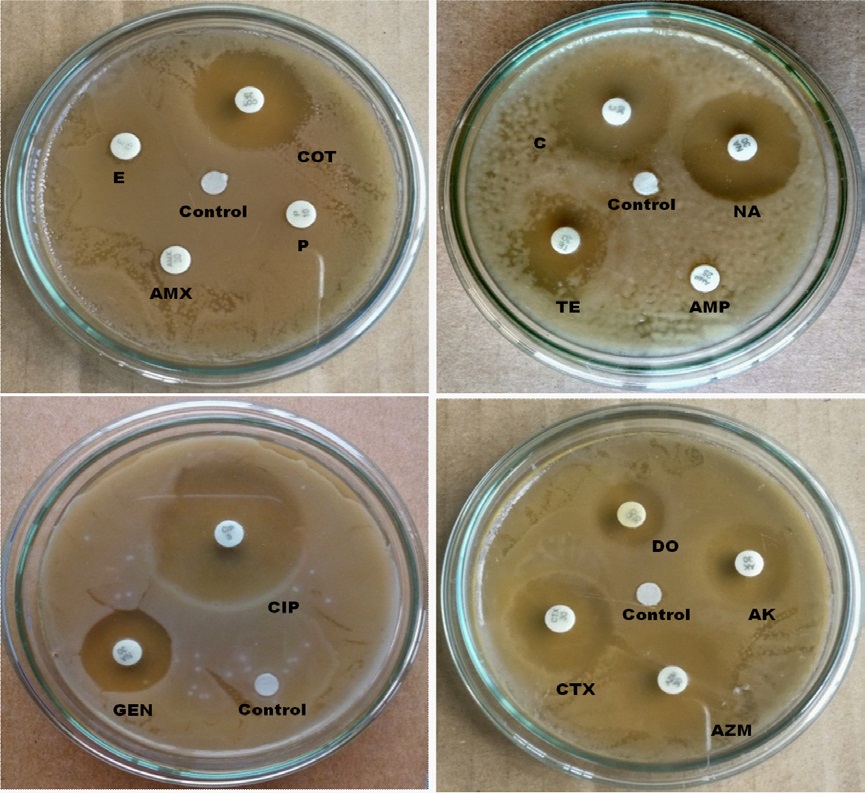
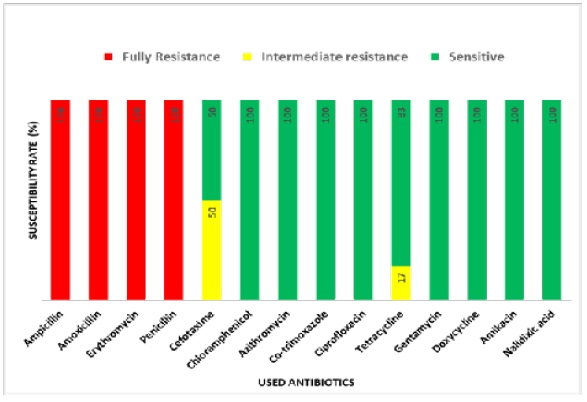
In the case of rural samples, our disc diffusion results show that E, P and AMX are completely resistant, and AMP is intermediate resistant whereas AK, TE, AZM, CTX, COT, GEN, C, DO, NA and CIP are sensitive to E.coli isolated from carrot sample (Figure 6) E.coli from tea stall water is completely resistant to E, AMX, P and AMP and intermediate resistant to TE whereas sensitive to COT, C, NA, GEN, CIP, CTX, DO, AZM and AK. (Figure 7). Our statistical analysis of rural samples show that E. coli is resistant to AMP (100%), AMX (100%), E (100%) and P (100%), and intermediate resistant to CTX (50%) and TE (17%) whereas sensitive to CTX (50%), C (100%), AZM (100%), COT (100%), CIP (100%), TE (83%) GEN (100%), DO (100%), AK (100%) and NA (100%) (Figure 8).
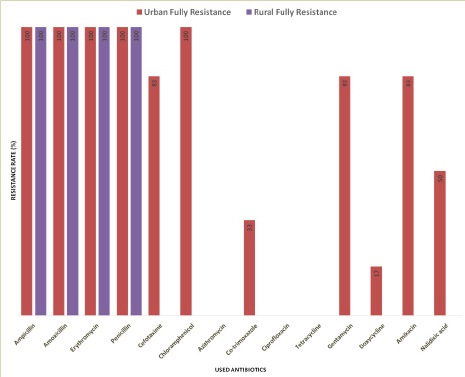
In summary, AMP, AMX, E and P all are resistant (100%) in both the urban and rural area in Sylhet division. However, CTX (83%), C (100%), COT (33%), GEN (83%), DO (17%), AK (83%) and NA (50%) are resistant only in the urban but not rural area of this division (Figure 9). On the other hand, AZM (100%) CIP (100%) and TE (100%) are completely sensitive to E.coli in both the urban and rural area (Figure 9). In addition, rural E.coli is sensitive to CTX (100%) and urban E.coli to CTX (17%), COT (66%), GEN (17%), DO (83%), AK (17%) and NA (50%) (Figure 9). Importantly, multiple antibiotic resistance (MAR) of E. coli is significantly higher in urban than rural samples (Table 3). Together, our results suggest geographical variations of antibiotics resistance exist in Sylhet Division.
Occurrence of antibiotic resistance associated factors and its related diseases
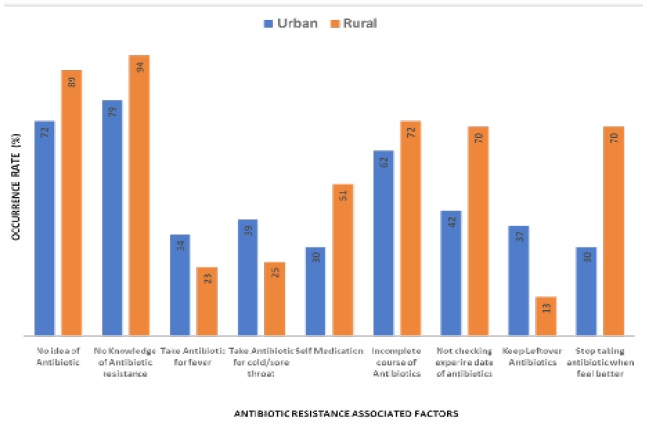
We analyzed total 577 questionnaires from urban (n=283) and rural (n=284) areas in this study. Male and female children (n=100), adolescent (n=82) and old (n=102) from urban and children (n=100), adolescent (n=102) and old (n=91) from rural areas were included in this study. A structured question naire form was used to collect information about factors associated with ABR and its related negative health outcomes like infectious diseases, chronic diseases and antibiotic allergy. In this study, we considered following factors associated with ABR are: no idea of antibiotic, no knowledge of antibiotic resistance, take antibiotic for fever, take antibiotic for cold/sore throat, self medication, incomplete course of antibiotics, not checking expire date of antibiotics, keep leftover antibiotics and stop taking antibiotic when feel better (Figure 10).
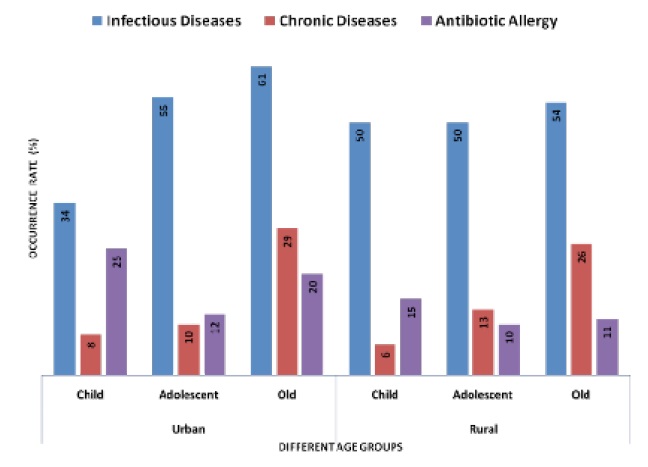
Our data indicates that the higher rate of no idea of antibiotic (in urban 72% and rural 89%), no knowledge of antibiotic resistance (in urban 79% and rural 94%), self medication (in urban 30% and rural 51%), incomplete course of antibiotics (in urban 62% and rural 72%), not checking expire date of antibiotics (in urban 42% and rural 70%), and stop taking antibiotic (in urban 30% and rural 70%) in rural than urban people. Nevertheless, our data also indicates that the higher rate of take antibiotic for fever (in urban 34% and rural 23%), take antibiotic for cold/sore throat (in urban 39% and rural 25%) and keep leftover antibiotics (in urban 37% and rural 13%) in urban than rural people (Figure 10). We next aimed in this study, whether or not those ABR associated factors have any effect on negative health outcomes (Figure 11). In child, we found that the higher occurrence rate of chronic disease and antibiotic allergy in urban than rural area. However, the occurrence rate of infectious disease in child was higher in rural than urban area. In the case old, the occurrence rate of infectious disease, chronic disease and antibiotic allergy were higher in urban than rural area (Figure 11).
Table 1: Measurement of water Quality parameters in urban and rural areas. Mild difference in temperature and No difference of PH between urban and rural filtered and tube well water. The value of BOD is increased in urban filtered and tube well water compared to rural water. The DO value is little high in rural water than urban.
Table 2: Prevalence of E. coli in urban and rural samples. In urban, 12 samples have E. coli among 36 samples (33.33%). In rural, 4 samples have E. coli among 36 samples (11.11%).
Table 3: Prevalence of E. coli in urban and rural samples. In urban, 12 samples have E. coli among 36 samples (33.33%). In rural, 4 samples have E. coli among 36 samples (11.11%).
Discussion
In this study, we compare the geographical variation of the antibiotic resistance of E. coli first time in Bangladesh. Our research group aims to make antibiotic resistance variation profiles 8 divisions of Bangladesh. Accordingly, our first study is in the urban and rural Sylhet division of Bangladesh. We also assessed the presence in this study if the antibiotic resistance and its associated factors lead to negative health outcomes.
In this study, we found the prevalence of E. coli is higher in urban than rural area suggesting that faecal contamination could be higher in urban region (Table 2). E. coli, a member of the faecal coliform group, has been used as an indicator of human enteric pathogens for many years [36]. A study among adults from eight developing countries found the contrasting result that the prevalence of resistance in fecal E. coli was more common in urban than in rural areas [38]. We also found that E. coli is resistant (100%) to AMP, AMX, E and P in both the urban and rural areas in Sylhet division. However, CTX (83%), C (100%), COT (33%), GEN (83%), DO (17%), AK (83%) and NA (50%) are resistant only in the urban but not rural area of this division (Figure 9). On the other hand, AZM (100%) CIP (100%) and TE (100%) are completely sensitive to E. coli in both the urban and rural area. In addition, rural E. coli is sensitive to CTX (100%) and urban E. coli to CTX (17%), COT (66%), GEN (17%), DO (83%), AK (17%) and NA (50%) (Figure 9). ABR pattern and their criteria are quite different in developed and developing countries [47, 48]. E. coli isolated from patient’s urine is resistant to Azithromycin, Ampicillin, Amoxicillin, Tetracycline, Doxycycline, Ciprofloxacin, Cephradine, PenicillinG and Chloramphenicol in Rajshahi city Bangladesh [49]. The geographical variation of ABR in E. coli from environmental samples has found in many studies [50-58]. Thus, our results suggest a geographical variation of ABR is existed in Sylhet Division. In addition, we found higher MAR index of E. coli from drinking water and foodstuffs in urban area than rural area of Sylhet division (Table 3). Notably, a MAR index ˃0.2 indicates the high-risk source of contamination [46]. E. coli from environmental samples has shown MAR profiles in India [58]. Multi drug resistance (MDR) E. coli was also reported from poultry farms, wild birds, pigeons, dogs, and humans [59]. We found water quality is relatively bad in urban area might be passively leading E.coli to be MAR (Table 1). Study has found the relation between water quality an antibiotic resistant [60]. Importantly, bacteria can develop MDR patterns through different genetic mechanisms [61,62]. Accordingly, our results suggest that the urban E. coli has high contamination rate leading poor antibiotic treatment for the E. coli infections.
Furthermore, our study found that the higher rate of no idea of antibiotic, no knowledge of antibiotic resistance, self medication, incomplete course of antibiotics, not checking expire date of antibiotics and stop taking antibiotic in rural than urban people. Our data also indicates that the high rate of take antibiotic for fever, take antibiotic for cold/sore throat and keep leftover antibiotics in urban than rural people (Figure 10). In addition, we also found that in child the higher occurrence rate of chronic disease and antibiotic allergy in urban than rural area. However, the occurrence rate of infectious disease in child was higher in rural than urban area. In the case old, the occurrence rate of infectious disease, chronic disease and antibiotic allergy were higher in urban than rural area (Figure 11). Accordingly, our results suggest that the correlation between the occurrence rates of ABR associated factors and its negative health outcomes is not exhibited. One reason, the urban area possibly posse’s high prevalence of pathogenic E. coli leading high ABR and negative health outcomes although having the lower occurrence of ABR associated factors. Infections caused by resistant bacterial strains lead to up to two-fold higher rates of adverse outcomes compared with similar infections caused by susceptible strains [63]. Antibiotic overdose can also cause some other health threats such as skin problems, severe allergies, hypersensitivity etc. Improved knowledge and more awareness among the nonmedical population about allopathic drugs, especially an understanding of the usage of antibiotics can help in limiting the microbial resistance issues globally [25]. Due to aberrant use of antibiotics, numerous bacterial human pathogens have evolved into MDR types. Therapeutic preferences for these resistant microbes are less, and these are associated with a prolonged stay at hospital and increased economic cost. MDR types bacteria become more pathogenic over times. Antibiotic resistance enables bacteria able to overcome therapeutics and virulence is necessary to fight the host immune system leading infections in human [64, 65].
Conclusions
In conclusion, this study shows first time in Bangladesh the prevalence of antibiogram in E. coli isolated from food stuffs and drinking water in Sylhet division. In addition, this study also shows the presence of antibiotic resistance, its associated factors and negative health outcomes in Sylhet division.
1. Our findings suggest that fecal contamination is higher in urban region than rural in Sylhet division.
2. Our findings indicate the geographic variation of antibiotic resistance of E.coli is existed in Sylhet division.
3. Azithromycin, Ciprofloxacin and Tetracycline are sensitive to E. coli in both rural & urban region in Sylhet.
4. Ampicillin, Amoxicillin, Erythromycin and Penicillin are resistant to E. coli in both rural & urban region in Sylhet.
5. Cefotaxime, Chloramphenicol, Co-trimoxazole, Gentamicin, Doxycycline, Amikacin and Nalidixic acid are sensitive and resistant to E. coli in rural and urban area respectively in Sylhet division.
6. The prevalence rate of antibiotic resistance associated factors is higher in rural area than urban area.
7. The negative health outcomes related with antibiotic resistance and its risk factors are relatively higher in urban than rural area.
Declarations
Acknowledgements: The team is grateful to the Microbiology lab, Department Biochemistry and Molecular Biology, Shahjalal University of Science and Technology, Sylhet, Bangladesh.
Funding: This research was conducted by the support of SUST research center grant (Grant number LS/2020/01/07).
References
- Aslam B, Wang W, Arshad MI, Khurshid M, Muzammil S, Rasool MH, et al. Antibiotic resistance: a rundown of a global crisis. Infect Drug Resist. 2018; 11: 1645-58.
- O’Neill J. Tackling drug-resistant infections globally: final report and recommendations. HM Government and Wellcome Trust: UK 2016.
- Rather IA, Kim BC, Bajpai VK, Park YH. Self-medication and antibiotic resistance: Crisis, current challenges, and prevention. Saudi J Biol Sci. 2017; 24(4): 808-12.
- Gootz TD. The global problem of antibiotic resistance. Crit Rev Immunol. 2010; 30(1): 79-93.
- Nathan C, Cars O. Antibiotic resistance--problems, progress, and prospects. N Engl J Med. 2014; 371(19): 1761-3.
- Organization WH. Global Action Plan on Antimicrobial Resistance. 2015.
- Chen B, Zheng W, Yu Y, Huang W, Zheng S, Zhang Y, et al. Class 1 integrons, selected virulence genes, and antibiotic resistance in Escherichia coli isolates from the Minjiang River, Fujian Province, China. Appl Environ Microbiol. 2011; 77(1): 148-55.
- Heuer H, Schmitt H, Smalla K. Antibiotic resistance gene spread due to manure application on agricultural fields. Curr Opin Microbiol. 2011; 14(3): 236-43.
- Jiang L, Hu X, Xu T, Zhang H, Sheng D, Yin D. Prevalence of antibiotic resistance genes and their relationship with antibiotics in the Huangpu River and the drinking water sources, Shanghai, China. Sci Total Environ. 2013; 458-460: 267-72.
- Ribeiro AF, Laroche E, Hanin G, Fournier M, Quillet L, Dupont JP, et al. Antibiotic-resistant Escherichia coli in karstic systems: A biological indicator of the origin of fecal contamination? FEMS Microbiol Ecol. 2012; 81(1): 267-80.
- Flores Ribeiro A, Bodilis J, Alonso L, Buquet S, Feuilloley M, Dupont JP, et al. Occurrence of multi-antibiotic resistant Pseudomonas spp. in drinking water produced from karstic hydrosystems. Sci Total Environ. 2014; 490: 370-8.
- Guo X, Li J, Yang F, Yang J, Yin D. Prevalence of sulfonamide and tetracycline resistance genes in drinking water treatment plants in the Yangtze River Delta, China. Sci Total Environ. 2014; 493: 626-31.
- Machado A, Bordalo AA. Prevalence of antibiotic resistance in bacteria isolated from drinking well water available in GuineaBissau (West Africa). Ecotoxicol Environ Saf. 2014; 106: 188-94.
- Mohanta T, Goel S. Prevalence of antibiotic-resistant bacteria in three different aquatic environments over three seasons. Environ Monit Assess. 2014; 186(8): 5089-100.
- Organization WH. Antimicrobial resistance. 2020.
- Davies J, Davies D. Origins and evolution of antibiotic resistance. Microbiol Mol Biol Rev. 2010; 74(3): 417-33.
- Munita JM, Arias CA. Mechanisms of Antibiotic Resistance. Microbiol Spectr. 2016; 4(2).
- Ramirez MS, Tolmasky ME. Aminoglycoside modifying enzymes. Drug Resist Updat. 2010; 13(6): 151-71.
- Holmes AH, Moore LS, Sundsfjord A, Steinbakk M, Regmi S, Karkey A, et al. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet. 2016; 387(10014): 176-87.
- French GL. The continuing crisis in antibiotic resistance. Int J Antimicrob Agents. 2010; 36 Suppl 3:S3-7.
- Michael CA, Dominey-Howes D, Labbate M. The antimicrobial resistance crisis: causes, consequences, and management. Front Public Health. 2014; 2:145.
- Read AF, Woods RJ. Antibiotic resistance management. Evol Med Public Health. 2014; 2014(1): 147.
- Stokes HW, Gillings MR. Gene flow, mobile genetic elements and the recruitment of antibiotic resistance genes into Gram-negative pathogens. FEMS Microbiol Rev. 2011; 35(5): 790-819.
- Bennadi D. Self-medication: A current challenge. J Basic Clin Pharm. 2013; 5(1): 19-23.
- Casadevall A. Crisis in infectious diseases: time for a new paradigm? Clin Infect Dis. 1996; 23(4): 790-4.
- Casadevall A. Crisis in Infectious Diseases: 2 Decades Later. Clin Infect Dis. 2017; 64(7): 823-8.
- Edwards DJ, Richman PB, Bradley K, Eskin B, Mandell M. Parental use and misuse of antibiotics: are there differences in urban vs. suburban settings? Acad Emerg Med. 2002; 9(1): 22-6.
- Friedman ND, Temkin E, Carmeli Y. The negative impact of antibiotic resistance. Clin Microbiol Infect. 2016; 22(5): 416-22.
- Gyssens IC. Quality measures of antimicrobial drug use. Int J Antimicrob Agents. 2001; 17(1): 9-19.
- Joshi S, Unni J, Vijay S, Khanijo V, Gupte N, Divate U. Obesity and pregnancy outcome in a private tertiary hospital in India. Int J Gynaecol Obstet. 2011; 114(1): 82-3.
- Kuperman AA, Koren O. Antibiotic use during pregnancy: how bad is it? BMC Med. 2016; 14(1): 91.
- R Sharma UV, CL Sharma, B Kapoor. Self-medication among urban population of Jammu city. Indian Journal of Pharmacology. 2005; 37(1): 40-3.
- Turta O, Rautava S. Antibiotics, obesity and the link to microbes - what are we doing to our children? BMC Med. 2016; 14: 57.
- Vaishali Phalke DPaPD. Self-medication practices in rural Maharashtra. Indian Journal of Community Medicine 2006; 31(1): 34- 5.
- Geldreich EE. Sanitary significance of fecal coliforms in the environment (Water pollution control research series) Unknown Binding – January 1, 1966. USA: U.S. Dept. of the Interior, Federal Water Pollution Control Administration; 1966 January 1, 1966. 122 p.
- Koneman EW AS, Janda WM, Schreckenberger PC and Winn WC. . The Enterobacteriaceae. . 6th edition ed: Philadelphia: Lippincott Raven Publisher.; 2006.
- Nys S, Okeke IN, Kariuki S, Dinant GJ, Driessen C, Stobberingh EE. Antibiotic resistance of faecal Escherichia coli from healthy volunteers from eight developing countries. J Antimicrob Chemother. 2004; 54(5): 952-5.
- Peralta G, Sánchez MB, Garrido JC, De Benito I, Cano ME, Martínez-Martínez L, et al. Impact of antibiotic resistance and of adequate empirical antibiotic treatment in the prognosis of patients with Escherichia coli bacteraemia. J Antimicrob Chemother. 2007; 60(4) : 855-63.
- Maal-Bared R, Bartlett KH, Bowie WR, Hall ER. Phenotypic antibiotic resistance of Escherichia coli and E. coli O157 isolated from water, sediment and biofilms in an agricultural watershed in British Columbia. Sci Total Environ. 2013; 443: 315-23.
- Pereira A, Santos A, Tacão M, Alves A, Henriques I, Correia A. Genetic diversity and antimicrobial resistance of Escherichia coli from Tagus estuary (Portugal). Sci Total Environ. 2013; 461-462: 65-71.
- Tzialla C, Borghesi A, Perotti GF, Garofoli F, Manzoni P, Stronati M. Use and misuse of antibiotics in the neonatal intensive care unit. J Matern Fetal Neonatal Med. 2012; 25 Suppl 4: 35-7.
- Talukdar PK, Rahman M, Rahman M, Nabi A, Islam Z, Hoque MM, et al. Antimicrobial resistance, virulence factors and genetic diversity of Escherichia coli isolates from household water supply in Dhaka, Bangladesh. PLoS One. 2013; 8(4): e61090.
- CLSI. Performance Standards for Antimicrobial Disk Susceptibility Tests; Approved Standard—Eleventh Edition. CLSI document M02-A11. Wayne, PA: Clinical and Laboratory Standards Institute. 2011.
- Bauer AW, Kirby WM, Sherris JC, Turck M. Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol. 1966; 45(4): 493-6.
- Krumperman PH. Multiple antibiotic resistance indexing of Escherichia coli to identify high-risk sources of fecal contamination of foods. Appl Environ Microbiol. 1983; 46(1): 165-70.
- Chokshi A, Sifri Z, Cennimo D, Horng H. Global Contributors to Antibiotic Resistance. J Glob Infect Dis. 2019; 11(1): 36-42.
- Shibl AM, Memish Z, Osoba A. Antibiotic resistance in developing countries. J Chemother. 2001; 13 Suppl 1: 40-4.
- Nurunnabi SM, Sutradhar J, Topa FB, Noman A, Rahman A, Sakib RT, Nousin NT, Mahmud S, Disha SS, Imran A, Shahjahan M. Resistance profiles of Azithromycin in Escherichia coli isolated from human urine. Bioresearch Communications. 2019; 5(2): 707-14.
- Abera B, Kibret M. Azithromycin, fluoroquinolone and chloramphenicol resistance of non-chlamydia conjunctival bacteria in rural community of Ethiopia. Indian J Ophthalmol. 2014; 62(2): 236-9.
- Apanpa MOEaMO. Status of contamination and antibiotic resistance of bacteria from well water in Ago- Iwoye, Nigeria. Journal of Applied Biosciences. 2010; 35: 2244-50.
- Coleman BL, Salvadori MI, McGeer AJ, Sibley KA, Neumann NF, Bondy SJ, et al. The role of drinking water in the transmission of antimicrobial-resistant E. coli. Epidemiol Infect. 2012; 140(4): 633-42.
- Erb A, Stürmer T, Marre R, Brenner H. Prevalence of antibiotic resistance in Escherichia coli: overview of geographical, temporal, and methodological variations. Eur J Clin Microbiol Infect Dis. 2007; 26(2): 83-90.
- Gherbawy AMAaYA. Antibiotic resistance in Escherichia coli strains isolated from water springs in Al-Ahsa Region. African Journal of Microbiology Research. 2010; 5(2): 123-30.
- Ibrahim ME, Bilal NE, Hamid ME. Comparison of phenotypic characteristics and antimicrobial resistance patterns of clinical Escherichia coli collected from two unrelated geographical areas. Glob J Health Sci. 2014; 6(6): 126-35.
- Lyimo B, Buza J, Subbiah M, Smith W, Call DR. Comparison of antibiotic resistant Escherichia coli obtained from drinking water sources in northern Tanzania: a cross-sectional study. BMC Microbiol. 2016; 16(1): 254.
- Parveen S, Lukasik J, Scott TM, Tamplin ML, Portier KM, Sheperd S, et al. Geographical variation in antibiotic resistance profiles of Escherichia coli isolated from swine, poultry, beef and dairy cattle farm water retention ponds in Florida. J Appl Microbiol. 2006; 100(1): 50-7.
- Sahoo KC, Tamhankar AJ, Sahoo S, Sahu PS, Klintz SR, Lundborg CS. Geographical variation in antibiotic-resistant Escherichia coli isolates from stool, cow-dung and drinking water. Int J Environ Res Public Health. 2012; 9(3): 746-59.
- Mohsin M, Raza S, Roschanski N, Guenther S, Ali A, Schierack P. Description of the First Escherichia coli Clinical Isolate Harboring the Colistin Resistance Gene mcr-1 from the Indian Subcontinent. Antimicrob Agents Chemother. 2017; 61(1).
- Chen Z, Yu D, He S, Ye H, Zhang L, Wen Y, et al. Prevalence of Antibiotic-Resistant Escherichia coli in Drinking Water Sources in Hangzhou City. Front Microbiol. 2017; 8: 1133.
- Uma KP, Rajendran S, and Sarayu YL. Antimicrobial Activity and Phytochemical Analysis of Coriander sativum Against Infectious Diarrhea. Ethnobotanical Leaflets 2009; 13: 590-4.
- Ibrahim ME, Magzoub MA, Bilal NE, Hamid ME. Distribution of Class I integrons and their effect on the prevalence of multi-drug resistant Escherichia coli clinical isolates from Sudan. Saudi Med J. 2013; 34(3): 240-7.
- Cosgrove SE, Carmeli Y. The impact of antimicrobial resistance on health and economic outcomes. Clin Infect Dis. 2003; 36(11): 1433-7.
- Feldman MW, Laland KN. Gene-culture coevolutionary theory. Trends in ecology & evolution. 1996; 11(11): 453-7.
- Martínez JL, Baquero F. Interactions among strategies associated with bacterial infection: pathogenicity, epidemicity, and antibiotic resistance. Clin Microbiol Rev. 2002; 15(4): 647-79.