Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
piRNAs and PIWI proteins as potential biomarkers in colorectal cancer
Mandana Ameli Mojarad; Melika Ameli Mojarad*
Faculty of biological science, University of Kharazmi, Tehran Iran.
*Corresponding Author: Melika Ameli Mojarad
Faculty of biological science, University of Kharazmi,
Tehran Iran.
Email: melikaamelimojarad@gmail.com
Received : Oct 08, 2021
Accepted : Nov 25, 2021
Published : Dec 02, 2021
Archived : www.jcimcr.org
Copyright : © Mojarad MA (2021).
Abstract
PIWI-interacting RNAs (piRNAs) are another subgroup of small non-coding RNAs emerging as key players in different biological activities, including regulating gene and protein expression, epigenetic silencing of transposable elements, and controlling the spermatogenesis by interacting with PIWI proteins. Recently, with the help of next-generation sequencing, abnormal piRNA expression has been observed in different types of cancer, such as colorectal cancer (CRC). A new investigation proposing piRNA as a prognostic and diagnostic biomarker in the treatment of CRC Therefore, this review focuses on the role of piRNAs in the initiation, progression, and metastasis of CRC and its molecular mechanisms to understand its function and provide a better therapeutic strategy.
Keywords: PIWI-interacting RNA; piRNA; small RNA; colorectal cancer.
Abbreviations: CRC: Colorectal Cancer; HSF1: Heat Shock Factor 1; HSP: Heat Shock Protein; ncRNAs: Non-Coding RNA; lncRNA: Long Non-Coding RNA; nt: nucleotide; OS: Overall Survival; pi-RISC: piRNAs-induced Silencing Complex; piRNA: Piwi-interacting RNA; TE: Transposable Element; Zac: Zucchini.
Citation: Mojarad MA, Mojarad MA. piRNAs and PIWI proteins as potential biomarkers in colorectal cancer. J Clin Images Med Case Rep. 2021; 2(6): 1447.
Background
Colorectal cancer (CRC) is the third most diagnosed cancer and the primary health concern among both men and women worldwide. According to the 2020 cancer statistic, CRC has the most morbidity and mortality rate after lung cancer, with 2 million new cases reported in 2020 [1]. Emerging evidence shows different risk factors can increase CRC incidence, including overweight, age, alcohol drinking, cigarette smoking, family history, and inflammatory bowel disease (IBD) [2,3]. Unfortunately, based on Inadequate prognostic and diagnostic biomarkers, most patients are diagnosed in advanced stages, losing their chance for early treatment; therefore, finding sensitive and stable therapeutic markers is a crucial Approach in controlling the CRC. Recent discoveries on RNAs reported that non-coding RNAs contain 98% of the human genome, involving different biological pathological activities [4-6]. PIWI-interacting RNAs (piRNAs) are a new type of recently discovered small non-coding RNAs with 24-31 nucleotides (nt) in length [7]. They were first discovered in 2001 in Drosophila melanogaster as small RNAs [8]. piRNAs can play a vital role in physiological processes both at the transcriptional or post-transcriptional level [9]. piRNA binds to PIWI proteins to form a ribonucleoprotein silencing complex, RISC (RNA-induced silencing complex) to influence genome rearrangement, epigenetic regulation, transposon silencing, spermiogenesis, protein regulation, and, most importantly, preservation of genome integrity [10,11]. A member of the argonaut protein family encoded by the Drosophila piwi gene is involved in DNA methylation, transposon silencing, germline development, and fertility [12-14]. Recently several studies have reported several abnormally expressed piRNA and PIWI proteins in different tumors, including CRC; it has been shown that high piRNA expression may be relevant for cancer tumorigenesis and cancer prognosis in various types of human tumors [15-17]. Therefore, this review summarizes recent studies on piRNAs and discusses their emerging roles in CRC as potential biomarkers.
piRNAs biogenesis
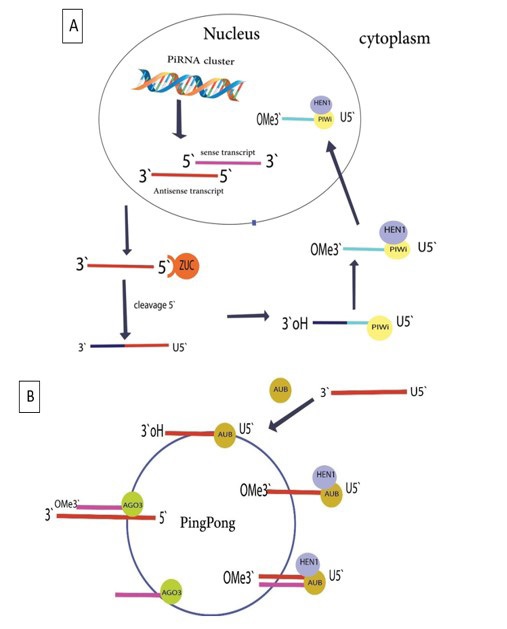
The investigation found that there are three subclasses for piRNAs that based on their origin derived from transposons, derived from lncRNAs, and derived from mRNAs, piRNA biogenesis is initiated by Pol II copying RNA from its cluster, and then, with more turnover, the piRNA precursor is transferred to the cytoplasm [18,19]. piRNAs are mostly transcribed as extensive single-stranded transcripts resulting from single-stranded precursors in the nucleus. Nascent piRNAs (pre-piRNAs) require post-transcriptional modification independent of Dicer to become mature piRNAs. Two mechanisms are involved in the generation of mature piRNAs: a primary synthesis mechanism that is carried out in both somatic and germ cells and a ‘ping pong’ amplification mechanism that is only formed in the germline [9,20]. Primary piRNAs, cleaved by Riboendonuclease called Zucchini (Zuc) with the help of Minotaur (Mino) to produce piRNA intermediates with a 5′ uracil in the cytoplasm. piRNA has a unique structure of 2’Omethyl at the 3’ end, which is methylated by the Hen1 enzyme, so the PIWI protein binds to this structure at the 3’ end to create the piRNA/PIWI complex [21]. Then the piRNA/PIWI complex migrates back to the nucleus, reaching its target transcript and repressing transposons during transcription and post-transcription, which protects germline genome integrity. In a ping pong mechanism, piRNAs bind to the proprietary AGO3 or AUB proteins to the PIWI protein subfamily in the cytoplasm to form piRNA/Ago3 or piRNA/AUB complexes, generating new piRNAs using the piRNA and piRNA/ Ago3 or piRNA/AUB complexes as templates, using the newly developed piRNAs. to synthesize other piRNAs as substrates in the same method [15,22] (Figure 1A). Ping pong structures have been identified in zebrafish, D. melanogaster, and other species. However, recent data have reported that piRNA biogenesis is independent of the ping-pong mechanism in mice during adult spermatogenesis. Therefore, piRNA biogenesis requires further investigation in mammals [20]. (Figure 1B).
piRNAs and colorectal cancer
Recent studies have found abnormal expression of the piRNA/PIWI complex in various types of cancer, especially colon cancer, in correlation with different pathological processes, suggesting a role for piRNAs in tumor-promoting or tumor suppression in cancer. A theory also indicates that piRNAs might play a tissue-specific role based on their high expression in different tissues. However, the mechanism by which piRNAs contribute to cancer is still not fully understood [20,23,24].
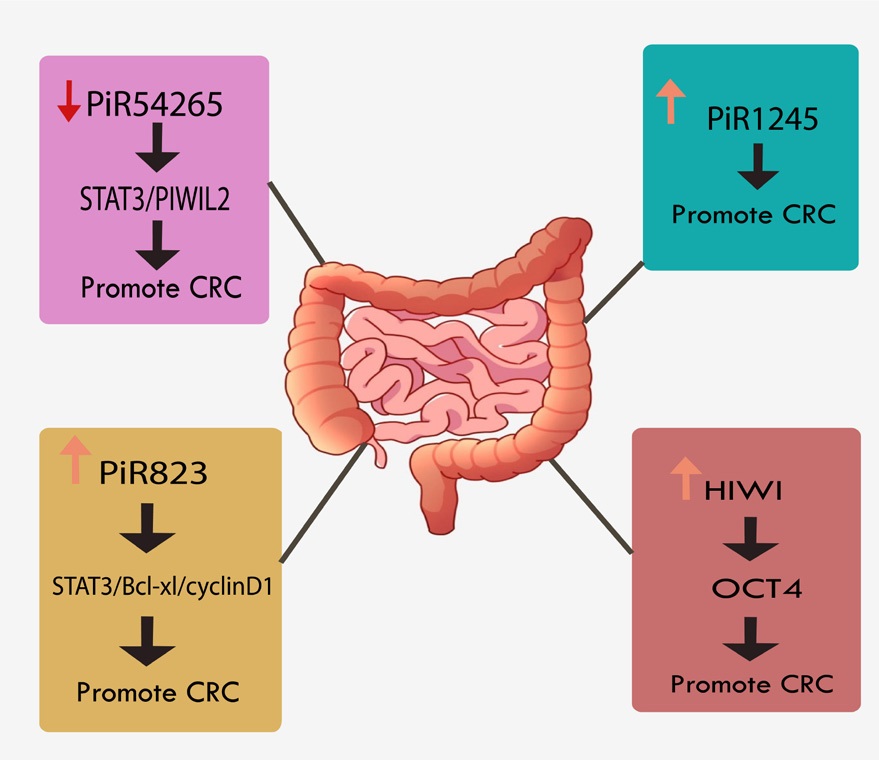
Several unregulated piRNAs have been reported in colorectal cancer tumor tissue, which is a heterogeneous disease. Largescale piRNA expression profiling in serum samples from colon cancer patients compared with regular groups found that 31 deregulated piRNAs, including piR5937 and piR28876, had the highest sensitivity and specificity, suggesting early diagnostic markers for differentiation between cancer and healthy patientsdonors for clinical use applications [25]. Dysregulation of piR-1245 and piR-823 were significantly discovered in the CRC tissues, promoting migration and invasion and inhibiting apoptosis in CRC. Upregulation of piR-1245 was down-regulation has associated with lymph node metastasis, poor differentiation, and shortens overall survival time of the patient; it has been elucidated that piR-1245 binds to the intronic regions of targeted mRNAs with the tumor-suppressive role and promote mRNA degradation via nuclear exosomes [26]. Furthermore, piR-1245 is found to be associated with genes involved in tumor-suppressive pathways [27]. piR-823 up-regulation promotescell proliferation by modifying the heat shock factor 1 (HSF1) at a posttranslational level.
In contrast, piR-823 inhibitions topped tumor cells at the G1 stage in CRC [28]. piR-54265 is another overexpressed piRNA in CRC discovered through the experiment in xenograft mice model, with the oncogenic role by promoting metastasis, chemoresistance, and inhibiting the apoptosis by forming aPIWIL2/ STAT3/phosphorylated-SRC (p-SRC) complex. High expression of piR-54265 is also correlated with shorter overall survival (OS) and progression-free survival (PFS) both in tumor and serum. Inhibition of piRNA-54265 significantly suppressed tumor metastasis and growth [29]. The piR-18849 and piR-1952 were both upregulated in CRC tissues by using small RNA sequencing, suggesting them as a prognostic biomarker; the overexpression of piR-18849 and piR-1952 was correlated with poor differentiation while only piR-18849 was associated with lymph node metastasis [30]. Upregulation of piR-823 in the CRC tissues enhances cell proliferation and tumorigenesis by upregulation of heat shock factor 1 (HSF1), which is a strong driver of carcinogenesis by affecting STAT3 phosphorylation. STAT3/Bcl-xl/cyclinD1 signaling pathway will be activated by piR-823/piwil2 complex and regulate G1 phase progression. Suppress of piR-823 showed inhibition of cell proliferation, cell-cycle arrest in the G1 phase, and induced apoptosis [28]. A schematic illustration of the function of piRNAs in CRC is shown in (Figure 2).
piRNAs as biomarkers in CRC cancer
Several studies have been performed to find new biological markers for the early diagnosis of CRC. Recently, piRNAs similar to miRNAs are very stable and resistant to degradation by ribonucleases in body fluids. As noted above, different piRNAs express differently between paired tumor and normal tissues, associated with aggressive biological behavior [31]. The expression of certain piRNAs related to cancer proliferation and metastasis, including piR1245, piR823, piR015551. piRNA has also been found to have great potential as an early minimally invasive biomarker in CRC due to its stability and tissue abundance, being referred to as an attractive prognostic and candidate diagnostic biomarker [26,31,32]. The clinical phase I study suggested these two piRNAs as promising biomarkers for the early diagnosis of colon cancer [32]. The mechanism of action for piRNAs in CRCis listed in (Table 1).
Table 1:The role of piRNAs in different cancer.
piRNA |
Cancer type |
Expression |
Clinical correlation |
Reference |
Colorectal Cancer |
up |
Promotingmigration, invasioninhibitingapoptosis. |
[26] |
|
Colorectal |
up |
Promoting proliferation and suppressing apoptosis via HSF1 activation |
[33] |
|
Colorectal cancer |
up |
Promoting the colorectal cancer development |
[34] |
|
piR-28876 |
Colorectal cancer |
down |
Recurrence-free survival |
[35] |
piR-54265 |
Colorectal cancer |
up |
Associating with TNM stage |
[17] |
piR-5937 |
Colorectal cancer |
down |
Associating with TNM stage |
[36] |
piR-18849 |
Colorectal cancer |
up |
Associating with poor degree differentiation |
[30] |
PIWI proteins and CRC
The PIWI protein is a member of the highly conserved family of Argonaute proteins found in mice, Drosophila, and humans. The PIWI protein in humans is made up of four proteins, including PIWIL1 (HIWI), PIWIL2 (similar to Miwi), PIWIL3 (HIWI3), and PIWIL4 (HIWI2) [37]. Among all four PIWI proteins, PIWIL1 is the most studied protein regulating gene expression, apoptosis, the cell cycle, and cell proliferation [38]. Recent studies have shown that piwil2 plays a role in various tumors by inhibiting apoptosis through the Stat3 / BclXL pathway; Stat3 has an oncogenic role in anti-apoptotic and metastatic processes. Another study showed that piwil2, in interaction with EIF2C1, is related to CRC tumors' formation, development, and metastasis, suggesting that it is a possible early diagnostic biomarker [38-40]. It has also been suggested that Piwil1 and Piwil2 as potential biomarkers for colon cancers in humans [41]. Upregulation of Piwil1 and Piwil2 has been demonstrated in the early stages of injury (adenoma stage) in CRC patients, suggesting that increased expression of Piwil1 and Piwil2 plays a vital role in the development of CRC [42]. The dysregulation of Piwil1 and Piwil2 plays a crucial role in the pathological process of several human cancers and, often associated with poor survival and aggressive clinicopathological properties of patients; the PIWI protein turned out to be a stem cell protein that plays a crucial role in cancer stem cell (CSC) differentiation, stem cell self-reproduction, DNA methylation, spermatogenesis playing gene regulation [43,44].
Conclusions
With the development of bioinformatics and sequencing technologies, more discoveries have been made about the critical role of non-coding RNAs (ncRNAs) in cell biology. In particular, ncRNAs and their acute anti-cancer effects. The strategy to use ncRNAs specifically for cancer treatment has recently developed. piRNAs, which are members of small non-coding RNAs, perform different regulatory functions in the cytoplasm and the nucleus by interacting with PIWI proteins. Furthermore, piRNAs and PIWI proteins have emerged as a new therapeutic target with a tissue-specific role in developing human cancers, with high levels of expression in both tissue and blood emerging as a promising biomarker in several cancers, includingcolorectal cancer. Although piRNA / PIWI has become a hot topic in cancer development, the study of its function and biosynthesis has not yet been fully explored. More research is required for its possible therapeutic applications of proteins in the development of various cancers.
Declarations
Ethics approval and consent to participate: Not applicable.
Consent for publication: Not applicable.
Availability of data and materials: Not applicable.
Competing interests: The authors declare no conflict of interest.
Funding: Not applicable.
Authors' contributions: M.A and M.A contribute equally to drafting, editing, and approving the final submitted version Mandana Ameli Mojarad was the corresponding author.
Acknowledgments: Not applicable.
References
- Siegel RL, Miller KD, Goding Sauer A, Fedewa SA, Butterly LF, Anderson JC, et al. Colorectal cancer statistics, 2020. CA Cancer J Clin. 2020; 70(3): 145–64.
- Zheng Y, Hua X, Win AK, MacInnis RJ, Gallinger S, Le Marchand L, et al. A new comprehensive colorectal cancer risk prediction model incorporates family history, personal characteristics, and environmental factors. Cancer Epidemiol Biomarkers Prev. 2020; 29(3): 549–57.
- CM J, C W, JEE, DJ S, CI A, B L, et al. Meta-analyses of colorectal cancer risk factors. Cancer Causes Control 2013; 24(6): 1207–22.
- Romano G, Veneziano D, Acunzo M, Croce CM. Small non-coding RNA and cancer [Internet]. Vol. 38, Carcinogenesis. Oxford University Press; 2017. p. 485–91.
- Chandra Gupta S, Nandan Tripathi Y. Potential of long non-coding RNAs in cancer patients: From biomarkers to therapeutic targets. Vol. 140, International Journal of Cancer. Wiley-Liss Inc.; 2017. p. 1955–67.
- Ameli-Mojarad M, Ameli-Mojarad M, Nourbakhsh M, Nazemalhosseini-Mojarad E. Circular RNA hsa_circ_0005046 and hsa_ circ_0001791 May Become Diagnostic Biomarkers for Breast Cancer Early Detection. J Oncol. 2021; 2021: 1–7.
- Liu P, Dong Y, Gu J, Puthiyakunnon S, Wu Y, Chen XG. Developmental piRNA profiles of the invasive vector mosquito Aedes albopictus. Parasites and Vectors. 2016; 9(1): 1–15.
- Lin H, Spradling AC. A novel group of pumilio mutations affects the asymmetric division of germline stem cells in the Drosophila ovary. Development 1997; 124(12): 2463–76.
- Ku HY, Lin H. PIWI proteins interact with piRNA biogenesis, germline development, and gene expression [Internet]. Vol. 1, National Science Review. Oxford University Press; 2014. p. 205– 18.
- Xu M, You Y, Hunsicker P, Hori T, Small C, Griswold MD, et al. Mice deficient for a small cluster of Piwi-interacting RNAs Implicate Piwi-interacting RNAs in transposon control. Biol Reprod [Internet]. 2008; 79(1): 51–7.
- Le Thomas A, Rogers AK, Webster A, Marinov GK, Liao SE, Perkins EM, et al. Piwi induces piRNA-guided transcriptional silencing and establishing a repressive chromatin state. Genes Dev. 2013; 27(4): 390–9.
- Ku HY, Lin H. PIWI proteins interact with piRNA biogenesis, germline development, and gene expression [Internet]. Vol. 1, National Science Review. Oxford University Press; 2014. p. 205– 18.
- J B, AA A, A S, M D, M K, R S, et al. Discrete small RNA-generating loci as master regulators of transposon activity in Drosophila. Cell. 2007; 128(6): 1089–103.
- HL, H Y. A novel epigenetic mechanism in Drosophila somatic cells mediated by Piwi and piRNAs. Cold Spring Harb Symp Quant Biol. 2008; 73: 273–81.
- Weng W, Liu N, Toiyama Y, Kusunoki M, Nagasaka T, Fujiwara T, et al. Novel evidence for a PIWI-interacting RNA (piRNA) as an oncogenic mediator of disease progression, and a potential prognostic biomarker in colorectal cancer. Mol Cancer. 2018; 17(1).
- Tan L, Mai D, Zhang B, Jiang X, Zhang J, Bai R, et al. PIWI-interacting RNA-36712 restrains breast cancer progression and chemoresistance by interaction with SEPW1 pseudogene SEPW1P RNA. Mol Cancer. 2019; 18(1).
- Mai D, Ding P, Tan L, Zhang J, Pan Z, Bai R, et al. PIWI-interacting RNA-54265 is oncogenic and a potential therapeutic target in colorectal adenocarcinoma. Theranostics. 2018; 8(19): 5213– 30.
- T T, H L. The biogenesis and function of PIWI proteins and piRNAs: progress and prospect. Annu Rev Cell Dev Biol. 2009; 25: 355–76.
- Yamashiro H, Siomi MC. PIWI-Interacting RNA in Drosophila: Biogenesis, Transposon Regulation, and beyond. Chem Rev. 2018; 118(8): 4404–21.
- Guo B, Li D, Du L, Zhu X. piRNAs: biogenesis and their potential roles in cancer. Vol. 39, Cancer and Metastasis Reviews. Springer; 2020. p. 567–75.
- Iwasaki YW, Siomi MC, Siomi H. PIWI-interacting RNA: Its biogenesis and functions. Annu Rev Biochem. 2015; 84: 405–33.
- Weick EM, Miska EA. piRNAs: From biogenesis to function [Internet]. Vol. 141, Development (Cambridge). Company of Biologists Ltd; 2014. p. 3458–71.
- Cheng J, Guo JM, Xiao BX, Miao Y, Jiang Z, Zhou H, et al. piRNA, the new non-coding RNA, is aberrantly expressed in human cancer cells. Clin Chim Acta. 2011 Aug 17;412(17–18):1621–5.
- Y C, Q W, W J, Y B, Y Z, A G, et al. Emerging roles of piRNAs in cancer: challenges and prospects. Aging (Albany NY). 2019; 11(21): 9932–46.
- Vychytilova-Faltejskova P, Stitkovcova K, Radova L, Sachlova M, Kosarova Z, Slaba K, et al. Circulating PIWI-interacting RNAs piR5937 and piR-28876 are promising diagnostic biomarkers of colon cancer. Cancer Epidemiol Biomarkers Prev. 2018; 27(9): 1019–28.
- Weng W, Liu N, Toiyama Y, Kusunoki M, Nagasaka T, Fujiwara T, et al. Novel evidence for a PIWI-interacting RNA (piRNA) as an oncogenic mediator of disease progression, and a potential prognostic biomarker in colorectal cancer. Mol Cancer. 2018; 17(1).
- Weng W, Liu N, Toiyama Y, Kusunoki M, Nagasaka T, Fujiwara T, et al. Novel evidence for a PIWI-interacting RNA (piRNA) as an oncogenic mediator of disease progression, and a potential prognostic biomarker in colorectal cancer. Mol Cancer. 2018; 17(1).
- Yin J, Jiang XY, Qi W, Ji CG, Xie XL, Zhang DX, et al. piR-823 contributes to colorectal tumorigenesis by enhancing the transcriptional activity of HSF1. Cancer Sci. 2017; 108(9): 1746–56.
- Mai D, Zheng Y, Guo H, Ding P, Bai R, Li M, et al. Serum piRNA-54265 is considered a new biomarker for early detection of patients suffering from Colorectal Cancer. Theranostics. 2020; 10(19): 8468–78.
- Yin J, Qi W, Ji C, Zhang D, Xie X, Ding Q, et al. Small RNA sequencing revealed aberrant piRNA expression profiles in colorectal cancer. Oncol Rep. 2019; 42(1): 263–72.
- Mei Y, Clark D, Mao L. Novel dimensions of piRNAs in cancer [Internet]. Vol. 336, Cancer Letters. Elsevier Ireland Ltd; 2013. p. 46–52.
- Vychytilova-Faltejskova P, Stitkovcova K, Radova L, Sachlova M, Kosarova Z, Slaba K, et al. Circulating PIWI-interacting RNAs piR5937 and piR-28876 are promising diagnostic biomarkers of colon cancer. Cancer Epidemiol Biomarkers Prev. 2018; 27(9): 1019–28.
- Yin J, Jiang XY, Qi W, Ji CG, Xie XL, Zhang DX, et al. piR-823 contributes to colorectal tumorigenesis by enhancing the transcriptional activity of HSF1. Cancer Sci. 2017; 108(9): 1746–56.
- Chu H, Xia L, Qiu X, Gu D, Zhu L, Jin J, et al. Genetic variants in non-coding PIWI-interacting RNA and colorectal cancer risk. Cancer. 2015; 121(12): 2044–52.
- Martinez VD, Enfield KSS, Rowbotham DA, Lam WL. An atlas of gastric PIWI-interacting RNA transcriptomes and their utility for identifying signatures of gastric cancer recurrence. Gastric Cancer. 2016; 19(2): 660–5.
- Vychytilova-Faltejskova P, Stitkovcova K, Radova L, Sachlova M, Kosarova Z, Slaba K, et al. Circulating PIWI-interacting RNAs piR5937 and piR-28876 are promising diagnostic biomarkers of colon cancer. Cancer Epidemiol Biomarkers Prev. 2018; 27(9): 1019–28.
- Sasaki T, Shiohama A, Minoshima S, Shimizu N. Identification of eight members of the Argonaute family in the human genome. Genomics. 2003; 82(3): 323–30.
- Li L, Yu C, Gao H, Li Y. Argonaute proteins: potential biomarkers for human colon cancer. BMC Cancer. 2010; 10(1): 1–8.
- JH L, C J, P J-E, S S, DS, MS, et al. Pathways of proliferation and antiapoptosis driven in breast cancer stem cells by stem cell protein piwil2. Cancer Res. 2010 ; 70(11): 4569–79.
- Li D, Sun X, Yan D, Huang J, Luo Q, Tang H, et al. Piwil2 modulates the proliferation and metastasis of colon cancer via regulation of matrix metallopeptidase 9 transcriptional activity. Exp Biol Med. 2012; 237(10): 1231–40.
- Weng W, Li H, Goel A. Piwi-interacting RNAs (piRNAs) and cancer: Emerging biological concepts and potential clinical implications [Internet]. Vol. 1871, Biochimica et Biophysica Acta - Reviews on Cancer. Elsevier B.V.; 2019. p. 160–9.
- Wang H-L, Chen B-B, Cao X-G, Wang J, Hu X-F, Mu X-Q, et al. The clinical significances of the abnormal expressions of Piwil1 and Piwil2 in colonic adenoma and adenocarcinoma. Once Targets Ther. 2015; 8: 1259–64.
- Oh S-J, Kim S-M, Kim Y-O, Chang H-K. Clinicopathologic Implications of PIWIL2 Expression in Colorectal Cancer. Korean J Pathol. 2012; 46(4): 318.
- Tan Y, Liu L, Liao M, Zhang C, Hu S, Zou M, et al. Emerging roles for PIWI proteins in cancer. Acta Biochim Biophys Sin (Shanghai). 2015; 47(5): 315–24.