Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Acute undifferentiated/unclassifiable leukemia or disseminated non-hematologic neoplasm? When treatment is the compelling decision
Luca Frison1; Anna M Scattolin1; Matteo Leoncin1; Giorgio Farina2; Francesca Carobolante1; Cristina Salbe3; Pietro M Donisi4; Renato Bassan1*
1 UOC Ematologia, Ospedale dell’Angelo, Mestre Venezia, Italy
2 UOC Laboratorio Analisi, Ospedale dell’Angelo, Mestre Venezia, Italy
3 UOS Citogenetica/Biologia Molecolare, Ospedale Ss. Giovanni e Paolo, Venezia, Italy
4 UOSD Anatomia Patologica, Ospedale Ss. Giovanni e Paolo, Venezia, Italy
*Corresponding Author: Renato Bassan
UOC Ematologia, Ospedale dell’Angelo, MestreVenezia, Italy.
Email: renato.bassan@aulss3.veneto.it
Received : Oct 18, 2021
Accepted : Nov 29, 2021
Published : Dec 06, 2021
Archived : www.jcimcr.org
Copyright : © Bassan R (2021).
Keywords: acute undifferentiated leukemia; bone marrow metastasis; differential diagnosis.
Citation: Frison L, Scattolin AM, Leoncin M, Farina G, Bassan R, et al. Acute undifferentiated/unclassifiable leukemia or disseminated non-hematologic neoplasm? When treatment is the compelling decision. J Clin Images Med Case Rep. 2021; 2(6): 1456.
Introduction
The rapid collection of exact diagnostic and prognostic data is essential to guide therapy in patients diagnosed with acute leukemia. Basic diagnostic steps confirm the hematopoietic nature of the illness and allow distinction between Acute Lymphoid And Myeloid Leukemia (ALL and AML) throughout morphology, immunophenotype and cytogenetic/molecular analysis. However, because the complete results from the latter assays are not readily available, only morphology and Flow Cytometry (FC) permit to define quickly the cell lineage of the disease, thus orientating treatment. A small group of cases is referred to as Acute Undifferentiated Leukemia (AUL), because unclassifiable by current diagnostic markers [1]. AUL makes up 2-7% of Myeloperoxidase (MPO)-negative leukemias and poses a difficult treatment challenge [1,2]. The importance of a correct diagnostic input is further emphasized by the different results obtained with AML or ALL regimens in mixed lineage acute leukemia, another rare subset with debated diagnosis to treatment relationships [3]. Moreover, although rare in the modern era of improved diagnostic methodology, a pure leukemic presentation of an unrecognized metastatic carcinoma may need to be differentiated from AUL, AML or ALL, an issue which may be aggravated if FC results are inconclusive but treatment cannot be deferred until the availability of the other time-consuming diagnostic procedures.
Case report
A sixty-six year-old male was admitted to our Hospital because of anemia, thrombocytopenia, fever and hypotension. Hemoglobin was 8.8 g/dL, total white blood cells were within normal ranges (with neutrophils and monocytes 4.4 and 2.4 X 109 /L, respectively, and no circulating blast cell) and platelets 4 X 109 /L. Routine biochemistry assessing kidney [3] and liver function, as blood coagulation tests, were within normal ranges. Physical examination was unremarkable apart from the detection of spontaneous mucosal and cutaneous bleeding. No abnormal finding was detectable on Computed Tomography (CT) of head, neck, chest and abdomen. The patient had a previous history of neuroendocrine carcinoma of the nasopharynx and paranasal sinuses with localized nodal metastases, diagnosed and treated one year earlier, that was still in complete response after chemotherapy (cisplatin, 5-flurouracile, etoposide) and localized radiation therapy.
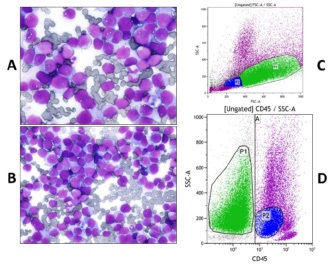
The patient bone marrow was almost totally repleted with immature, medium-sized cells of undifferentiated/monocytoid appearance, with high nucleo-cytoplasmic ratio, dispersed nuclear chromatin with inconspicuous nucleolus, occasional multinuclearity and basophilic agranular and at times vacuolated cytoplasm (Figure 1A and B). The FC analysis demonstrated two abnormal cell Populations (P) constituting the infiltrative marrow process (Figure 1C & 1D). P1 represented 50% of all gated cells and consisted of larger cells that were CD45 unreactive, minimally CD117 reactive (7%), partially HLA-DR positive (37.7%), CD56 positive (97%), and totally negative for all other markers (immufluorescence staining <3% for stem cell antigens CD34 and TdT; myelo-monocytic antigens cyMPO, CD13, CD33, CD4, CD11b, CD14, CD15, CD16, CD64, CD65; B- and T-lymphoid antigens CyCD79a, CD19, CD10, CD20, CyCD3, CD7, CD2, CD5; erythroid antigens CD235a, CD36, CD71; other antigens CD25, CD38). P2 consisted of smaller cells (15% of all gated cells) that expressed low-intensity CD45, minimally CD117 reactive (4%), weakly HLA-DR (17%) and CD11b (13%) positive, CD56 positive (66.2%) and negative for all other markers studied. Altogether, these findings, the past medical history and the absolute blood monocytosis raised the possibility of a secondary AUL, weakly CD45+, minimally CD117+, HLA-DR+ and CD56+, in an elderly patient who had previously been treated with chemotherapy and radiotherapy.
After discussion of the diagnostic issue, in agreement with the patient’s own and his relatives’ wish, AML-type chemotherapy was initiated because of the severity of the clinical picture dominated by the hemorrhagic risk. Our standard chemotherapy program for older adults with AML was started on day 3 from admission: mitoxantrone 10 mg/m2 /d intravenously (i.v.) on days 1-3, etoposide 100 mg/m2 /d i.v. and cytarabine 100 mg/m2 /bd i.v. on days 1-5, plus G-CSF from day 8. On day 6 the patient developed a hypertensive crisis that was soon followed by loss of consciousness, with myosis and right gaze deviation, resulting in a Glasgow Coma Scale equal to 5. A cerebral CT scan documented an extensive right frontal brain hemorrhage with ventricular discharge and left deviation of the midline structures. Craniotomy was performed, but despite an effective blood drainage the clinical status deteriorated and the patient died two days later in the intensive care unit.
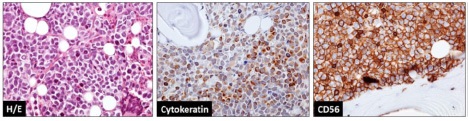
The bone marrow biopsy report was ready on day 8 from start of therapy (2 days following the patient’s death). The final diagnosis was diffuse metastatic neuroendocrine carcinoma with reactive marrow fibrosis (grade 2). Positive immunostaining was confirmed for MNF116 (cytokeratin) and CD56, while synaptophysin, chromogranin, S100 and hematopoietic cell markers including CD45, CD34, CD3, CD10, CD79a and nuclear TdT were all negative (Figure 2). Cytogenetics assay results were received on day 16, reporting a hyper-tetraploid karyotype (4n+[18]/46,XY[10]) in approximately 65% of the metaphases analyzed.
Discussion
The detection of carcinoma cells mimicking acute leukemia is a rare event. This phenomenon is named “Carcinocythemia” (CCA) when circulating tumor cells are detected, and needs to be differentiated from secondary acute leukemia due to chemotherapy and/or radiation therapy [4-6]. Generally, CCA occurs in far advanced stages of breast cancer, small cell lung carcinoma [5], and rhabdomyosarcoma [7] (Supplemental file), heralding refractoriness to treatment and a median interval between CCA diagnosis and death of only 14 days.
When only the bone marrow shows diffuse infiltration by neoplastic cells with uncertain morphology and immunophenotype, the differentiation from an AUL [1,2] is more difficult, again relying on a positive immuno-histochemical staining with antigenic cell markers expressed by either the primary tumor or leukemic cells [5]. The grade of diagnostic ambiguity is even higher if there is no apparent clinical evidence of an associated metastatic solid tumor and becomes of special concern when the clinical condition mandates for a prompt start of treatment, a challenge we had to deal with in the present case.
This 66 year-old patient was initially managed as having AUL/ ambiguous lineage leukemia but had a final diagnosis of massive marrow dissemination of a neuroendocrine malignancy that had been assessed in complete remission. On presentation, he had a massive leukemia-like marrow involvement by blast-looking cells expressing an unclear immunophenotype that suggested AUL/unclassifiable secondary acute leukemia with weak CD45 expression and positive CD56 antigen expression. CD56 is widely expressed across normal and malignant myelo-monocytic and lymphoid cells, but is also compatible with the final diagnosis, while the very weak CD45 expression could be possibly related to autofluorescence or other technical reason. The cytokeratin plus CD56 antigen staining of a core marrow biopsy confirmed a metastatic neuroendocrine tumor, and ruled out any CD45 antigen expression.
Once the final diagnosis was made the patient had already died of intracranial hemorrhage. This was in keeping with the very short survival reported for CCA and highlights the urge we had to treat him without any delay because of a rapidly deteriorating clinical picture and without any clinical evidence of locoregional or distant recurrence of his previous neuroendocrine cancer. With regard to the pathogenesis and diagnosis of bone marrow involvement by CCA, it is known that primary tumors can create pre-metastatic marrow niches through an interplay among tumor-derived factors, tumor-mobilized bone marrowderived cells, macrophages, mesenchymal stem cells and local stromal components [8-10]. From the diagnostic standpoint, immunostaining for epithelial markers such as EpCAM, cytokeratins, human epidermal growth factor receptor 2, estrogen receptors and immune-checkpoint molecules is essential to detect even small amounts of CCA in marrow or blood samples [9].
Conclusion
In conclusion, because the differential diagnosis of a suspected acute leukemia may be difficult in cases with unclear and/or poor surface markers expression, the application of these tests in flow cytometry should allow to identify rapidly an unrecognized CCA in a patient with a prior history of solid tumor still in apparent clinical remission.
References
- Van’t Veer MB. The diagnosis of acute leukemia with undifferentiated or minimally differentiated blasts. Annals of Hematology. 1992; 64: 161-165.
- Lee E, Pollak A, Leavitt R, Testa J, Schiffer C, et al. Minimally differentiated acute nonlymphocytic leukemia: A distinct entity. Blood. 1987; 70: 1400-1406.
- Matutes E, Pickl WF, Veer MVNT, et al. Mixed-phenotype acute leukemia: Clinical and laboratory features and outcome in 100 patients defined according to the WHO 2008 classification. Blood. 2011; 117: 3163-3171.
- Myerowitz RL, Edwards PA, Sartiano GP. Carcinocythemia (carcinoma cell leukemia) due to metastatic carcinoma of the breast. Report of a case. Cancer. 1977; 40: 3107-3111.
- Gallivan MVE, Lokich JJ. Carcinocythemia (carcinoma cell leukemia). Report of two cases with english literature review. Cancer. 1984; 53: 1100-1102.
- Rowan DJ, Logunova V, Van Tuinen P, Olteanu H, Peterson JF. Circulating Breast Carcinoma Cells Mimicking Therapy-Related Acute Myeloid Leukemia: A Potential Cytogenetic and Flow Cytometry Pitfall. International Journal of Surgical Pathology. 2017; 25: 87-93.
- Misawa R, Kobayashi M, Ito M, Kato M, Uchikawa Y, Takagi S. Primary Colonic Signet Ring Cell Carcinoma Presenting Carcinocythemia: An Autopsy Case. Case Reports in Gastroenterology. 2008; 2(3): 301-307.
- Liu Y, Cao X. Characteristics and Significance of the Pre-metastatic Niche. Cancer Cell. 2016; 30: 668-681.
- Masuda T, Hayashi N, Iguchi T, Ito S, Eguchi H, Mimori K, et al. Clinical and biological significance of circulating tumor cells in cancer. Molecular Oncology. 2016; 10: 408–417.
- Shiozawa Y, Eber MR, Berry JE, Taichman RS. Bone marrow as a metastatic niche for disseminated tumor cells from solid tumors. BoneKey Reports. 4.