Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
The use of low dose methylprednisolone in severe Covid-19 induced ARDS to facilitate oxygen weaning: Results from a case series and mini review
E Doherty; L Ilves; C Cheeld; A Elradi; VA Varney*
St Helier Hospital, Wrythe Lane, Carshalton, Surrey SM5 1AA, UK.
*Corresponding Author: Veronica Varney
Department of Respiratory Medicine, St Helier Hospital,
Wrythe Lane, Carshalton, Surrey SM5 1AA, UK.
Email: veronica.varney@btinternet.com
Received : Sep 27, 2021
Accepted : Dec 01, 2021
Published : Dec 08, 2021
Archived : www.jcimcr.org
Copyright : © Varney V (2021).
Abstract
We describe 8 patients with confirmed Covid-19 infection requiring high flow oxygen and respiratory support for adult Respiratory distress syndrome (ARDS) for a mean duration of 12 days. These patients had already received standard treatments for Covid-19, including broad spectrum antibiotics, dexamethasone, heparin and remdesivir along with entry to the recovery trial according to patient choice. Due to on-going high oxygen demands and respiratory support, these patients were at increasing risk of significant lung injury and the development of fibrotic lung changes. Intravenous low dose methylprednisolone was therefore given in an attempt to assist resolution of the ARDS. The low dose was chosen to reduce the cytokine storm and minimise the risk of a super-infection. This led to improvement in 6 out of 8 patients allowing oxygen weaning (mean duration 4.5 days) and discharge home within a mean duration of 11 days. Five patients were discharge home with low flow oxygen (mean 2.8 L/min) with community based weaning. The 2 non-responders subsequently deteriorated further and were managed palliatively. We describe the cases and discuss the current literature with regard to the use of methylprednisolone in Covid-19 ARDS.
Keywords: ARDS; Covid 19; methylprednisolone; respiratory failure; fibrotic lung injury.
Abbreviations: ARDS: Adult Respiratory Distress Syndrome; CPAP: Continuous Positive Airway Pressure; Optiflow: High Flow Nasal Oxygen Device; Po2: Arterial Oxygen Level; CT Scan: Computerised Tomography Scan; TNFα: Tumour Necrosis Factor –Alpha; SARS: Severe Acute Respiratory Syndrome; MERS: Middle East Respiratory Virus; MP: Methylprednisolone; CRP C: Reactive Protein; A&E: Accident & Emergency Dept.
Citation: Doherty E, Ilves L, Cheeld C, Elradi A, Varney V. The use of low dose methylprednisolone in severe Covid-19 induced ARDS to facilitate oxygen weaning: Results from a case series and mini review. J Clin Images Med Case Rep. 2021; 2(6): 1461.
Introduction
Forty percent of admitted patients with Covid 19 develop adult respiratory distress syndrome (ARDS); in 20% of these cases ARDS is severe [1]. Severity is linked to increasing age and co morbid factors such as obesity, diabetes and ischaemic heart disease [2,3]. The viral-mediated mechanisms implicated in causing ARDS, include chronic inflammation related to the cytokine storm from a dysregulated immune response [3]. This phase is exudative with hyaline membrane formation that can give diffuse alveolar damage, followed by an organising phase and a fibrotic phase [4]. Epithelial and endothelial injury occurs in the inflammatory phase of ARDS due to the release of proteases and cytokines such as matrix metalloproteases and vascular endothelial growth factor along with interleukin-6 and TNF-α (tumour necrosis factor -α) [4].
ARDS is the main predictor of pulmonary fibrosis in Covid 19. The exact extent of post-Covid fibrotic injury will become apparent with time, but the data so far suggests that up to 30% of hospital discharges show fibrotic abnormalities post ARDS [5,6]. Previous studies suggest that the duration of ARDS is an important determinant of fibrotic lung injury [5]. In post-Covid ARDS lasting <21 days, up to 24% of patients show some fibrotic changes, while 61% of post-Covid-ARDS lasting >21 days develop fibrosis [7]. It is unclear as to whether high levels of oxygen use in ARDS plays a role in the fibrotic injury through the generation of reactive oxygen radicals, or if genetics and other biological factors are more important [4,8].
Currently there are no established treatment pathways for prolonged ARDS in Covid-19, and it is yet to be established whether anti-viral’s, anti-inflammatory drugs or anti- fibrotic drugs may reduce the development of fibrotic injury, and reduce remodelling [4,5,9].
In this case series, we present 8 patients with Covid 19-induced ARDS with significant radiological airspace changes for a mean period of 12 days, increasing their risk of fibrotic lung injury. These patients were pulsed with relatively low doses of intravenous methylprednisolone for up to 3 doses. This was in an attempt to hasten resolution of the ARDS and allow oxygen weaning to off-set the risk of fibrotic lung injury and progressive ventilatory failure. We describe the cases, their response to this treatment and review the recent literature on methylprednisolone in Covid -19 induced lung injury.
Case series
Case I
A 58 year old lady was admitted with a 5-day history of worsening shortness of breath and lethargy. She was hypoxic with saturations of 86% on air, and raised inflammatory markers C - reactive protein (CRP) of 71 mg/L (normal <5.0). Her chest x-ray showed patchy consolidation in the lower zones suggesting a viral pneumonitis and her COVID-19 swab was positive. Oxygen was commenced via a reservoir bag mask at 15 L/min, despite this her arterial pO2 was only 7.7 KPa and CPAP (continuous positive airway pressure) was commenced (6 cms H2 0) with an oxygen flow of 15 L/min. Intravenous Benzyl penicillin, clarithromycin and prophylactic dalteparin were commenced along with dexamethasone 6mgday following randomisation to the Recovery Trial.
Her past medical history was rheumatoid arthritis, for which she took hydroxychloroquine. She also had hypertension, a hiatus hernia and anxiety. She was normally independent despite her rheumatoid with a 40-pack year smoking history.
CPAP continued for 9 days, but pressure was gradually increased from 6 to 12 cms water. Despite this oxygen saturations and pO2 remained low. Acute chest pain developed but cardiac troponins and serial ECG’s were normal. Her D-dimer was raised >6000 (μg/L) therefore full treatment dose dalteparin was commenced. Her CT pulmonary angiogram excluded central pulmonary emboli but confirmed bilateral ground glass change. A decision was made to continue full treatment dose dalteparin due to high risk for micro-emboli. On day 10, her oxygen was changed to Optiflow (at 60%; 60 L/ min) but her requirement increased to 90% at 60 l/min rapidly. A return to CPAP was not tolerated and discussions with her and the Intensive Care doctors led to a decision to avoid invasive ventilation and to continue with optiflow.
On day 13, a 3-day regime of intravenous Methylprednisolone (MP) was commenced starting at 40 mg on day 1, 125 mg on day 2 and 250 mg on day 3. Immediately prior to these infusions, saturations were only 89% with Optiflow treatment (p02 of 7.5 KPa). By the 3rd infusion saturations had improved to 92% on optiflow (FiO2 47%:60 L/min). This improved further and at day 4 her arterial pO2 was 11.6 KPa on nasal cannulae at 4 L/ min.
Intravenous hydrocortisone 50 mg TDS continued for 72 hours followed by a regime of oral prednisolone (30 mg for 1 week, 20 mg 1 week, 10 mg 1 week then stop). This facilitated complete weaning of optiflow at day 6 following the start of MP leading to discharge with 1 litre/min nasal oxygen that was weaned off in the community.
Case II
A 55 year old lady presented with a 2-week history of progressive shortness of breath, a productive cough and diarrhoea. She was normally healthy until her family members tested positive for COVID-19. Upon admission saturations were only 50% on air and she required 15 L/min via a Hudson mask to achieve saturations of 88%. Her chest-x-ray showed bilateral pulmonary infiltrates and her CT pulmonary angiogram confirmed the presence of pulmonary emboli with extensive bilateral ground glass shadowing in keeping with Covid 19 pneumonitis. Her inflammatory markers, were raised (CRP 174 mg/L) along with a ddimer of >6000 μg/L; so full-dose dalteparin was commenced along with broad spectrum antibiotics. Entry to the recovery trial was declined.
Arterial gases showed a pO2 of 5.9 KPa on 15 Lmin (Hudson mask) therefore CPAP was commenced (8 cms H2 O; 15 L/min). CPAP continued for 10 days with short breaks and proning. Despite this, hypoxaemia continued (pO2 of 8.7 KPa).
On day 10 she was commenced on a 3-day course of intravenous MP regime (40 mg on day 1, 125 mg on day 2 and 250 mg on day 3). Improvement was seen by day 2 when CPAP was changed to optiflow (FiO2 50%, 40 L/min) maintaining her saturations at 94% but further arterial gas measurement was declined.
Following the intravenous MP , improvement occurred over a further 10 days with oxygen weaned down to 2L/min via nasal cannulae leading to discharge (day 24) with continued nasal oxygen (2L/min) successfully wean off in the community.
Case III
An 80 year old man was admitted with shortness of breath and malaise along with oxygen saturations of only 70% on air and a chest X-ray showing bilateral patchy opacification. His inflammatory markers were raised with a CRP of 161 mg/l and a swab positive for COVID-19.
His past history included type 2 diabetes, hypertension, glaucoma and osteoarthritis. He had never smoked and was independent in activities of daily living.
He was immediately commenced on high flow oxygen (15 L/ min) via a reservoir bag mask. Arterial blood gases showed: pH 7.44, pCO2 4.5 KPa and pO2 8.5.KPa. Standard treatment for COVID-19 pneumonitis including dexamethasone, remdesivir, and broad spectrum antibiotics (tazocin and clarithromycin) were given. Due to ongoing hypoxia CPAP was commenced (10 cms H2 O; 15 L/min) with proning. CPAP pressures were increased (14 cms H2 O;15 L/min oxygen) to maintain oxygen saturations. Attempts to wean CPAP led to de-saturations to down to 85% and during CPAP-breaks saturations fell as low as 70% despite high flow oxygen.
During this time he was comfortable at rest, but short of breath on minimal movements. On day 6 following admission, he was commenced on full dose dalteparin in case of pulmonary micro-emboli.
At day 10 arterial gases showed pH 7.46, pCO2 4.82 KPa and pO2 7.4 KPa with continued CPAP (14 cms, 15 L/min). An additional antibiotic was added (Teicoplanin for 7 days).
At day 12, a CT pulmonary angiogram excluded pulmonary emboli but confirmed severe COVID pneumonitis with interlobular septal thickening suggestive of fibrotic change despite completing 10 days of dexamethasone.
Methylprednisolone intravenously was trialled: using 40 mg on day 13, 125 mg on day 14, and 250 mg on day 15. Upon completion a repeat arterial blood gas taken on 12 L/min oxygen (reservoir bag mask) demonstrated improvement pO2 10.9 KPa. Further oral prednisolone 40 mg commenced with nocturnal CPAP only (14 cms; 10-15 L/min).
By day 17, his CRP rose to 73 mg/L and a repeat Chest X ray confirmed a new right upper lobe consolidation, so meropenem was commenced for a hospital-acquired pneumonia. Despite initial improvement, by day 21 shortness of breath had increased again. Discussions with his family established maximal therapy would be ward-based as invasive ventilation was not advisable. Therapy was simplified with help from palliative care using supplemental oxygen without CPAP and he died peacefully
Case IV
A 60 year old man attended A&E and tested positive for COVID-19 with cough and shortness of breath without hypoxia and was therefore discharged with oral antibiotics. Subsequently his dyspnoea increased with saturations of 70% on air and fever leading to hospital admission. His past medical history was benign prostatic hyperplasia with a past cholecystectomy; he had never smoked.
His chest X-ray showed widespread air space opacification consistent with viral pneumonitis and his CRP was raised at 70 mg/l (normal <5.0).
Arterial blood gases on 15 L/min of oxygen via a Hudson mask showed: pH 7.5, pCO2 4.52 KPa, pO2 7.6 KPa with saturations of 88-92% at this flow rate.
He commenced dexamethasone and antibiotics (tazocin and clarithromycin) and remdesivir. CPAP was commenced (10 cms H2 O: 15 L /min oxygen), improving saturations to 96% (Arterial pO2 10.2 KPa). Treatment dose dalteparin was commenced in view of his D-dimer >6000 μg/L as he was not sufficiently stable for a CT pulmonary angiogram.
On day 1, saturations varied between 81-91% despite CPAP (12 cms; 15 L/min). He found CPAP difficult to tolerate so pressure was varied (8-14 cms H2 O) with 4 hour breaks to Optiflow (60 L/min: FiO2 70%).
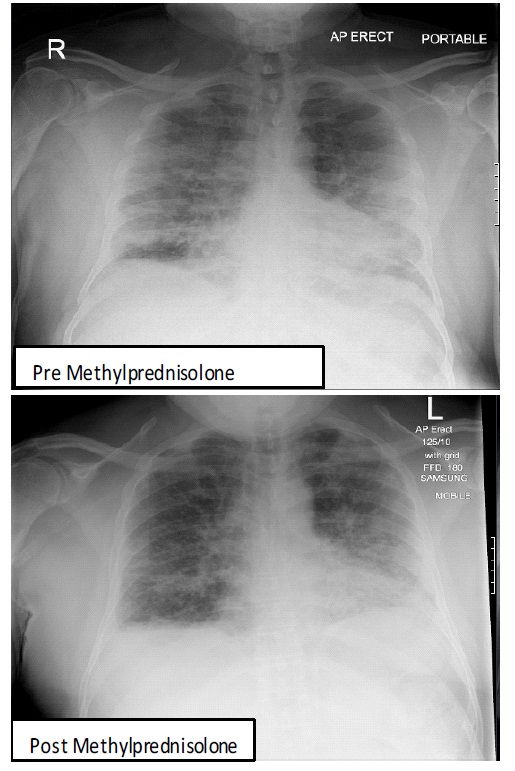
On day 6, following admission, MP was commenced intravenously due to high oxygen requirements that were failing to improve. He received 40 mg on day 6, 125 mg on day 7 and 250 mg on day 8. Following the 3rd dose of methylprednisolone, intermittent CPAP support was discontinued and Optiflow continued. Figure 1 shows the chest-X-ray pre and 10 days post MP.
A high resolution CT chest on day 9 confirmed moderately severe viral pneumonitis with early fibrotic change. Prednisolone 40 mg once daily for 5 days commenced with further oxygen weaning to a Venturi mask (FiO2 60%) at day 13. Prednisolone was reduced to 20 mg/day and reduced by 5 mg every 7 days to zero. By day 24, oxygen requirement was 4 L/min (nasal cannulae) with saturations 93% at rest but falling to 65-75% on mobilising . He was discharged with nasal oxygen which weaned off completely within 8 weeks.
Case V
A 91 year old man was admitted with two weeks of cough, progressive breathlessness and anosmia. His wife had tested positive for COVID-19 two weeks beforehand. On admission he was profoundly hypoxic (p02 6.67 KPa) requiring oxygen at 15 L/min via a Hudson mask. His chest x-ray showed bilateral infiltrates in keeping with Covid pneumonitis. A CT pulmonary angiogram showed a right lower lobe pulmonary artery embolus and bilateral patchy consolidation. CRP was raised (118 mg/L ). CPAP was commenced (12 cms: 15 L/min).
His past medical history was type-2 diabetes mellitus and glaucoma. He was independent in activities of daily living. He had never smoked.
Intravenous antibiotics, dexamethasone, remdesivir and treatment dose dalteparin were commenced. He entered the recovery trial and randomised to standard care. A ward based ceiling of care was established as intubation and resuscitation were deemed inappropriate.
On day 5, his fevers increased. CPAP continued as before and antibiotics were changed to meropenem. At day 12 his chest xray appearances had worsened so vancomycin was added.
By day 22 his oxygen requirement continued at 15 L/min with nocturnal CPAP. An echocardiogram estimated pulmonary artery systolic pressure of 29-34 mmHg and left ventricular ejection fraction 55%. A second CT chest scan showed worsening radiological appearances, with increased bilateral subpleural reticular changes suggesting fibrotic injury. His CRP had reduced to 55 mg/L.
A 3 day regime of intravenous MP started on day 23, with 40 mg, 125 mg on day 24 and 250 mg on day 25. Following his 2nd dose of MP his oxygen requirement began to decrease, allowing weaning to a Venturi mask (40%) and nocturnal CPAP support was discontinued.
On day 27, his oxygen was further reduced to Venturi mask 35%. Home oxygen was arranged and discharge occurred on day 28 with nasal oxygen at 5 l/min that was completely weaned off in community.
At 3 months, a repeat CT chest showed a significant reduction in consolidation with bilateral upper lobe fibrotic change and traction bronchiectasis.
Case VI
A 48 year old lady was admitted with a 6 day history of breathlessness and a dry cough following contact with COVID-19. She was unvaccinated, with a past medical history of asthma, hypertension, type-2 diabetes and obesity. She had never smoked.
She was hypoxic with oxygen saturations of 66% on room air, and commenced 15 L/min oxygen via Hudson mask giving an arterial oxygen valve of only P02 7.4 KPa. CPAP was commenced (10 cms; Fi02 70%). Her chest x-ray showed extensive pulmonary infiltrates consistent with covid-19 pneumonitis. She was treated with regular nebulised salbutamol and ipratropium in addition to intravenous remdesivir, tazocin and clarithromycin and oral dexamethasone. She was transferred to the high dependency unit. Serum CRP level was raised at 152 mg/L.
On day 2, CPAP was continued (10 cms; 12 L/min). Her procalcitonin level was normal (0.2 ug/L) and therefore intravenous sarilumab (an anti-interleukin-6 monoclonal antibody) was administered as per recovery -3 trial randomisation.
At day 3 a CT pulmonary angiogram excluded pulmonary emboli and confirmed diffuse bilateral pulmonary infiltrates consistent with covid-19 pneumonitis and ARDS. A transthoracic echocardiogram was normal.
Oxygen requirements remained high without weaning despite treatment for 9 days. Following discussion with the respiratory team, intravenous MP commenced, with 40 mg on day 9 and 80 mg MP on day 10.
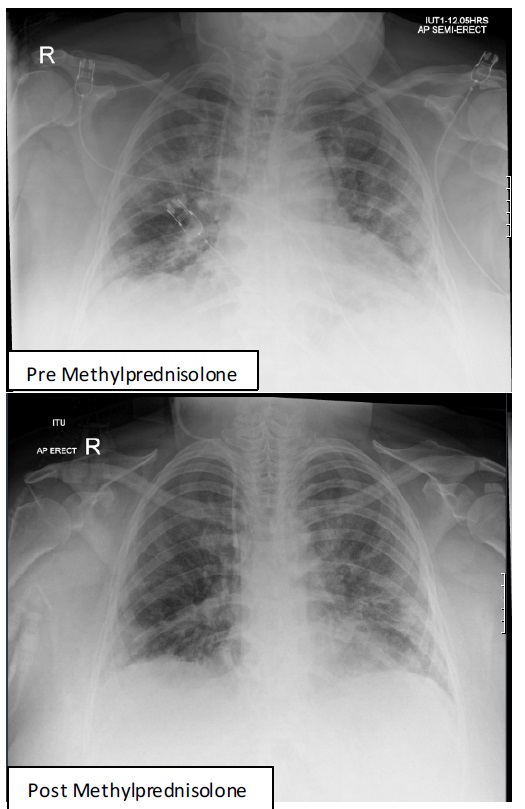
At day 10, she showed significant signs of improvement with reduced oxygen requirements allowing transfer to the respiratory ward. With CPAP replaced by Optiflow, (Fi02 35%; 40 L/ min). Figure 2 shows the chest-X-ray pre and 4 days post MP. Oral prednisolone 40 mg/d commenced on day 11 and reduced 5 mg every 7 days to zero. At day 33, she was discharged on nasal oxygen (2 L/min) with weaning in community
Case VII
A 48 year old man was admitted with 7 days of fever, decreased appetite, myalgia, breathlessness with a productive cough for 2 days. A Covid 19 swab had been positive 5 days previously. His past medical history included mild asthma, type 2 diabetes and gout. He had not had a Covid vaccine.
In A&E, his oxygen saturations were 84% on room air and he required 15 L/min oxygen (Hudson mask) to obtain oxygen saturations of 93%. His chest was wheezy on auscultation. Arterial blood gas showed pO2 5.1 KPa on 15 L/min oxygen. CRP was elevated (75 mg/L) and a chest x-ray showed bilateral pulmonary infiltrates.
CPAP was commenced (10 cms; 15 L/min) along with intravenous ceftazidime and clarithromycin,. Regular nebulised salbutamol, dexamethasone and remdesivir were added and he was transferred to intensive care unit for closer monitoring.
On day 2 of admission he received tocilizumab.
Between days 1-7, CPAP continued at the same settings with intermittent optiflow used during meal breaks. On day 8, a CT pulmonary angiogram confirmed Covid pneumonitis with a pneumo-mediastinum, probably secondary to asthma and barotrauma from CPAP. Therefore he continued with Optiflow (Fi02 75%,; 60 L/min) only.
On day 11 the patient was reviewed by the respiratory team and intravenous MP 40 mg was administered intravenously. At day 12, his oxygen requirement had significantly reduced allowing weaning to a 35% Venturi mask with oxygen saturations 95%.
Arterial P02 had improved to 10.5 KPa on this flow rate and no further intravenous pulses of MP were deemed necessary.
Oral prednisolone 40 mg/d was commenced with transfer to the respiratory ward where his chest was clear of wheeze and nebulizers stopped.
Within 48 hrs (day 15) he was oxygen free and discharged home to complete prednisolone weaning by 5 mg every 5 days to zero.
Case VIII
A 76 year old lady with a 3 week history of fatigue, anorexia and dry cough was admitted febrile (38.1oc) without hypoxia (saturations 95% on air).
She tested positive for Covid-19 despite being fully vaccinated for Covid several months before. Her chest x-ray was clear. CRP was 22.5 mg/L. She had a background of Waldenstrom’s macrogloblinaemia (on active chemotherapy) and osteoporosis.
On day 2 of admission, mild hypoxia required 3 L/min nasal oxygen via nasal cannulae. Intravenous tazocin, clarithromycin, remdesivir and dexamethasone were commenced. At day 7 hypoxia increased, requiring oxygen at 15 L/min (Hudson mask). This corrected oxygen saturations to 88% with a measured arterial oxygen of PO2 7.2 PKa.
A CT pulmonary angiogram excluded pulmonary emboli but confirmed severe ground glass opacities consistent with a Covid pneumonitis. She was transferred to the intensive care unit to commence CPAP (6 cms; 15 L/min ).
CPAP continued between day 8-14 (8 cms; 15 L/min). A repeat CTPA showed increased ground glass changes with a small right apical pneumothorax and pneumo-mediastinum. At day 14 she required mechanical ventilated. Her antibiotics were escalated to intravenous meropenem.
On day 19, after discussion with respiratory team she was commenced on a 3 day regime of intravenous MP, 40 mg was administered on day 19, 125 mg on day 20 and 250 mg on day 21.
She developed multi-organ failure (acute kidney, liver and respiratory) and support was withdrawn following discussions with family. She died on day 25.
Discussion
In 1967 an association between respiratory failure and noncardiac pulmonary oedema was first described, leading to the name “adult respiratory distress syndrome or ARDS”. Studies have suggested that the condition is related to inadequate cellular oxygenation which can lead to multisystem failure necessitating a wide range of therapeutic support. The clinical picture is that of refractory hypoxaemia, unresponsive to an increasing inspired oxygen fraction with reduced lung compliance and bilateral pulmonary infiltrates without raised pulmonary wedge pressure [1]. Mortality is high at 50-60%. The condition can follow a wide variety of insults to the pulmonary vascular epithelium or alveolar epithelium directly (toxic fumes, aspiration of gastric contents, viral infections) or indirectly (via sepsis, massive haemorrhage, blood transfusion, major trauma, obstetric crisis) [1].
Large numbers of neutrophils have been isolated from bronchoalveolar lavage fluid in patients with ARDS. In animals depleted of neutrophils, there is attenuated responses and reduced oedema [1]. Despite evidence of a key role for neutrophils, the precise mechanism underlying their action is only partly understood. The complement system is stimulated in many disorders associated with ARDS and can increase vascular permeability if given experimentally to animals without other effects on the lungs. Chemotactic mediators for neutrophils (TNF-α, interleukin-1, 6, and 8) successfully recruit neutrophils to the lung, leading to the generation of toxic oxygen free radicals with damage to the capillary endothelium giving alveolar oedema from disturbed vasomotor control along with, fibroblast replication and collagen deposition that forms the basis of ARDS [10,11].
There has been considerable research on ARDS in the past decade with better understanding of its pathogenesis. Despite this, there are limited therapeutic measures to decrease mortality in ARDS except for using low-tidal volume ventilation, prone ventilation for severe cases; and Extra Corporeal Membrane Oxygenation rescue technology in severe cases [11].
No clear benefit of specific drug therapy has been established apart from antibiotics. Steroids traditionally used for their anti-inflammatory effects are not shown to improve survival and to be harmful in some situations leading largely to their discontinuation in this condition [11].
In the SARS (severe acute respiratory syndrome) and MERS (Middle East respiratory syndrome) outbreak in 2003, the same risk factors of advanced age and co-morbid diseases were associated with severe disease [9]. ARDS was associated with increased mortality and steroid use in SARS and MERS gave mixed results and delayed viral clearance [12-16]. Radiological abnormalities were noted in both diseases and associated with severity and the duration of mechanical ventilation. Fibrotic changes in SARS were noted in 9.4% of severe cases in the early recovery period that decreased to 3.2% over a 15 yr. follow-up suggesting absorption of some damaged tissue with time. Similar observations were seen with MERS [7,9,17].
To date there is no long-term data for Covid-19 induced fibrotic lung injury, but concerns exist due to the potential morbidity given the global infection rate. Current data shows fibrotic change in 72% of cases after mechanical ventilation, compared with 20% without along with a linear relationship of fibrotic changes with the duration and severity of the ARDS [7]. Management of fibrotic lung injury in Covid-19 is currently unexplored as no clinical trials exist and treatment is symptomatic [4,18].
The UK ‘National Recovery Trial‘ gave the first indication that the steroid dexamethasone (6 mg/day) showed some benefit in oxygen dependant Covid-19 patients and reduced mortality in ventilated patients with ARDS by 11% [11,12,19]. There are suggestions from CT scan findings that Covid-19 infection does not produce a “classical ARDS” and may represent an endothelial cell injury in contrast to the usual alveolar cell injury, but details are sketchy [9]. The positive effect of dexamethasone in Covid-19 has led to reported cases of ARDS being treated with Methylprednisolone (MP). There are so far 7 small randomised studies published this year, with benefit demonstrated from the meta-analysis [1,12]. The doses used were low and ranged between 80-250 mg/day for 3-7 days. There was clear evidence of benefit for oxygen dependant Covid-19 patients who were developing a respiratory injury or ARDS.
The findings suggested that MP in the earlier stages of respiratory injury reduced mortality by 71%, along with the need for ITU admission and ventilation [1,20]. MP gave faster improvement in arterial oxygen levels and allowed weaning in 55% of cases by day 3 compared with 21% on standard care without MP [1]. C-reactive protein and interleukin-6 levels also fell quickly and the need for mechanical ventilation was reduced to 8.7% in MP-cases with ARDS versus 33% with standard care [1].
This response was better than that seen in the Recovery Trial and may relate to the optimal lung penetration of MP giving near complete saturation of the glucocorticoid receptors. Dexamethasone has a longer half-life (36-72hrs) compared with MP (12-36 hrs), but the MP dose used was the equivalent of 12-24mg of dexamethasone [12,13,19]. The avoidance of prolonged respiratory support along with shortening of the ARDS illness would suggest that fibrotic injury should be reduced also. This will need a longer term follow-up of treated cases to be certain.
In our cases series, treatment with MP was commenced later into the ARDS injury, when oxygen requirements were high and respiratory support on-going for a mean of 12 days without demonstrated improvement. Six cases show noticeable improvements following MP (mean of 4.5 days) with weaning of oxygen and ventilatory support leading to discharge (mean period 11 days). Five cases required low flow nasal oxygen upon discharge with subsequent weaning in community. Our treatment was initiated, due to the lack of clinical progress in our cases and without other published data on MP available. Relatively low doses of MP were used in our cases due to the natural anxiety of inducing a super-infection. Prednisolone followed MP in 4 cases, two were asthmatic and the others showed slower improvements after MP.
The published case reports are now showing benefit from low dose MP treatment without significant adverse effects, along with a suggestion that earlier treatment of Covid induced ARDS will avoid mechanical ventilation and earlier oxygen weaning [1]. Theoretically this may also reduce the risk of fibrotic scarring from protracted ARDS. At present, the long-term persistence of the interstitial lung changes seen in our patients secondary to their ARDS is unknown. Resolution as seen in SARS and MERS may possibly occur with time. Overall the data from MP use suggests that larger trials of treatment in hospitalised patients with Covid-19 induced ARDS may show benefit in mortality, reduced mechanical ventilation and length of hospital stay, with these outcomes hopefully reducing the possibility of fibrotic lung injury. All patients recently contacted at home report themselves as well.
Declarations
No Ethics Committee approval was required for this reported case series. Written and verbal consent was obtained from the patients. The data described can be available on individual request. No external funding was used in the treatment of these National Health Service patients. The authors report no conflict of interest.
References
- Edalatifard M, Akhari M, Salehi M, Naderi Z, Jamshidi A, et al. Intravenous methylprednisolone pulse as a treatment for hospitalised severe COVID-19 patients: Results from a randomised controlled clinical trial. Eur Respir J. 2020: 562002808.
- Wu C, Chen X, Cai Y et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan China. JAMA Intern Med, 2020; 180: 934-943.
- Lui X, Zhou H, Zhou Y et al. Risk factors associated with disease severity and length of hospital stay in COVID-19 patients. J Infect. 2020; 81(10): E95-97.
- Alhiyari MA, Ata F, Alghizzawi MI, Bilala AB, Abdulhadi AS, et al. Post-COVID-19 fibrosis, an emerging complication of SARSCoV-2 infection. IDCases 2021; 23: e01041.
- Scelfo C, Fontana M, Casalini E, Menzella F, Piro R, et al. A dangerous consequence of the recent pandemic: Early lung fibrosis following COVID-19 pneumonia-Case reports. Therapeutics and clinical risk management. 2020: 16; 1039-1046.
- Sheng g, Chen P, Wei Y et al. Viral infection increases the risk of idiopathic pulmonary fibrosis: A meta-analysis. Chest. 2019.
- Mcgroder CF, Zhang D, Choudery MA, Salvatore MM, et al. Pulmonary fibrosis 4 months after Covid-19 is associated with the severity of the illness and blood leucocyte telomere length. Thorax. 2021.
- George PM, Wells AU, Jenkins RG. Pulmonary fibrosis and COVID-19: the potential role of anti-fibrotic therapy. Lancet respire Med. 2020: 8; 807-815.
- Rai DK, Sharma P, Kumar R. Post Covid 19 pulmonary fibrosis. Is it a real threat?. Indian J of TB. 2021: 68; 330-333.
- Stockman LJ, Bellamy R, GarnerP. SARS; Systematic review of treatment effects. PLOS Med: 2006; 3; e343.
- Rawal G, Yadar S, Kumar R. Acute Respiratory distress syndrome: An update and review. J Translational Int Med. 2019; 6; 74-77.
- NIH US National Library of Medicine-Clinical Trials.gov. Methylprednisolone for patients with Covid-19 severe acute respiratory syndrome. March 2020.
- Hamed DM, Belhoul KM, Al Maazmi NA, Ghayoor F, Moin M, et al. Intravenous methylprednisolone with or without tocilizumab in patients with severe COVID-19 pneumonia requiring oxygen support: A prospective comparison. J infection and public health. 2021; 14: 985-989.
- Sung JJY, Wu A, Joynt GM etal. Severe acute respiratory syndrome : report of treatment and outcome after a major outbreak. Thorax. 2004; 59: 414-420.
- Arabi YM, Mandourah Y, Al-Hameed F, et al, Corticosteroid therapy for critically ill patients with middle east respiratory syndrome. Am J respire Crit Care Med. 2018; 197: 757-767.
- LeeN, Chan KC, Hui DS et al. Effects of early corticosteroid treatment on plasma SAR-associated coronavirus RNA concentration in adults patients. J Clin Virol. 2004: 31: 1247-1249.
- Das KM, Lee EY, Singh R. Et al. Follow-up chest radiographic findings in patients with MERS-CoV after recovery. Indian j Radiol Imag. 2017; 27: 342-349.
- Gentile F, Aimo A, Forfori F et al. Covid-19 and risk of pulmonary fibrosis: the importance of planning ahead. Eur J Prev Cardiol. 2020; 27; 1442-1446.
- G- Pinzon MA, Ortiz S, Holguin H, Betancur JF, Arango DC, et al. Dexamethasone VS methylprednisolone high dose for Covid-19 pneumonia. PLOS ONE. 2021.
- . Salton F, Confalonieri P, Meduri GU, Santus P, Harari S, et al. Prolonged low-dose methylpredisolone in patients with severe COVID-19 pneumonia. Open forum infectious disease. 2021. DOI 10.1093/ofid/ofaa421.