Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Responding well to corticosteroid in an acitretin treatmentRelated acute lung injury patient with psoriasis
Min Cao1; Hui Li1,2*; Yong Long Xiao1; Hou Rong Cai1
1 Division of Pulmonary and Critical Care Medicine, The Affiliated Drum Tower Hospital, Nanjing University Medical School
2 The Seventh Affiliated Hospital of Sun Yat-Sen University, China.
*Corresponding Author: Hui Li
Division of Pulmonary and Critical Care Medicine, 321
Zhongshan Rd., The Affiliated Drum Tower Hospital,
Nanjing University Medical School, China.
Email: huil36@163.com
Received : Oct 26, 2021
Accepted : Dec 09, 2021
Published : Dec 16, 2021
Archived : www.jcimcr.org
Copyright : © Li H (2021).
Abstract
Acitretin is retinoic acid derivative that is approved for treatment of severe psoriasis. The side effect of retinoic therapy is all-trans retinoic acid syndrome. Here, we report a case of acitretin treatment associated acute lung injury in a psoriasis patient. Exhibited as a diffuse lung parenchymal injury, the differential diagnosis of infection, heart failure, and alveolar hemorrhage should be ruled out. A definite medication history is prerequisite for diagnosis of the disease. After correct diagnosis, the patient responded well to corticosteroid and the symptom and radiological changes of acitretin-induced acute lung injury alleviated soon.
Citation: Cao M, Li H, Xiao Y, Cai H. Responding well to corticosteroid in an acitretin treatment-related acute lung injury patient with psoriasis. J Clin Images Med Case Rep. 2021; 2(6): 1484.
Introduction
Differentiation Syndrome (DS) or Retinoid Acid Syndrome (RAS) is a common complication that is described in patients with Acute Promyelocytic Leukemia (APL) who are treated with All-Trans Retinoic Acid (ATRA) [1]. Within 2 to 21 days of ATRA treatment initiation, patients may develop RAS manifested as fever, pulmonary infiltrates, hypoxemia, respiratory distress, weight gain, pleural and pericardial effusions; hypotension, and multi-organ failure [2]. Acitretin is retinoic acid derivative that is approved for treatment of severe psoriasis [3]. Here, we report a case of acitretin treatment associated acute lung injury, which responded well to corticosteroid in a psoriasis patient.
Case presentation
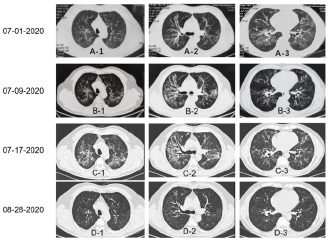
A 56-year-old man, having psoriasis over 10 years, presented to our division with dyspnea, non-persistent fever, and productive cough within a period of 20 days, but denied chest pain, palpitations, syncope or body edema. Before he came to our facility, he was admitted to hospital A because of complaining productive cough, dyspnea, and fever for 5 days (Tmax 38.4o C). Computed tomography scanning showed diffused ground glass opacity, and local consolidation (Figure 1, A-1~A-3). Brain Natriuretic Peptide (BNP) was slightly elevated at 180 pg/ml. Then, pneumonia was suspected and bronchoscope was performed in that hospital for withdrawing Bronchoalveolar Lavage Fluid (BALF) for microorganism screening including Next Generation Sequencing Technology (NGS). There were no positive findings for bacteria, fungi, virus, and parasites. However, the patient was still empirically administered with imipenem and Linezolid. Several days later, voriconazole was also prescribed for recurrence of fever. After one-week treatment, the patient still had symptoms of dyspnea and productive cough. CT scan was rerun and the results showed as (Figure 1, B-1~B-3), ground glass opacity was getting thicken and traction bronchiectasis was obviously shown. He was then transferred to our hospital.
Laboratory testing showed a white blood cell count of 13.1 X 103 /μL, lymphocytes of 1100/μL, hemoglobin levels of 13.9 g/ dL, platelet count of 339 X 103 /μL, and C-reactive protein levels of 0.6 mg/dL. Aminotransferase levels of 17.8 U/I, alanine transaminase levels of 21.3 U/I, total bilirubin levels of 0.54 mg/dL, total protein levels of 6.48 g/dL, albumin levels of 4.0 g/dL, blood urea nitrogen levels of 4.6 mmol/L, and creatinine levels of 49 μmol/L was detected. The Erythrocyte Sedimentation Rate (ESR) increased to 40 mm/h. Carcinoembryonic Antigen (CEA) was normal as 4.57 ng/ml, Neuron-Specific Enolase (NSE) increased to 33.40 ng/ml, and CYFRA21-1 was elevated as 19.80 ng/ml.Microbiological study showed a negative culture result for bacteria. He was also negative for T-SPOT, β-D glucan and galactomannan antigen test. The serum antibodies for virus including Influenza A, influenza B, COVID-19, cytomegalovirus, EB virus were also negative. He was hypoxemic with an oxygen saturation of 88% on room air at rest. Blood-gas testing showed a decreased PaO2 at 53 mmHg (room temperature, breath at quiescent status).
High resolution CT scanning revealed bilateral central distributed, diffused ground glass opacity and consolidation. More diffused traction bronchiectasis was also seen in the CT scan (Figure 1, C1~C3). Pulmonary function testing revealed a normal ventilation function, moderate decreased diffuse capacity (DLCO 3.66 mmol/min/kPa; 52.5% predicted value). Echocardiography was performed to rule out heart failure. The results showed no significant valvular disease and signs of heart failure (ejection fraction value of 63.0%, left ventricular end diastolic/ systolic diameter of 49/38 mm, inferior vena cava diameter during inspiration of 18 mm, and normal right ventricular internal diameter of 36 mmHg).
Based on above parameters, infection and heart failure were ruled out for the abnormality on CT scan and respiratory failure. Because he had psoriasis, we then inquired the therapeutic detail for the treatment. He was received about 10 years external acitretin treatment, and two months ago, he was advised to receive oral acitretin because of refractory skin lesion. After receiving 20-30 mg/day acitretin for two months, he got fatigue, dry mouth, and subsequently fever, dry cough, and progressive dyspnea. He then went to hospital A. Finally, the cause for his respiratory failure was diagnosed as acitretin-associated lung injury. Acitretin was strictly stopped, even for the external use. 40 mg methylprednisolone was intravenous administered to the patient for 5 days. And then he discharged with 40 mg oral prednisone for 15 days and 30 mg oral prednisone for another 15 days. A month later, he revisited to our outpatient setting, and CT scan was re-performed. As shown in (Figure 1, D-1~D-3), ground glass opacity and consolidation diminished and vanished, and traction bronchiectasis was largely alleviated.
Discussion
The most important complication of all trans retinoid acidbased therapy is the Retinoic Acid Syndrome (RAS), a cardiorespiratory syndrome manifested asdyspnea, pulmonary infiltrates, pleural or pericardial effusions, episodic hypotension, and multi-organ failure [1]. As previous report, ATRA administration may induce RAS in an incidence of approximately 25% cases [4]. Hence, RAS was broadly concerned during the treatment of acute promyelocytic leukemia using ATRA. Due to the awareness, the mortality rate of the patients with this syndrome has declined from approximately 30% to 2~10% [5]. Diagnosis of the RAS is under the premise of using ATRA, occurrence one of the following clinical findings in the absence of alternative explanations: Fever, weight gain, respiratory distress, pulmonary infiltrates, pleural or pericardial effusions, hypotension and renal failure [6]. In our case, patient was diagnosed with RAS under the occurrence of dyspnea, laboratory confirmed respiratory failure, and pulmonary infiltrates after two months acitretin treatment. The causative for misdiagnosis of pneumonia in hospital A was due to absence of inquiring the treatment detail for his psoriasis. After discontinuing acitretin and taking corticosteroid therapy, the symptoms relieved and CT image alleviated promptly in this patient.
Acitretin is a derivative of retinoic acid, it is rarely associated with RAS as indicated by previous report. Limited case reports showed all the acitretin-associated RAS appeared in psoriasis patient [3,7,8]. As reported previously, the doses of acitretin used in those patients were at range of 35~50mg daily [3]. Here the patient in our report received only 20~30mg daily oral administration for two months and then RAS occurred. Although limited data regarding the dosage of acitretin for inducing RAS occurrence, it may still have variance dependent on personal sensitivity. Furthermore, this patient had used external acitretin for 10 years, it is unknown whether it has accumulating and/or synergistic effect of external and oral acitretin. The time course of onset of RAS was about two months in this patient which was longer than previous reports. Dose accumulation and gradually increased dosage of acitretin could be probably accounted for the longer onset time course.
In advantage of acitretin stop and corticosteroid usage, this patient recovered rapidly from respiratory failure to normal activity within a month. Intriguingly, traction bronchiectasis was almost disappeared after a month’s glucocorticoids administration. Traction bronchiectasis is noted as dilatation of airwaywithin the lung areas. Pathologically, traction bronchiectasis is thoughtto be the result of contraction of lung tissue surrounding airway becauseof fibrosis, inflammation, and scarring [9]. The relatively early intervention of glucocorticoid might be effectively interrupt and reverse the fibrotic procedure.
Conclusion
In conclusion, we report here a rare case of acitretin-associated RAS and acitretin-related lung injury responding well to corticosteroid.
Declarations
Conflict of interest statement: The authors declare they have no conflict of interest.
Informed Consent: Informed consent was not obtained from parents because the person anonymously mentioned in this case report.
References
- Tallman MS, Andersen JW, Schiffer CA, Appelbaum FR, Feusner JH, et al. All-trans-retinoic acid in acute promyelocytic leukemia. N Engl J Med. 1997; 337: 1021-1028.
- Nicolls MR, Terada LS, Tuder RM, Prindiville SA, Schwarz MI. Diffuse alveolar hemorrhage with underlying pulmonary capillaritis in the retinoic acid syndrome. Am J Respir Crit Care Med. 1998; 158: 1302-1305.
- Vos LE, Vermeer MH, Pavel S. Acitretin induces capillary leak syndrome in a patient with pustular psoriasis. J Am Acad Dermatol. 2007; 56: 339-342.
- Tallman MS, Andersen JW, Schiffer CA, Appelbaum FR, Feusner JH, et al. Clinical description of 44 patients with acute promyelocytic leukemia who developed the retinoic acid syndrome. Blood. 2000; 95: 90-95.
- Montesinos P, Bergua JM, Vellenga E, Rayón C, Parody R, et al. Differentiation syndrome in patients with acute promyelocytic leukemia treated with all-trans retinoic acid and anthracycline chemotherapy: Characteristics, outcome, and prognostic factors. Blood. 2009; 113: 775-783.
- Larson RS, Tallman MS. Retinoic acid syndrome: manifestations, pathogenesis, and treatment. Best Pract Res Clin Haematol. 2003; 16: 453-461.
- Liu D, Cao F, Yan X, Chen X, Chen Y, Tu Y, Furue M. Retinoic acid syndrome in a patient with psoriasis. Eur J Dermatol. 2009; 19: 632-634.
- Gu W, Zhao G, Shi F. Acitretin-induced retinoic acid syndrome. J Am Acad Dermatol. 2011; 65: e148-149.
- Hida T, Nishino M, Hino T, Lu J, Putman RK, et al. Traction Bronchiectasis/Bronchiolectasis is Associated with Interstitial Lung Abnormality Mortality. Eur J Radiol. 2020; 129: 109073.