Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Therapeutic dilemma: When to start anticoagulation for cardiac thrombi in acute ischemic stroke? Lessons from cases
Rajish Sanjit Kumar Shil1*; Mohamed Salah Gamea2; Noor Ullah Hussaini Mohammed3; Renganathan Radhakrishnan4
1 Department of Internal Medicine, Al Ain Hospital, Al Ain, United Arab Emirates.
2 Specialist Neurologist, Department of Neurology, Madinat Zayed Hospital, Abu Dhabi, United Arab Emirates.
3 Consultant Cardiologist, Department of Cardiology, Madinat Zayed Hospital, Abu Dhabi, United Arab Emirates.
4 Consultant Neurologist, Department of Neurology, Madinat Zayed Hospital, Al Ain Hospital, Abu Dhabi, United Arab Emirates.
*Corresponding Author: Rajish Sanjit Kumar Shil
Department of Internal Medicine, Al Ain Hospital, Al
Ain, United Arab Emirates.
Email: rajishrsk@gmail.com
Received : Nov 11, 2021
Accepted : Dec 14, 2021
Published : Dec 21, 2021
Archived : www.jcimcr.org
Copyright : © Shil RSK (2021).
Abstract
Ischemic stroke has been one of the major reason for morbidity and mortality all over the world. Anticoagulation treatment in patients with cerebral ischemic events caused by cardiac emboli is no longer a matter of debate. The problem is when to start. Because of a recurrence rate of 15 to 20% in the first few weeks after the initial event, direct anticoagulation by means of intravenous heparin followed by oral anticoagulants seems to be indicated. We are presenting 2 cases of patients who suffered cardio-embolic stroke due to cardiac thrombosis and had a therapeutic dilemma on when to start the anticoagulation treatment. Early anticoagulation treatment was associated with good clinical and neurological outcome.
Keywords: anticoagulation; stroke; cardiac thrombus; cardio-embolic stroke; hemorrhagic stroke; warfarin.
Abbreviations: MI: Myocardial Infarction; CT: Computed Tomography; MRI: Magnetic Resonance Imaging; NIHSS: National Institute of Health Stroke Scale; ADC: Apparent Diffusion Coefficient; INR: International Normalized Ratio; MV: Mitral Valve; MCA: Middle Cerebral Artery; OAC: Oral Anticoagulation; ECG: Electrocardiography; DWI: Diffusion Weighted Imaging; FLAIR: Fluid Attenuated Inversion Recovery; SWI: Susceptibility Weighted Imaging.
Citation: Shil RSK, Gamea MS, Mohammed NUH, Radhakrishnan R. Therapeutic dilemma: When to start anticoagulation for cardiac thrombi in acute ischemic stroke? Lessons from cases. J Clin Images Med Case Rep. 2021; 2(6): 1497.
Introduction
The cardiac conditions most commonly associated with cerebral embolism are rheumatic heart disease, atherosclerotic heart disease (myocardial infarction and atrial arrhythmias) and other kinds of non-valvular atrial fibrillation. Of patients with a myocardial infarction, 5-12% will have a clinically apparent systemic embolus, and one-third to one-half have a recurrence, usually early. Anticoagulation diminishes the incidence of emboli in myocardial Infarction to 25% of the natural Incidence. However, there is no clear-cut guideline to address, when to start the treatment with anticoagulation for patients with cardio-embolic stroke [1]. We are presenting two cases of patients who suffered from cardio-embolic stroke and early initiation of anticoagulation treatment resulted in a good clinical and neurological outcome. It also reduces the risk of recurrent thrombo-embolism as the risk of embolism is highest in the first two weeks of stroke.
Case description
Case 1
A 53 years old, man, known to have past medical history of ischemic heart disease, hypertension, dyslipidemia, heart failure with reduced ejection fraction, dilated cardiomyopathy, recent ST segment elevation myocardial infarction, status post angioplasty, presented to emergency department of tertiary care stroke center of Al Ain city with acute onset left sided weakness on January 2019. On neurological examination, his NIHSS score was 9 along with left hemiplegia and right sided upper motor neuron facial palsy. Since he arrived within the window period, he was treated with intravenous thrombolysis with Alteplase. Following thrombolysis, his NIHSS score improved to 2 within 24 hours clinical monitoring. Treatment as per the stroke protocol was continued in the form of physiotherapy, speech therapy and occupational therapy along with medications.
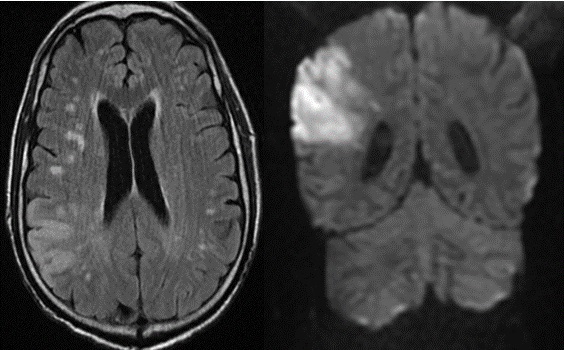
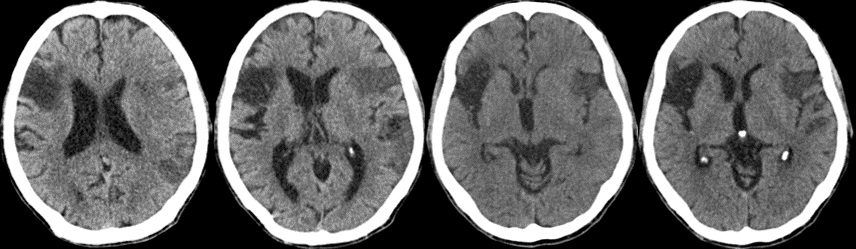
On arrival, his computed tomography scan of brain showed wedge shaped infarction on the right parietal region, with no evidence of hemorrhagic transformation. Following this, MRI brain revealed diffusion weighted image wedge shape high signal intensity seen involving the right frontal, temporal and parietal regions grey and white matter with corresponding ADC map low signal intensity changes; these findings are consistent with acute infarction of the right middle cerebral artery. It also revealed bilateral periventricular white matter hyper-intense signal changes in T2 and FLAIR images, which are suggestive of small vessel disease (Figure 1).
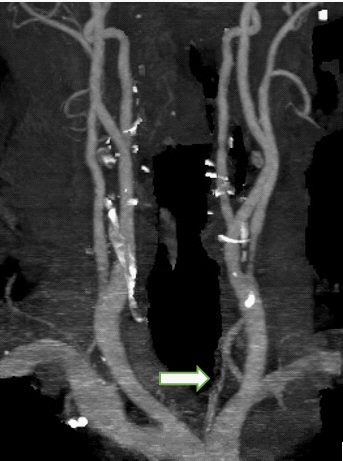
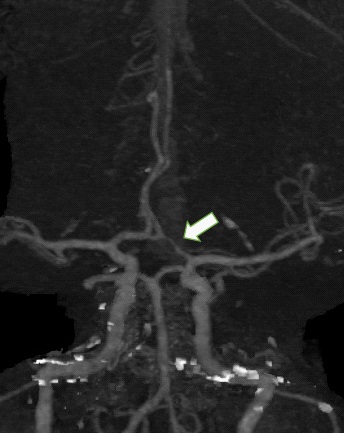
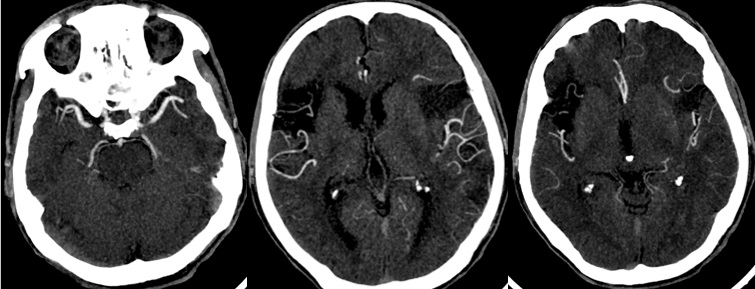
As a workup of stroke, CT Angiogram of the head and neck was performed. Neck angiogram revealed both common carotid arteries and internal carotid arteries of normal course and caliber with no significant stenosis. However, the origin and proximal segment of the left external carotid artery showed about 2 cm long significant stenosis, estimated up to 55% (Figure 2). Head angiogram also revealed a suspected small focal narrowing at left posterior communicating artery at the P2 -P3 junction (Figure 3). Otherwise major intracranial arterial system were found to be patent withno significant stenosis or aneurysmal dilatation.
On echocardiography, it was revealed that he had dilated left ventricle with reduced ejection fraction of 15%, thin akinetic apex, hypokinetic anterior-septal and inferior-septal walls.
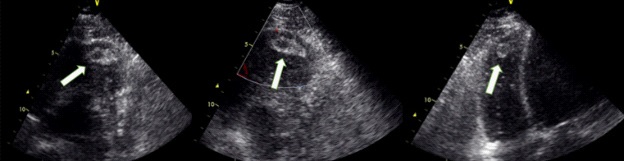
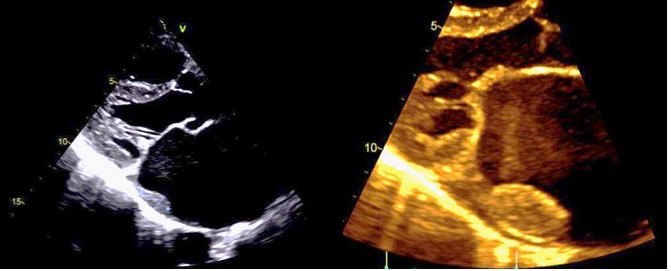
There was also multiple echogenic masses seen in the right ventricle, measuring around 5 X 6 mm and 4 X 6 mm in diameter, which represented most likely as thrombi (Figure 4). He also had moderate mitral regurgitation and left ventricular diastolic dysfunction grade 3.
In view of the fact that the patient had a recent ST elevation MI and underwent angioplasty to two vessels (right coronary artery and left anterior descending artery), patient was already on dual antiplatelet agents with aspirin and clopidogrel. So given his current clinical findings and radiological investigations, he was a candidate for anticoagulation as he had a cardio-embolic stroke. After discussion with cardiology and hematology, he was started on therapeutic low molecular weight heparin from day 3 of his presentation along with the dual antiplatelet agents. Later on, he was started on warfarin from day 7, with target INR of 2.0-2.5. It was decided by the multidisciplinary team to treat with dual antiplatelet agents and warfarin for 1 month, followed by long-term warfarin and clopidogrel, with close monitoring of INR and any hemorrhagic complications. He improved with our treatment strategy and was discharged from the hospital with modified Rankin score of 1. He continued to follow up in the clinic on monthly basis initially, followed by three monthly basis and he showed a good clinical outcome and did not suffer from any side effects of bleeding. He continued to take warfarin and clopidogrel after the first month and repeat echocardiography after six months showed resolution of the cardiac thrombi.
Case 2
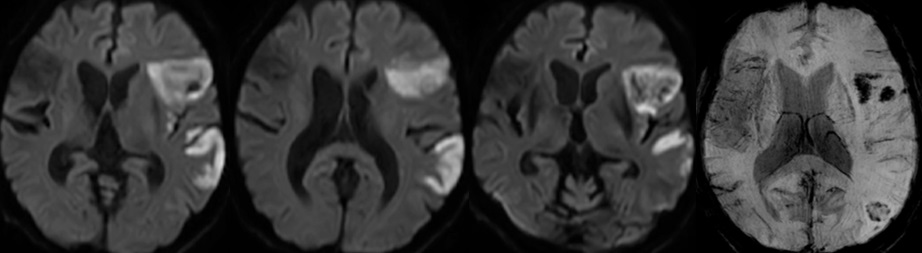
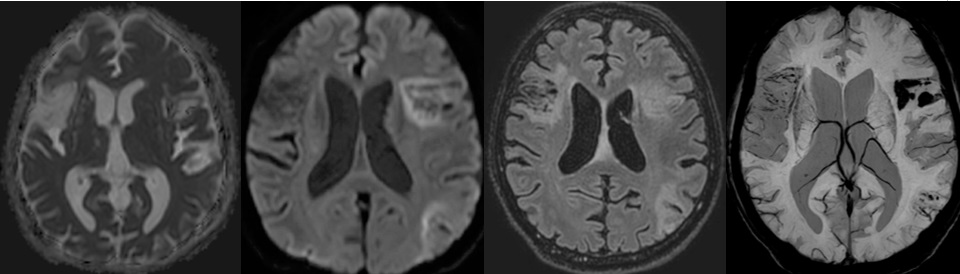
A 65 years old, male, not known to have past medical history, was brought in to tertiary care center in Abu Dhabi city, with altered mental status and difficulty in speech by his neighbor, with unknown time of onset on January 2021. On neurological examination, he had evidence of severe aphasia with left facial twitching. Motor and sensory examination of the limbs were within normal. NIHSS score was 1 and was hemodynamically stable. He had a normal ECG and troponin levels. Initial CT Brain showed signs of acute left Middle Cerebral Artery (MCA) infarction with hypodensity on the left fronto-parietal lobes (Figure 5), along with evidence of old right MCA infarct. MRI Brain showed acute left MCA infarct with hemorrhagic transformation along with findings of old right MCA hemorrhagic infarct (Figure 6). CT angiogram head and neck showed bilateral middle cerebral artery opacification with contrast, along-with some attenuation of sub-segmental branches of M4 segment in the superior trunks bilaterally and the inferior trunk on the left side (Figure 7).
The patient was admitted to stroke unit, started on Aspirin and prophylactic enoxaparin for venous thrombo-embolism. Four days post admission, he developed generalized tonic - clonic seizure for few minutes, which was self-limiting. He was started on Levetiracetam 500 mg twice daily. Carotid Doppler ultrasound showed mildly stenotic left common carotid artery, along with bilateral minimal intimal thickening of common carotid arteries (left > right). Echocardiography of heart showed dilated left atrium with thrombotic clots in the atrium and the atrial appendage extending to left atrium and Mitral Valve (MV) stenosis. These findings were confirmed with trans-esophageal echocardiography (Figure 8) where MV peak gradient was 12 mmHg and mean gradient was 8 mmHg, MV Area 0.8 cm2 with moderate mitral regurgitation, which appeared to be rheumatic in origin. There was no regional wall motion abnormalities and he had good systolic function of 60%.
The patient was a candidate for anticoagulation treatment in view of cardiac thrombi and severe mitral stenosis to prevent further development of embolic stroke. However due to the presence of hemorrhage in the brain, there was a dilemma on starting of anticoagulation treatment. Following a MDT discussion in view of appropriate risk benefit ratio, patient was monitored in the intensive care unit, continued on aspirin and prophylactic enoxaparin for one week. Repeat CT brain did not show extension of hemorrhage, following which aspirin was stopped and warfarin + enoxaparin was introduced to achieve therapeutic INR 2-3. He was kept on 2.5 mg warfarin for maintaining the target INR. He continued speech rehabilitation treatment in the stroke unit, following which he had significant improvement in his speech and was discharged safely.
Repeat MRI brain after a month showed the infarcted left MCA area to be sub-acute in nature with no significant interval changes regarding hemorrhagic transformation of the previous study (Figure 9). Repeat ECHO showed presence of the left atrial clots but there was significant reduction in the size of the clots.
Discussion
Oral Anticoagulation (OAC) is the treatment of choice for secondary prevention after a cardio-embolic stroke [2,3]. Warfarin is the commonest OAC being used worldwide, although other OACs like acenocoumarol, phenprocoumon, or anisindione are frequently prescribed in many countries. The mechanisms of action of these medications are comparable, as they all inhibit the vitamin K-dependent post-translational carboxylation of glutamate residues on the N-terminal regions of coagulation factors VII, II, IX, and X via inhibiting the conversion of vitamin 2, 3 epoxide to reduced vitamin K [4]. Although the benefits of OAC are supported by a high degree of evidence for stroke prevention in cardio-embolic entities, such as atrial fibrillation [4], they carry certain limitations in the form of narrow therapeutic index, drugs and dietary interactions, along-with a significant risk of serious bleeding, including hemorrhagic stroke [5].
In the first case, given presence of recent large vessel infarction, early initiation of anticoagulation would have resulted in hemorrhagic complications, and hence it was decided to defer anticoagulation with warfarin after a week of presentation. There was no other anticoagulants approved for such cases apart from few case reports in the literature. However, given the fact that the patient had a recent STEMI with angioplasty to two vessels, it was mandatory to be on dual antiplatelet agents, and to add anticoagulation early on would have increased his risk of hemorrhagic complications. Therefore, he was initially started on subcutaneous low molecular weight heparin, which was later substituted to oral anticoagulation with warfarin after a week. In the second case, the patient had a hemorrhagic transformation of the ischemic infarct, along with cardiac thrombi and mitral valve stenosis. Also no anticoagulation was approved for such cases, if not warfarin, but the timing to start anticoagulation was a challenging decision. We started warfarin cautiously after a week, and it resulted in positive outcome with no further hemorrhagic complications later.
Hemorrhagic transformation is not uncommon and patients with cardioembolic infarcts are particularly prone to develop hemorrhagic transformation. Hemorrhagic transformation has been reported in up to 40% of patients with cerebral embolism on CT scans [6], and more than 60% on MRI scans [7]. Elderly patients with large sized infarcts are known to be associated with particularly high risks of hemorrhagic transformation [6- 8]. Following an acute cardioembolic stroke, which occurs due to left ventricular thrombi, the risks for recurrent early embolic event is usually high. Hence, in order to decide about starting anticoagulant treatment, it is prudent to balance the benefit of reduction in early recurrent embolic events against the risks of potentiating secondary brain or other organ hemorrhage. Cardioembolic strokes were found to have a higher propensity for secondary hemorrhagic transformation and, therefore, no consensus has been reached on the optimum strategyin this regard [9]. Font et al suggested that, however, based on the fact as the usual timing of secondary hemorrhagic transformation is usually between 12 hours and 4 days after stroke onset, it would be reasonable to begin warfarin as soon as the patient is declared medically and neurologically stable. This happens often on 2 to 3 days after stroke, and physicians can aim to achieve therapeutic anticoagulation by 7 to 10 days after the stroke onset. Some experts, however,tends to repeat a CT brain, before initiating warfarin and tends to delay warfarin therapy if hemorrhagic transformation is evident. Minor degrees of hemorrhagic transformation are frequently seen (particularly on MRI brain), and the clinical significance regarding initiation of warfarin at that point is unclear and controversial [9].
The risk of other major thromboembolic events like pulmonary embolism or recurrent ischemic stroke or transient ischemic attacks were weighed against the hemorrhagic complications for both of our patients, and hence was initiated with anticoagulation early on. As per the guidelines for any cardioembolic stroke, it is usually recommended to start on anticoagulation after 14 days of the cerebrovascular event, but in our patient, in view of the recent STEMI, poor ejection fraction, and major vessel infarction in first patient and multiple cardiac thrombus with severe mitral valve stenosis, we have started the treatment earlier with the benefit outweighing the risks of treatment modality.Our first patient continued to be on longterm warfarin and clopidogrel after the first month of triple therapy with aspirin; second patient continued only on single agent long-term warfarin treatment. Both treatment approach have resulted in a good clinical outcome, as we have closely followed up the patients in the anticoagulation and stroke clinic, where they did not suffer any morbidities from the treatment plan.
The frequency of embolic cerebral infarction varies from 20 to 50 percent in recent studies. The recurrence rate is high, especially during the first 3 weeks after the initial event and therefore anticoagulation treatment is indicated. A matter of debate has been when to start such treatment. In pathology studies, a high frequency of small petechial hemorrhages, mainly in gray matter, was observed in cases with cerebral embolic infarction. It is usually the fragmentation of the embolic clots with reperfusion through the distal affected vessels, which was hypothesized to be the responsible mechanism for such hemorrhagic infarction.This finding plus incidental clinical dataled to the fear of a bleeding tendency if anticoagulation treatment should be started immediately after cerebral embolic ischemia. Moreover, experimental studies showed an increase in hemorrhagic infarction and clinical deterioration in animals under anticoagulation treatment after reperfusion of temporarily occluded cerebral arteries. Therefore some authors advised to wait a period ranging from a few days to weeks before initiating anticoagulation treatment. Since the feared dangerous complication of direct anticoagulation treatment in embolic stroke has never been confirmed in clinical studies, others advised to start immediate anticoagulation treatment by means of intravenous heparin to prevent early recurrences [10]. We had similar approach in our patients and they had a good clinical outcome based on this approach.
Conclusion
In patients who develops stroke due to cardiac embolism, who also has recent history of ischemic heart disease, early treatment with dual antiplatelet and anticoagulation may be considered to prevent further thromboembolism after properly weighing benefits and risks of anticoagulation. Our patient has tolerated the treatment well with such approach and it can be prudent to apply this approach for similar patients in future and needs further studies to implement a treatment protocol for similar patients.
Declarations
Conflicts of interest: The authors declare no conflicts in interest for the mentioned case report.
Consent: Informed written consent was obtained from both the patients for reporting the case report.
Data availability: The radiological imaging and laboratory data were collected from electronic medical records and were used to support the findings of this study for publication of this case report.
Presentation: Case series was presented as “E-POSTER” in the 13th World Stroke Congress, held Virtually in Spain, October 2021 and have received acknowledgement and interest from global researchers.
References
- Easton J, Sherman D. Management of cerebral embolism of cardiac origin. Stroke. 1980; 11: pp.433-442.
- “Guidelines for management of ischaemic stroke and transient ischaemic attack 2008,” Cerebrovascular Diseases. 2008; vol. 25: pp. 457–507.
- A Cervera, S Amaro, V Obach, and A. Chamorro. “Prevention of ischemic stroke: Antithrombotic therapy in cardiac embolism,” Current Drug Targets. 2007; vol. 8, no. 7, pp. 824–831.
- D. S. Whitlon, J. A. Sadowski, and J. W. Suttie, “Mechanism of coumarin action: Significance of vitamin K epoxide reductase inhibition,” Biochemistry. 1978; vol. 17, no. 8, pp. 1371–1377.
- S. Schulman, R. J. Beyth, C. Kearon, and M. N. Levine, “Hemorrhagic complications of anticoagulant and thrombolytic treatment: American College of Chest Physicians evidence-based clinical practice guidelines (8th edition),” Chest. 2008; vol. 133, supplement 6, pp. 257S–298S.
- Okada Y, Yamaguchi T, Minematsu K, Miyashita T, Sawada T, Sadoshima S, et al. Hemorrhagic transformation in cerebral embolism. Stroke 1989; 20: 598-603.
- Hornig CR, Bauer T, Simon C, Trittmacher S, Dorndorf W. Hemorrhagic transformation in cardioembolic cerebral infarction. Stroke 1993; 24: 465-468.
- Hornig CR, Dorndorf W, Agnoli AL. Hemorrhagic cerebral infarction–a prospective study. Stroke.1986; 17: 179-185.
- M. À. Font, J. Krupinski, and A. Arboix, “Antithrombotic medication for cardioembolic stroke prevention,” Stroke Research and Treatment. 2011; vol. 2011, Article ID 607852, 23 pages.
- Lodder J, van der Lugt PJ. Evaluation of the risk of immediate anticoagulant treatment in patients with embolic stroke of cardiac origin. Stroke. 1983; 14: 42-46.