Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Pneumothorax and lung metastases cavitation during cabozantinib treatment for metastatic clear cell renal cell carcinoma: A clinical case with literature overview
Marco FM Cavallaro1*; Anna Di Giusto2; Chiara Bigal3; Valentina Orlando3; Alessandra Guglielmi3; Maria A Cova3
1 Department of Radiology, Maggiore Hospital, Trieste, Italy.
2 Department of Oncology, Maggiore Hospital, Trieste, Italy.
3 Department of Radiology, Cattinara Hospital, University of Trieste, Trieste, Italy.
*Corresponding Author: Marco FM Cavallaro
Department of Radiology, Trieste University Hospital,
34139 Trieste, Italy.
Email: mrc.cavallaro@virgilio.it
Received : Nov 08, 2021
Accepted : Dec 22, 2021
Published : Dec 29, 2021
Archived : www.jcimcr.org
Copyright : © Cavallaro MFM (2021).
Abstract
Cabozantinib is an oral multikinase inhibitor approved for the treatment of advanced renal cell carcinoma. Radiological response evaluation may be difficult because, like other multikinase inhibitors, cabozantinib causes vascularization reduction and, in the lung, it may be associated with metastasis cavitation instead of a pure size reduction. Moreover treatment may be complicated with pneumothorax development, a life threatening condition, that has to be clinically suspected and promptly recognized on imaging.
We present a clinical case of a spontaneous pneumothorax in a patient with metastatic clear cell renal carcinoma treated with cabozantinib.
Keywords: cabozantinib; lung metasteses; pneumothorax; computed tomography
Citation: Cavallaro MFM, Di Giusto A, Bigal C, Orlando V, Guglielmi A, et al. Pneumothorax and lung metastases cavitation during cabozantinib treatment for metastatic clear cell renal cell carcinoma: A clinical case with literature overview. J Clin Images Med Case Rep. 2021; 2(6): 1528.
Introduction
Renal Cell Carcinoma (RCC) is the most common renal malignancy, accounting for 3.7% of all new cancers, and Clear Cell RCC (ccRCC) represents the more frequent subtype (75%). Localized disease is treated with partial or radical nephrectomy, heat or cold ablation, or active surveillance, but approximately 20% of patients have Metastatic RCC (mRCC) at diagnosis and require systemic treatment. Target therapies represent a well established option for the treatment of mRCC, and cabozantinb, a multikinase inhibitors of VEGFR2, MET, RET and AXL, has been approved in Europe both for initial therapy in intermediate and poor risk groups, and for second or third-line therapy in all risk groups [1]. The introduction of moleculartargeted therapies, including cabozantinib, poses challenges for the radiological assessment of treatment response and, althought Response Evaluation Criteria In Solid Tumors (RECIST) are currently used for the evaluation of therapeutic response to antineoplastic agents, these criteria have shown several limitations. Mainly, RECIST are based only on dimensional changes of tumoral lesions, while tyrosine kinase inhibitors induce first changes in attenuation and necrosis rather than a pure size reduction; moreover in lungs, metastases necrosis could appear as air-filled cavities, and these changes may be misinterpreted on radiological evaluation. Furthermore, in case of a rapid cavitation of lung metastasis, pneumothorax may develop and the radiological assessment could be more complex and doubtful.
We present a clinical case of mRCC treated with cabozantinib, with cavitation of lung metastases and pneumothorax development as hallmarks of drug response.
Clinical case
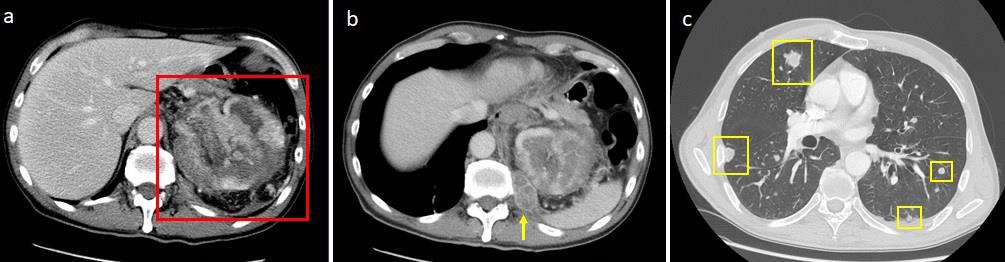
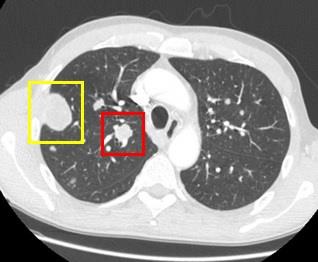
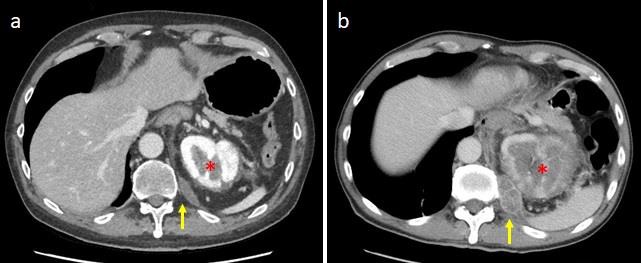
A 57 year-old man underwent a thoracoabdominal Computed Tomography (CT) for persistent left flank pain, anemia and haematuria. CT demonstrated a very large left renal mass with bilateral lung metastasis and soft tissue lesions (Figure 1); lung lesions were distributed both in the central and subpleural regions (Figure 2). Pathological diagnosis of clear cell RCC was obtained with a percutaneous renal biopsy, and so the patient started a medical therapy with oral cabozantinib 60 mg daily.
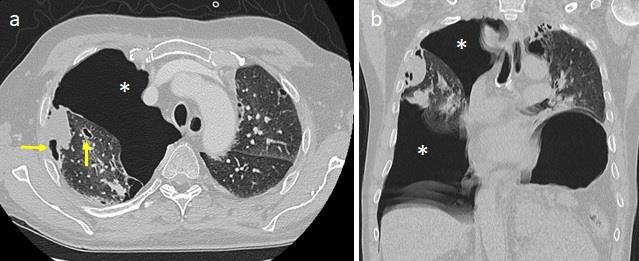
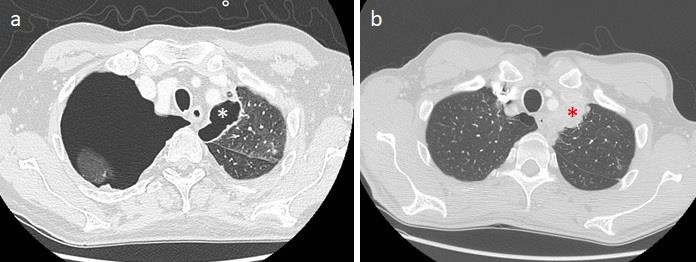
One month after starting treatment, he experienced acute chest pain and dyspnea, and so he was admitted to the Emergency Department where chest CT was performed, demonstrating a right pneumothorax and multiple cavitated lung lesions (Figure 3). Interestingly cavitated lesions were noted instead of originally solid lesions (Figure 4), and compared to the baseline CT both renal mass and the others already known soft tissue lesions were smaller and more hypodense due to vascularization reduction (Figure 5). Patient was therefore stabilized, treated with chest drainage placement, and finally discharged home after ten days.
Discussion
Cavitation of lung metastases is a known occurrence induced by cancer therapy, especially by antiangiogenic agents, but pneumothorax development is considered a quite rare complication. In a post-hoc analysis of the phase 3 CORRECT study, it is previously reported a 40.9% of incidence of lung metastases cavitation in 85 patients with Metastatic Colorectal Cancer (mCRC) treated with regorafenib [2]; no pulmonary events, such as dyspnea, pneumonia, hemoptysis or pneumothorax were reported. Lim et al. [3] published similar results in a prospective trial including 72 patients with mCRC treated with regorafenib; among 53 patients with lung metastasis, 17 (32.1%) developed cavitation at the first CT evaluation and this radiological sign was associated with better disease control rate. More recently Datar et al. [4] evaluated the incidence of cavitations in 83 patients with thyroid cancer metastases during treatment with antiangiogenic TKIs; authors reported a 12% of cavitations and described two cases of pneumothorax. Marom et al. [5] reported that 17 of 124 (14%) patients who received antiangiogenic therapy for lung cancer developed tumor cavitation, but they did not describe pneumothorax development. In another study by Intariano et al. [6] it was demonstrated a close association between pneumothorax and cavitation of pulmonary nodule, studying 33 patients with lung metastases treated with bevacizumab-sorafenib and low-dose cyclophosphamide, but authors analyzed a pediatric population. In another two studies considering patients with soft tissue sarcoma and treated with pazopanib, pneumothorax was reported in 3.3- 14% of cases [7,8]; it was frequently observed in presence of pleural or subpleural lung metastases, but was independent of tumor cavitation and therapeutic response. In two cases of pneumothorax related respectively to axitinib and sunitinib for mRCC [9,10], authors described the presence of cavitated lung metastases. Particularly, the patient treated with sunitinib developed bilateral spontaneous pneumothorax 3 weeks after start treatment, and CT revealed the presence of cavitation of previous bilateral lung nodules; authors hypothesized that pneumothorax could be induced by a bronchopleural fistula formation from one of the cavitated lesions. The axitinib-treated patient developed a left recurrent pneumothorax after one month; in this case the pathological analysis of a large subpleural cavitated lesion reported a RCC lung metastasis with extensive necrosis. In another case report of Iacovelli et al. [11], two cabozantinib-related pneumothorax were described in patients with mRCC; in both cases cavitated lesions were located in the subpleural space, so authors hypothesized that tumor necrosis could be the cause.
From these studies emerges that cavitation of lung nodules is a possible effect of multi tyrosine kinase inhibitors treatment, and it must be correctly evaluated on imaging even if it is not included in the evaluation of response with conventional RECIST criteria. In our clinical case abdominal CT findings were consistent with a good response to chemotherapy according to RECIST, while lung lesions showed a partial response, with a 30% decrease in size; however all metastases were air filled due to necrosis suggesting practically a very good response. In our case pneumothorax developed one month after starting treatment in patient without other risk factors, and lung lesions were distributed in both subpleural and central locations. Considering the very good response of primary renal mass and other abdominal lesions, we hypothesize pneumothorax as a consequence of a rapid tumour necrosis induced by cabozantinib, with fistula formation between the lung parenchyma and pleural space.
Conclusion
Lung metastases cavitation may be a consequence of cabozantinib treatment, like other anti TKI agents, and from the few available reported cases in this setting, it seems that cavity formation could be associated with therapy response. Moreover spontaneous pneumothorax could be a possible complication during cabozantinib treatment for mRCC, and so both Oncologists and Radiologists have to be aware of this eventuality.
References
- Astrid A M. van der Veldt et al. Targeted therapies in renal cell cancer: Recent developments in imaging. Targ Oncol. 2010; 5: 95–112.
- Ricotta R, Verrioli A, Ghezzi S et al. Radiological imaging markers predicting clinical outcome in patients with metastatic colorectal carcinoma treated with regorafenib: Post hoc analysis of the CORRECT phase III trial (RadioCORRECT study). ESMO Open. 2017; 1: 1–10.
- Lim Y, Han SW, Yoon JH et al. Clinical Implication of AntiAngiogenic Effect of Regorafenib in Metastatic Colorectal Cancer. PLoS ONE. 10: e0145004.
- Datar S, Cabanillas M, Dadu R et al. Pulmonary cavitation in patients with thyroid cancer receiving antiangiogenic agents. BMC Cancer. 2020; 20: 1181.
- Marom EM, Martinez CH, Truong MT et al. Tumor Cavitation During Therapy with Antiangiogenesis Agents in Patients with Lung Cancer. J Thorac Oncol. 2008; 3: 351–357.
- Interiano RB, McCarville MB, Wu J et al. Pneumothorax as a complication of combination antiangiogenic therapy in children and young adults with refractory/recurrent solid tumors. Send J Pediatr Surg. 2015; 50: 1484–1489
- Van der Graaf WT, Blay JY, Chawla SP et al. Pazopanib for metastatic soft-tissue sarcoma (PALETTE): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2012; 379: 1879– 1886.
- Verschoor AJ, Gelderblom H. Pneumothorax as adverse event in patients with lung metastases of soft tissue sarcoma treated with pazopanib: A single reference centre case series. Clin Sarcoma Res. 2014; 4: 14.
- Katta A, Fesler MJ, Tan A, et al. Spontaneous bilateral pneumothorax in metastatic renal cell carcinoma on sunitinib therapy. Cancer Chemother Pharmacol. 2010; 66: 409–412.
- Socola F, Loaiza-Bonilla A, Benedetto P. Axitinib Induced Recurrent Pneumothorax following Near-Complete Response of Renal Cell Carcinoma Lung Metastasis: An Unexpected Complication. Case Rep Oncol Med. 2012.
- Iacovelli R, Ciccarese C, Mosillo C et al. Cabozantinib-related pneumothorax in rapidly responding patients with renal cell carcinoma. Lancet Oncol. 2019; 20: e124.