Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Inflammatory cytokines and cell activation markers in HIV and HIV/TB co-infected patients attending AMPATH clinic at MTRH-Eldoret, Kenya
Rose Undisa1*; Silas O Awuor2; AK Chemtai1; LO Diero1
1 Moi university, School of medicine, Eldoret, Kenya.
2 Ministry of Health, Masogo Sub county hospital, Kisumu County, 12-40122, Kisumu, Kenya.
*Corresponding Author: Rose Undisa
Moi university, School of medicine, Eldoret,
Kenya.
Email: undisaros@gmail.com
Received : Nov 17, 2021
Accepted : Jan 06, 2022
Published : Jan 13, 2022
Archived : www.jcimcr.org
Copyright : © Undisa R (2022).
Abstract
Background: Infection with HIV and TB leads to inflammation. Markers of inflammation include TNF-α, IFN-γ, CD38 and HLA-DR. In HIV infection these markers create an environment which favours reactivation of latent TB to TB disease in patients on HAART. The markers TNF-α, IFN-γ, CD38, HLA-DR and CD4 cell count can be used to predict possible reactivation of latent TB to disease in HIV patients on HAART. The level of these inflammatory markers in HIV and HIV/TB co-infection is unknown. The purpose of this study was to investigate inflammatory markers TNF-α, IFN-γ, CD38, and HLA-DR levels in HIV and HIV/TB coinfected patients attending AMPATH clinic at MTRH-Eldoret.
Objective: To determine inflammatory cytokines and cell activation markers in HIV and HIV/TB co-infected patients attending AMPATH clinic at MTRH-Eldoret.
Methods: This was a cross sectional comparative study of 168 patients comprising of 84 HIV/TB co-infected and 84 HIV only patients who had been on HAART for more than 1 year. Clinical and demographic information was obtained from the patients hospital files and interview guide. Blood samples from each patient were collected into EDTA tubes and plasma separated within 2 hours of collection at 3,000 r.p.m for 3 minutes then kept at -800 C until analysis. Activation markers CD38, HLA DR and CD4 count were analysed by flow Cytometry (BD FAC’S calibur® ) on whole blood samples. Analysis of the inflammatory cytokines (TNF-α and IFN-γ) were done on the plasma samples by ELISA (Genway’s® ). The levels of inflammatory markers in the two groups were compared. Data was analysed using STATA version 13. P-value ≤ 0.05 was considered significant.
Findings: The mean age for both HIV/TB and HIV patients was 42 years. The median (IQR) levels for TNF-α (pg/ml) was 7.26 (6.76- 8.12), IFN-γ (pg/ml) 26.3 (25.18- 27.22), CD38(%) 4.36 (2.06-6.31), HLA-DR (%) 92.44 (90.52-95.51) and CD4 (cells/mm3 ) 231 (117-350) in HIV/TB co-infected patients, while in HIV patients the levels were TNF-α 10.72 (8.12-12.13), IFN-γ 25.40 (23.59-27.67), CD38 5.94 (4.44-7.41), HLA-DR 93.33 (90.24-96.56) and CD4 383 (318-543). HIV only patients had higher levels of TNF-α (p=<0.001), CD38% (p=0.0002) and CD4 (p=<0.001) compared to those of TB/HIV co-infected patients. No significant difference was observed in the levels of IFN-γ and HLA-DR (p=0.1559) and (p=0.1754) respectively.
Conclusion: Higher levels of inflammatory markers were found in HIV only patients compared to HIV/TB co-infected patients. This high level of inflammation leads to TB-reactivation.
Keywords: inflammatory cytokines; cell activation marker; HIV and HIV/TB co-infected.
Citation: Undisa R, Awuor SO, Chemtai AK, Diero LO. Inflammatory cytokines and cell activation marker in HIV and HIV/TB co-infected patients attending AMPATH clinic at MTRH Eldoret, Kenya. J Clin Images Med Case Rep. 2022; 3(1): 1559.
Introduction
In HIV/TB co-infection, the immunologic environment is characterised by cytokine shifts that has been shown to increase immune activation, viral replication, and problems associated with T- cell function such as autoimmunity [1]. Studies have shown that, immune response to MTB involves IFN-γ as a Th1 cytokine which plays the role of containment of mycobacterium within granulomas [2]. IFN- γ is induced early in infection, and is protective against many pathogens including HIV and TB infections [3]. IFN-γ also plays the function of immune regulation and pro inflammatory antiviral activity which has made it be able to be used as an alternative biomarker to assess immune capabilities and antiviral response in HIV patients [4]. IFN- γ and TNF-α levels were found to be high in HIV/TB and TB patients due to increased immune activation [5]. According to Barlett et al. 2013, HIV positive and negative TB patients displayed similar plasma cytokine pattern and, anti-TB treatment improved the Th1 cytokine levels but did not restore the immune response in HIV positive individuals. TNF-α release from infected cells was stimulated by both HIV and MTB, where the stimulated TNF-α enhanced HIV replication, and inhibited MTB growth [6]. It was shown that increase in HIV viral load led to development of active TB, in patients who were not on antiretroviral therapy [7]. This signifying that high viral load has negative effects on the immune system by weakening the natural immunity mechanisms, leading to development of opportunistic infections such as TB. TB associated Immune Reconstitution Inflammatory Syndrome (TB-IRIS) developed in many patients on Highly Active Antiretroviral Therapy (HAART), soon after being initiated on HAART [8]. IRIS was thought to be caused by rapidly recovering CD4 T cell count when the patient was on HAART, thus the rapid recovery being as a result of immune recovery, in which there was a high level of Th1 response to a latent opportunistic infection [9,10]. Immunological pattern of patients on HAART individuals was found to be similar to non-infected individuals, showing that when the patient was on HAART, immune recovery was returned to about normal [11]. Biomarkers of immune activation and inflammation in patients on HAART moved towards levels found in HIV negative patients [8]. Thus, HIV patients not on HAART were in an inflammatory state, which was shown by varying levels of Th1 cytokines and activation markers [11]. In patients on HAART there was reduced plasma levels of TNF-α. implying that there was immune recovery and reduced viral replication [12]. This however has been shown to reverse after some period of treatment with HAART, implying that there could be immune deterioration shown by increased viral replication as a result of high TNF-α levels [13]. Protection against Mycobacterium Tuberculosis infection required activation of Th1 cells and subsequent production of TNF-α and IFN-γ [14]. Studies have shown that, for TB to remain in a latent state, there should be higher levels of Th1 cytokines, to be able to contain the infection within granulomas [15]. However, high levels of TNF-α in patients on HAART has been shown to indicate treatment failure, and reflect immune activation [14,16]. Increased TNF-α levels were linked to increased viral replication, high viral load, acceleration to disease, and low CD4 count [17]. Progression of HIV infection was related to increases in levels of TNF-α as the high levels propagated viral replication [8,18]. TNF-α was elevated in HIV patients before HAART, and the levels were seen high in patients failing HAART, indicating treatment failure [19]. Thus, the immunological response involving high TNF-α levels may signify a failing immune system due to increase in viral load, which then weakens the immune system leading to exacerbation of latent TB to TB disease.
Key Th1 cytokines (TNF-α and IFN- γ ) in HIV patients can be investigated as a milestone in the quest to finding a solution in retaining the TB infection in a latent state, which would increase productivity and reduce mortality.
Methodology
Study area
The study was carried out at AMPATH clinic at MTRH Eldoret, Kenya. The clinic provides health services for HIV infected patients from the western region, covering Kitale, Kapenguria, Teso, Mosoriot, Turbo, Burnt-forest, Webuye, Naitiri, Chulaimbo and Amukura. MTRH is used as the teaching hospital for Moi university-school of medicine, and other institutions. AMPATH also works to address the epidemic of TB, with a keen emphasis on HIV co-infection. AMPATH has over 60 urban and rural clinic sites, running programmes such as research, social health, primary care and chronic diseases, maternal and child health, communicable diseases, agriculture and nutrition, and children’s services (Wikipedia, 2016). The United States Agency for International Development-Academic Model Providing Access to Health care (USAID-AMPATH) clinic is located in western Kenya, Eldoret headquarter [20].
Study design
The study was a cross sectional comparative study of representative Th1 cytokine (IFN-γ and TNF-α) levels and cell activation markers in HIV/TB co-infected and HIV only patients attending AMPATH clinic at MTRH-Eldoret.
Study population
The study population consisted of HIV/TB co-infected and HIV only patients attending AMPATH clinic at MTRH-Eldoret. A total of 168 patients were enrolled, of which 84 were HIV/TB co-infected and 84 were HIV positive patients on HAART. They both met inclusion and exclusion criteria.
Sampling procedure
Sampling was done consecutively, based on patient’s hospital number. All patients HIV and HIV/TB status were established first, and whether they were on HAART. The clinicians at the AMPATH and TB-clinics manage HIV positive patients, and those co-infected with TB. The clinical data was sought from the clinicians, as they saw the patients. This was filled in the interview guide and data sheet (appendix I), as the patients presented themselves for routine check-up and drugs collection. The two groups were matched on the basis of age, gender with the help of clinicians and records.
Data collection instruments and procedures
An interview guide was used to collect data. Blood samples from each patient were collected into EDTA tubes. Separation of plasma was done within 2 hours of sample collection at 3,000 r.p.m for 3 minutes and kept at -800C till the day of analysis. Activation markers CD38 and HLA DR and CD4 count were analysed by flow Cytometry on whole blood samples, while analysis for the representative Th1 cytokines (TNF-α and IFN-γ) was done on the plasma samples by ELISA (Genway’s®). The controls comprised of HIV positive patients on treatment for HIV infection only.
Determination of IFN- γ and TNF-α level
Genway’s ELISA kit was used. The ABC working solution and TMB colour developing agent was kept warm at 370 C for 30 minutes before use. 0.1 ml of samples and standards was added to each well and incubated at 370 C for 90 min. Biotinylated antibodies were added and incubation done at 370 C for 60 min. The plate was washed 3 times with 0.01 M TBS. ABC working solution was added and the plate incubated at 370 C for 30 min. It was washed 5 times with 0.01 M TBS. TMB colour developing agent was added and incubation of the plate done at 370 C in dark for 20-30 min. TMB stop solution was added, and reading was done at the O.D. absorbance at 450 nm in a micro plate reader within 30 min after adding the stop solution.
Data management and statistical analysis
Filled data forms were checked for completeness, forms that had gaps were taken back to the respective points for verification and re-filling. Data was entered in a spreadsheet. It was later exported to STATA V.13 for analysis. Since the subjects were matched on the basis of age and gender, these variables were not analysed. Normality tests were performed using the Shapiro wilks test. The measure of central tendency (Median (IQR) was used to summarize the data. Since the data was skewed, Mann Whitney U-test was used to compare the medians between the two groups (HIV/TB and HIV only). P-value less than 0.05 was considered significant. Data is presented in form of a Table 1 and box plots.
Table 1: Biographical data of the study population.
Age bracket (years) |
Males |
Females |
Total |
||||
HIV |
HIV/TB |
Total |
HIV |
HIV/TB |
Total |
||
18-27 |
5(2.98) |
7(4.17) |
12(7.14) |
6(3.63) |
6(3.66) |
12 (7.14) |
24(14.29) |
28-37 |
14(8.33) |
12(7.14) |
26(15.45) |
13(7.86) |
13(7.93) |
26 (15.45) |
52(30.95) |
38-47 |
15(8.93) |
16(9.52) |
31(18.45) |
16(9.67) |
15(9.15) |
31(18.45) |
62(36.90) |
48-57 |
8(4.77) |
7(4.17) |
15(8.94) |
8(4.83) |
7(4.25) |
15(9.08) |
30(17.86) |
TOTAL |
42(25) |
42(25) |
84(50) |
43(26) |
41(25) |
84 ( 50) |
168(100) |
Ethical considerations
The study protocol was approved by the Institutional Research and Ethics Committee (IREC) MTRH, Ref. No. RES/ STUD/1/2016 (appendix VII). Written informed consent was sought from each participant prior to sample collection (appendix II). Numbers were used on the data collection form. Hard copies were kept in a lockable cabinet while data in the computer was protected using password.
Results
A total of 168 patients comprising of 84 for HIV/TB (test group) and 84 for HIV only (control group) were analyzed from February to May 2016.
The age group range was between 22 and 55, a majority of the population being between ages of 38-47 with a total of 62 patients. The least age bracket was 18-27 years with a total of 24 patients.
Clinical data of the study population
All patients sampled had been on HAART for more than 1 year.
Main findings of this study indicated higher levels of TNF-α, CD38 and CD4 in HIV only patients and similar levels of IFN-γ and HLA DR in both HIV and HIV/TB co-infected patients.
Levels of TNF-α and IFN-γ in HIV/TB co-infected patients and HIV patients
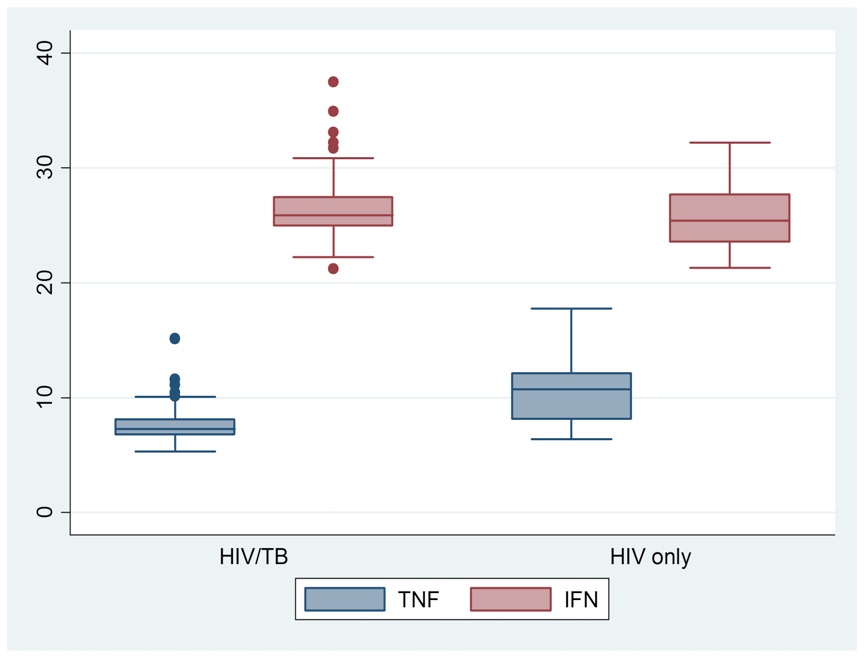
The median (IQR) TNF-α and IFN-γ in HIV/TB co-infected patients were 7.26 (6.76, 8.12) and 26.30 (25.18, 27.22) respectively as indicated in Figure 1.
Levels of CD38 and HLA-DR activation makers and CD4 in HIV/TB co-infected patients
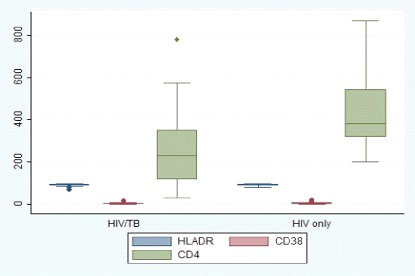
The median (IQR) CD38, HLA-DR and CD4 in HIV/TB co-infected patients were 4.36 (2.06- 6.31), 92.44 (90.52-95.51), 231 (117-350) respectively as indicated in Figure 2
Relationship in cytokine levels between HIV/TB co-infected patients and HIV only patients.
There was a significant difference in the levels of TNF-α, CD38 and CD4 between HIV/TB co-infected patients and HIV only patients as shown in Table 2. HIV only patients had higher levels of TNF-α, CD38 and CD4 compared to those of TB/HIV coinfected patients (p<0.05). However, no significant difference was observed in the levels of IFN-γ and HLA-DR (p=0.1559) and (p=0.1754) respectively.
Discussion & conclusion
There was a significant difference in the levels of TNF-α between HIV/TB co-infected patients and HIV only patients. HIV only patients had higher levels of TNF-α compared to those of HIV/TB co-infected patients. This was in line with similar findings by Palowski et al., (2012). According to Barlett et al., (2013), both HIV and MTB stimulated TNF-α release from infected cells, and TNF-α prevented bacterial growth but enhanced HIV replication. HIV replication lead to increase in viral load. Development of active TB was associated with increase in HIV viral load locally and systemically (Kwara et al., 2005). The excess of pro-inflammatory cytokines such as TNF-α lead to increased HIV virus replication [10]. This would imply that, higher levels of TNF-α created an environment that favoured HIV replication, increasing viral load, which in turn accelerated AIDS disease development. AIDS development subsequently leads to TB disease development, which is one of the opportunistic infections in HIV infected /AIDS patients. High levels of TNF-α in patients on HAART have been shown to indicate treatment failure, and reflect immune activation [8,21]. Increased TNF-α levels was linked to increased viral replication, high viral load, acceleration to disease, and low CD4 count [22]. Progression of HIV infection was related to increases in levels of TNF-α as the high levels propagated viral replication [17,23]. TNF-α was elevated in HIV patients before HAART, and the levels were seen high in patients failing HAART, indicating treatment failure [19]. Thus, the immunological response involving high TNF-α levels may signify a failing immune system due to virus replication. The control of the levels of TNF-α may help in the retaining of mycobacterium in a latent state.
There was no significant difference observed in the levels of IFN-γ. HIV positive and HIV positive Tuberculosis patients displayed similar IFN-γ levels. These results are similar with those attained by [20]. IFN- γ levels after active TB treatment indicated successful antibiotic-induced killing of all bacilli (Sauzullo et al., 2014). This implies that, in both HIV and HIV/TB co infection there was successful destruction of the bacilli, as a result, the levels of IFN-γ were similar. Also in the presence of TB in HIV patients, TB did not alter the level of IFN-γ.
Findings from the current study indicated a significant difference in the levels of TNF-α, CD38 and CD4 between HIV/TB co-infected patients and HIV only patients. HIV only patients had higher levels of TNF-α, CD38 and CD4 compared to those of HIV/TB co-infected patients. However, no significant difference was observed in the levels of IFN-γ and HLA DR. This findings are similar to those by [20] and [15]. The higher levels of TNF-α, CD38 and CD4 in the HIV only group show that there was viral replication, shown by high cytokine levels and activation markers, and a possibility of increased viral load, even when the patient was HAART. This may indicate failing HAART resulting into virus replication and problems in controlling the virus. This contrasted with findings by [26]. When the patient was on both HAART and Anti-TB, there was immune restoration, which tended to reduce the levels of Th1 cytokines. These findings suggest that, in the co-infected persons on HAART and Anti-TB, there is subsequent immune restoration, and in HIV only patients on HAART there is vigorous immune activation that leads to polarised cytokine production towards Th1 response. According to [27], TB infection had an effect on the reduction of CD4 cell count in HIV patients as the patient s immune system was at constant activation due to dual infection. Opportunistic infections in HIV patients with CD4 counts between 200-500 cells/ mm3 included pulmonary TB, and CD4 counts less than 200 cells included extra-pulmonary TB, (WHO, 2016). Lower CD4 count levels predisposed the patient to opportunistic infections such as TB.
References
- Barlett CP, DeWitt CC, Maronna B, Johnson K. Social media use as a tool to facilitate or reduce cyberbullying perpetration: A review focusing on anonymous and nonanonymous social media platforms. Violence and gender. 2018; 5: 147-152.
- Falvo JV, Jasenosky LD, Kruidenier L and Goldfeld AE. Epigenetic control of cytokine gene expression. Regulation of the TNF/LT locus and T helper cell differentiation. Advances in immunology. 2013; 118: 37-128.
- Nireekshana TG, Kesava Rao, Raju SS. Available transfer capability enhancement with FACTS using Cat Swarm Optimization.” Ain Shams Engineering Journal. 2016; 159-167.
- World Health Organisation, Systemic screening for active Tuberculosis: Principles and Recommendations. 2013.
- Pal Aukrust, Fredrick Muller, Egil Lien, Ingrild Nordoy, Nina-Beate Liabackk, dag Kvale; Tumor Necrosis Factor system levels in human immunodeficiency virus-infected patients during Highly Active Anteretroviral Therapy: Persistent TNF activation is associated virological and immunologic treatment failure. Journal of infectious diseases.1999; 179: 74-82.
- Abbas AK, Lichtman AH, Cellular and molecular Immunology, 5th edition, 2005. 243: 263-265.
- Dee Dee Wu, Can TNF inhibitors be used in HIV positive patients? Rheumatology Fellow, Hospital for special surgery. 2004.
- Emer JJ, Is there a potential role of anti-TNF Therapy in Patients with HIV? Journal of clinical and aesthetic dermatology. 2009; 2: 29-35.
- Keatingg SM, Golub ET, marek Nowicki, Mary young, Kathryn anastos, et al. The effect of HIV infection and HAART on inflammatory biomarkers in a population based cohort of US women, AIDS. 2011; 25: 1823-1832.
- Wada NI, Jacodson LP, Manqolick JB, Breen EL, Macatangay B, et al. The effect of HAART induced HIV suppression on circulating markers of inflammation and immune activation, AIDS. 2015; 20: 463-467.
- Epindola MS, Lima LJG, Soare LS, Cacemiro MC, Zambazi FA, et al. Dysregulated immune activation in second-line HAART HIV positive patients is similar to that of untreated patients, 2015; http:/dx.doi.org/10.1371/journal.pone.0145261.
- Qinglai meng, Ismail Sayin, Canaday DH, Harriet Mayanja Kizza, Joy Baseke, Zahra Toossi. Immune activation at sites of HIV/TB co-infection contributes to the pathogenesis of HIV-1 disease. 2016; http://dx.doi.org/10.1371/journal.pone .0166954.
- Da Silva TP, Giacoia Gripp CBW, Schmaltz CA, Sant Anna FM, Valeria Rolla, et al. T cell activation and cytokine profile of TB and HIV positive individuals during intituberculous treatment and Efavirenz- Based regimes, 2013; http://dx.doi.org/10.1371/ journal.pone.0066095. Nathalie
- Amirajan Chevilland, herve Tissor Dupornt, Yolande Obadia, Harve Gallais, Jean Louis mege, Christian capo. Highly Active Antiretroviral Therapy (HAART) and circulating markers of immune activation: Specific effect of HAART on neopterin. Clinical and diagnostic laboratory immunology. 2000.
- Amit Kumar, Wassim Abbas, Georges Herbein, TNF receptor super family members in HIV infection: New cellular targets for therapy? Mediators of imflammation. http://dx.doi. org/10.11155/2013/484378. Benito
- Lopez MJM, Lozano S, Martinez P, Ganzalez Lozano J, Soriano V. CD38 expression on CD8T lymphocytes as a marker of residual virus replication in chronically HIV-infected patients receiving Antiretroviral Therapy, AIDS res Hum Retroviruses. 2004; 20: 227-233.
- Fitzgerald Katherine A, Inke AIN, Gearing Andy IH. Cytokine facts book, academic press. 2001; 43-49.
- Chelty Nandini, Immunology: Introductory textbook, new age international. 2005.
- David Male, Jonathan Brostoff, Roth DB, Ivan Roitt, Immunology, 7th Edition. 2008; 487.
- Sivro A, Su RC, Plummer FA, Ball TB, AIDS review. Interferon responses in HIV infection: From protection to disease. 2014.
- Ugochukwu E. 2009; HIV/Tb co-infection in Nigerian children. vireo/ 5a899.
- Aluoch JA. HIV/TB co-infection: The challenges. East African Medical Journal. 90: 2013.
- Otieno CF, Otedo AEO, Othero D, Otiende MYC, Okoth P, et al. CD4+ cell response to ART in positive clinical care over one year period in a cohort of HAART Naïve, HIV positive Kenyan patients, East African Medical journal. 2013; 90.
- Bartlett JG, Redfield RR, Pharm PA, Sylvia Ojoo, MRCP. Medical management of HIV infection, CDC Kenya edition 16th edition. 2013; KEN -11.
- UNICEF- unite for Children, Integrating HIV/AIDS and sexual reproductive Health: Policy implications. 2009; 64-68.
- Jon Fielder, Tuberculosis in the era of HIV, a clinical manual for care providers working in Africa and other resource limited settings. 2010; 163-164.
- World Health Organisation, Implementation of the WHO stop TB strategy: A handbook for National TB control programme, regional office of Europe, 2008.