Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Renal cell carcinoma with leiomyomatous stroma: A case report and a review of the literature of a provisional entity distinct from conventional renal cell carcinoma
Nermeen Chaudhry; Nada Shaker; Monika Karera; Anil Parwani*
Department of Pathology, Ohio State University Wexner Medical Center, James Cancer Hospital and Richard Solove Research Institute, Columbus, OH USA.
*Corresponding Author: Anil Parwani
Professor of Pathology and Biomedical Informatics,
Vice-Chair of Anatomic Pathology, Director of Pathology Informatics Director, Digital Pathology Shared
Resources, Principal Investigator, Cooperative Human
Tissue Network (CHTN) Midwestern Division Wexner
Medical Center - Department of Pathology, The Ohio
State University, E409 Doan Hall, 410 West 10th Ave,
Columbus, OH, 43210, USA.
Email: anil.parwani@osumc.edu
Received : Nov 18, 2021
Accepted : Jan 06, 2022
Published : Jan 13, 2022
Archived : www.jcimcr.org
Copyright : © Parwani A (2022).
Abstract
Renal Cell Carcinoma with Leiomyomatous Stroma (RCCLMS) is a rare renal cancer with indolent behavior and favorable prognosis. We present a case of rare renal cell carcinoma with leiomyomatous stroma in a 49-year-old male, with an incidental finding of a 1.6 cm renal nodule on MRI scan. The patient underwent partial nephrectomy of the left kidney and subsequent histological examination showed an epithelial tumor composed of clear cell cytoplasm with low grade nuclear features, surrounded by an abundant smooth muscle stroma composed of fascicles of spindle cells. Immunohistochemistry revealed the positive reaction of tumor cells for CK7, CD10, CAIX, vimentin and desmin; negative for AMACR. Molecular testing via FISH study shows lack of 3p deletion. Final diagnosis of RCCLMS was made. The aim of this case report is to highlight the importance of awareness of this disease as well as to differentiate RCCLMS from other renal cancers with less favorable prognosis and higher degree of malignancy like clear cell renal cell carcinoma.
Keywords: renal cell carcinoma; leiomyomatous stroma; kidney.
Citation: Chaudhry N, Shaker N, Karera M, Parwani A. Renal cell carcinoma with leiomyomatous stroma: A case report and a review of the literature of a provisional entity distinct from conventional renal cell carcinoma. J Clin Images Med Case Rep. 2022; 3(1): 1560.
Introduction
Renal cell carcinoma accounts for approximately 3% of cancer cases in adults, and 90-95% of kidney neoplasms. It is also more common in males than females (2:1), and rare in children [1]. The most common subtype, clear cell renal cell carcinoma, accounts for 65-75% of all malignant renal tumors [2]. Renal Cell Carcinoma with Leiomyomatous Stroma (RCCLMS) is a rare entity that is thought to be distinct from clear cell renal carcinoma, though research is still ongoing. The first case report was described by Canzonieri et al. in 1993, and since then, The International Society of Urological Pathology 2012 Vancouver Classification of renal neoplasia and the World Health Organization 2016 Classification of renal tumors have classified renal cell carcinoma with leiomyomatous stroma as an emerging/provisional entity of renal cell carcinoma [3,4]. Less than 70 cases have been reported so far, with no gender predominance, and findings showing the right kidney is affected as commonly as the left [2].
Case report
A 49-year-old, Caucasian male with the history of GERD, HTN, WPW s/p ablation (1995), and VP shunt secondary to hydrocephalus (2003) presented to our institution for Urological evaluation of a left renal mass found incidentally during a previous MRI to evaluate lumbar radiculopathy. A follow up MRI of the abdomen w/wo contrast revealed a 1.6 X 1.2 cm enhancing mass in the left lower pole of the kidney. Physical examination and routine blood and serum analysis were unremarkable. The patient underwent laparoscopic partial left nephrectomy. The patient tolerated the procedure well and postoperative course was uncomplicated.
Pathological features
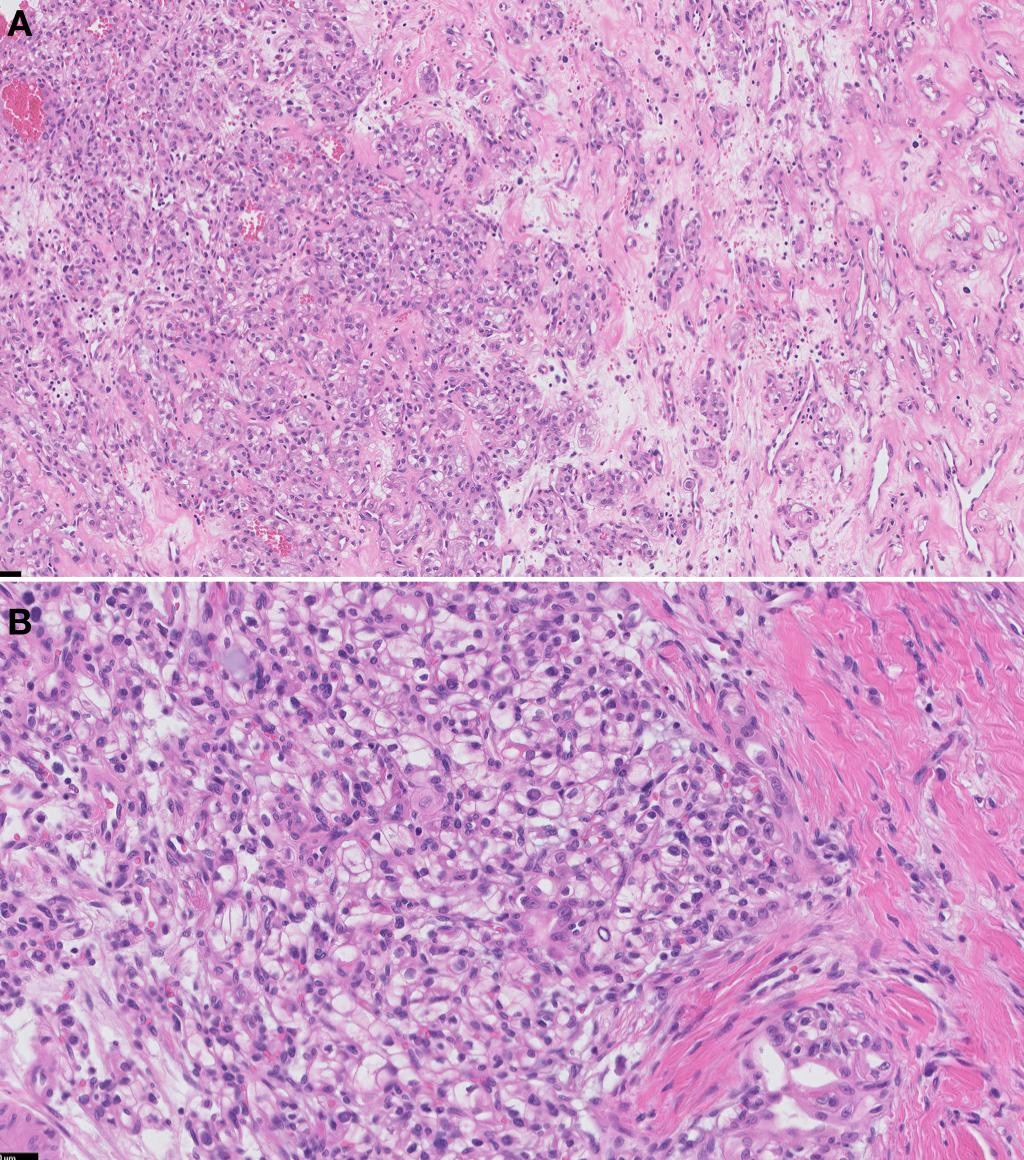
Macroscopically, the partial nephrectomy specimen weighed 2.89 grams and measured 2.4 X 1.9 X 1.4 cm in overall dimension. Serial sectioning revealed a 1.5 X 1.4 X 1.1 cm well-circumscribed, rubbery mass with tan-pink, homogeneous, solid cut surfaces that is less than 0.1 cm from the capsular surface and 0.2 cm from the renal parenchymal margin. The mass is limited to the kidney and does not appear to involve the perinephric fat. The surrounding uninvolved renal parenchyma is tan, rubbery, and exhibits significant pallor.
Microscopy
There are no characteristic morphological features unique for renal cell carcinoma with leiomyomatous stroma. The tumor is composed of anastomosing tubules of renal cells with clear cytoplasm. The cellular stroma was abundant and composed of intertwining bundles of smooth muscle with eosinophilic cytoplasm, elongated nuclei without significant pleomorphism, atypia, or mitoses. This morphology resembled benign smooth muscle tissue (Figure 1A,1B).
Results
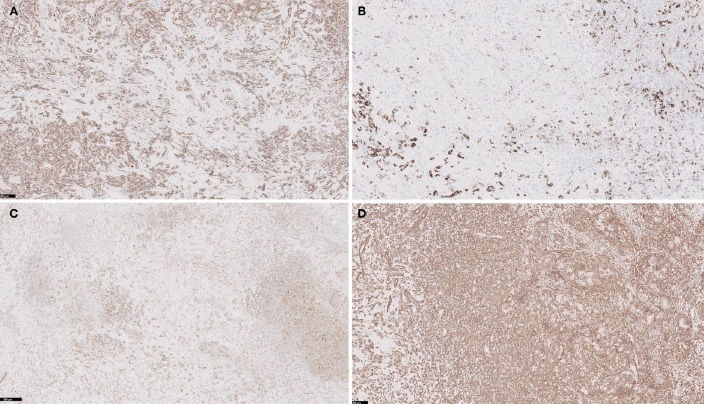
Studies were performed on one representative block of the lesion, and showed positive staining for vimentin, CAIX, CK7 (focal), CD10, desmin (highlights muscle in stroma); and negative staining for AMACR (Figure 2A-2D). A FISH Study was performed and showed no loss of Chromosome 3p which confirms that this is not clear cell subtype. Based on the morphology, immunoprofile and molecular genetics results, this tumor is best classified as Renal Cell Carcinoma with Leiomyomatous Stroma. The tumor was diagnosed as RCCLMS and staged as pT1aNx (TNM 7th edition).
Discussion
Renal Cell Carcinoma with Leiomyomatous Stroma (RCCLMS) is included as a provisional entity in the 2016 World Health Organization (WHO) classification of renal epithelial neoplasia. RCCLMS is a rare renal tumor with only a few cases reported so far. The cases reported show the age range of 31 to 79, with no gender predominance [2]. Most cases are found incidentally during routine imaging studies, with only 25% of the patients presenting with hematuria [5]. Most cases of RCCLMS were confined to the kidneys with patients having stage pT1a tumors [6-9]. Biological behavior of RCCLMSs is indolent with favorable prognosis.
Upon physical examination, RCCLMS presents as a well circumscribed, non-encapsulated tumor with a range of diameters from 0.8 to 5.0 cm. The cut surface is typically gray-white fibrotic tissues, with no yellow-tan or hemorrhagic areas and tumor necrosis, composed of small nests and nodules of renal epithelial cells with clear cytoplasms. Carcinoma cells are embedded in a cellular stroma composed of intertwining bundles of smooth muscle, showing no significant pleomorphism or atypia. The leiomyomatous stromal component is proposed to be polyclonal and therefore reactive, rather than neoplastic. A recent study analyzed the clonality of the smooth muscle component in 14 selected cases of renal epithelial neoplasms, rich in this type of stroma, and found them to be polyclonal in all analyzable cases (8 of 14). It is hypothesized to be derived from smooth muscle cells of large caliber veins located at the peripheral capsular region or within the collagenous septae of the tumors [10].
Immunohistochemically, the epithelial component is typically positive for pancytokeratin, EMA, CK7 and CD10, as well as CAIX, pankeratin, vimentin, and HIF1-alpha, as reported in the majority of RCCLSs published in the literature (Table 1). The stromal component is positive for smooth muscle actin, negative for HMB-45, MART-1, MiTF [2,11,12].
Table 1: Renal cell carcinoma with leiomyomatous stroma. Review of the literature.
Authors |
No. of cases |
Sex |
Age |
Tumor Diameter (cm) |
Stage |
Immunohisto- |
Molecular findings |
Martinoni et al. (2014) |
3 |
F (3) |
79, 74, 74 |
2-3
|
pT1a (3) |
CK7+ (3/3), AE1/AE3+ (3/3) 34BE12+ (3/3), CAIX+ (2/3) CD10+ (1/3), GLUT1+ (3/3) S100+ (2/3), AMACR, vimentin- |
3p deletion (0/3) VHL mutation (0/3) VHL methylation (0/3) VHL copy number changes (0/3) |
Pecova et al. (2014) |
5 |
M (2), F (3)
|
53, 63, 69, 63, 63 |
4
|
NA |
CK7+ (3/5), CD10+ (5/5), CAIX+ (5/5), vimentin+ (5/5) |
Absence of VHL mutation, hypermethylation, LOH3p (5/5), CEP7, CEP17 disomy (5/5), No TCEB mutation (4/5) |
Abrantes et al. (2015) |
1 |
F |
72 |
1.5 |
pT1a |
CK7+, CD10+, vimentin+, EMA+, vimentin, smooth muscle actin, HHF35, desmin and caldesmon + (stroma) |
NA |
Kiremit et al. (2017) |
1 |
M |
31 |
3.2 & 2.6 |
cT1a |
CK7+, pankeratin+, SMA, desmin+ (in stroma) |
NA |
Petersson et al. (2018) |
6 |
M(3) |
53- 69 |
1.5 - 2 |
NA |
NA |
Absence of VHL mutation, hypermethylation, No TCEB gene mutation (2/2) |
Bah et al. (2018) |
2 |
M(1) |
33, 60 |
1.5 |
pT1a |
CK7+ (1/1), CD10+ (1/1) CAIX+ (1/1), vimentin+ (1/1) |
TSC2 alteration |
Massaeli et al. (2019) |
2 |
M(1) |
50, 53 |
NA |
NA
|
MNF117+, PAX8+, CK7+ 34BE12-, CD10-, WT1-, HMB-45- |
NA
|
Shah et al. (2020) |
18 |
M-F (1:2) |
33-69 |
1.1 - 4.5 |
pT1a |
CK7+, CD10+ (18/18) |
Absence of VHL mutation (14/14), mutations (16/16): TSC1 (4), TSC2 (4), MTOR (6), and/or ELOC (2) |
Gournay et al. (2020) |
3 |
M (3) |
22-42 |
1-3 |
pT1a |
CK7+ (3/3), CK20+ (3/3), CAIX+ (3/3), CD10+ (3/3), AMACR- (3/3) |
TSC1 mutation (1/3), TSC2 mutation (2/3), absence of deletion of 3p and/or monosomy 8 (3/3), absence of Trisomy 7 or 17 (3/3) |
Ferguson et al. (2021) |
4 |
M (2) F(2) |
50-67 |
1.4 - 3.5 |
pT1a |
CK7+, CD10+ |
NA |
Current Case (2021) |
1 |
M |
49 |
1.5 |
pT1a |
CK7+, CD10+, vimentin+ and desmin+ (highlights muscle in stroma); AMACR- |
Absence of LOH3p |
Recent studies have attempted characterization of RCCLMS at a molecular level. Only tumors lacking alterations more characteristic of CCRCC, such as Von-Hippel Lindau (VHL) gene mutation or hypermethylation and loss of heterozygosity of chromosome 3p, were considered a distinct entity of RCCLMS. However, a TCEB1 mutation has been demonstrated in a subset of RCCLMS tumors with similar morphology, suggesting an alternative route of cell signaling in tumorigenesis [13,14].
Additionally, another study has demonstrated sporadic mutations in TSC1 and TSC2 resulted in gene mutations and allelic imbalance in patients with a family history of tuberous sclerosis [14]. There was a single case report of familial RCCLS associated with an atypical tuberous sclerosis complex, where researchers suggested TSC2 mutations support the hypothesis that RCCLMS is strongly related to abnormalities of the mTOR pathway [6]. The relationship between RCCLMS and similar tumors occurring in the setting of Tuberous Sclerosis Complex (TSC) was further evaluated, concluding that RRCLMS shows morphologic and molecular characteristics distinct from CCRCC and clear cell papillary RCC [15]. Furthermore, RCCLMS show recurrent mutations of TSC1/TSC2, MTOR, and/or ELOC, consistent with hyperactive MTOR complex, demonstrating morphologically identical tumors occurring in TSC patients. These findings suggest RRCLMS in TSC patients could constitute a distinct entity [15,16]. Lastly, a new study suggests intranuclear inclusions are a distinguishing morphological feature of RCCLMS. While intranuclear inclusions have been reported in RCC, with more frequency in papillary and chromophobe carcinoma, they are typically described as eosinophilic in color and were associated with grade 3/4 tumors. In contrast, the study found all cases of RCCLMS to have colorless inclusions in tumors with features consistent of grade 1/2 [18].
Prognosis of RCCLMS is generally good, with a low rate of tumor recurrence, highlighting the importance in accurate diagnosis. Clinical follow-up of many patients reported in the literature did not reveal tumor recurrence or metastasis, with surgical resection being curative and the treatment of choice [7,8,17]. The major differential diagnosis of RCCLS are other renal tumors with similar morphology, including Clear Cell Renal Cell Carcinoma (CCRCC), specifically with abundant smooth muscle stroma or sarcomatoid change. Clear Cell Papillary RCC (CCPRCC) and Renal Angiomyoadenomatous Tumor (RAT) also contains the smooth muscle component and are morphologically and immunohistochemically like each other, though the quantity of smooth muscle stroma is much greater in RAT than CCPRCC, and the malignant epithelial cellular features easily distinguish RCCLMS from both [2].
In contrast to clear cell RCC which macroscopically shows brown to golden yellow areas with hemorrhage or necrosis, the cut surface of RCCLMS shows solid white tissue witha “leiomyoma-like” appearance. Microscopically, clear cell RCCs have abundant cytoplasm that is vacuolated or granular, usually with indistinct cell borders. Clear cell RCC nuclei have variable atypia, irregular contours, haphazard orientation with abnormal chromatin and variably prominent nucleoli. In addition, clear cell RCC usually has no conspicuous smooth muscle stroma and is CK7 negative. Positive staining in RCC is usually PAX8, PAX2, and CAIX: Diffuse, membranous in 75-100% of clear cell RCC. Generally, positive for CD10 (proximal tubular marker), RCC, vimentin and epithelial markers including AE1 / AE3, CAM 5.2, EMA. Differential diagnostic methods from clear cell RCC include FISH, gene sequencing and methylation-specific multiplex ligation-dependent probe amplification analysis; where renal clear cell carcinoma will show deletion of chromosome 3p, VHL mutation, and VHL methylation abnormalities [7,12]. Ongoing research suggests additional differential diagnostic methods may include genetic testing specific to mutations of TSC1/TSC2, MTOR, and/or ELOC [15,16].
Declarations
Conflict of interest: The authors declare no conflicts of interest.
Disclosure of grants or other funding: No grants or funding was used in this study.
Consent: This case report has been completely anonymized and all tissue was obtained as part of the standard of care for the patient; hence no consent was required.
Data availability statement: The authors confirm that the data supporting the findings of this study are available within the article [and/or] its supplementary materials. Any additional information is available from the corresponding author, upon reasonable request.
References
- Cohen HT, McGovern FJ. Renal-cell carcinoma. N Engl J Med. 2005; 353: 2477–2490.
- Yeh YA, Constantinescu M, Chaudoir C, Tanner A, Serkin F, et al. Renal cell carcinoma with leiomyomatous stroma: A review of an emerging entity distinct from clear cell conventional renal cell carcinoma. American journal of clinical and experimental urology. 2019; 7: 321–326.
- Canzonieri V, Volpe R, Gloghini A, Carbone A, Merlo A. Mixed renal tumor with carcinomatous and fibroleiomyomatous components, associated with angiomyolipoma in the same kidney. Pathology, research and practice. 1993; 189; 951–959. https:// doi.org/10.1016/S0344-0338(11)81110-6
- Moch H, Humphrey PA, Ulbright TM, Reuter eds VE. World Health Organization classification of tumours of the urinary system and male genital organs. 4th ed. Lyon: IARC Press. 2016.
- Zhou M, Netto GJ, Epstein JI. Uropathology: High-Yield Pathology. Chapter G – Neoplastic Disease of the Kidney. People’s Republic of China: Elsevier-Sauders. 2012; 300-301.
- Bah I, Fahiminiya S, Begin LR, Hamel N, D’Agostino MD, Tanguay S, Foulkes WD. Atypical tuberous sclerosis complex presenting as familial renal cell carcinoma with leiomyomatous stroma. J Pathol Clin Res. 2018; 4: 167–174.
- Martignoni G, Brunelli M, Segala D, Gobbo S, Borze I, Atanesyan L, et al. Renal cell carcinoma with smooth muscle stroma lacks chromosome 3p and VHL alterations. Modern pathology: An official journal of the United States and Canadian Academy of Pathology, Inc. 2014; 27: 765–774. https://doi.org/10.1038/ modpathol.2013.180
- Kiremit MC, Acar Ö, Sağlıcan Y, Esen T. Bilateral renal cell carcinoma with leiomyomatous stroma: A rare entity diagnosed synchronously and treated surgically in a staged fashion. Turkish journal of urology. 2017; 43: 566–570. https://doi.org/10.5152/ tud.2017.68639
- Abrantes C, Oliveira R, Sepúlveda L, Figueiredo A, Marinho C, et al. A case of renal cell carcinoma with abundant smooth muscle (leiomyomatous) stroma and the differential diagnosis. Diagnostic Pathology. 2015; doi:10.17629/www.diagnosticpathology. eu-2015-1:28
- Petersson F, Branzovsky J, Martinek P, Korabecna M, Kruslin B, Hora M, et al. The leiomyomatous stroma in renal cell carcinomas is polyclonal and not part of the neoplastic process. Virchows Archiv: An international journal of pathology. 2014; 465: 89–96. https://doi.org/10.1007/s00428-014-1591-9
- Peckova K, Grossmann P, Bulimbasic S, Sperga M, Perez Montiel D, Daum O, et al. Renal cell carcinoma with leiomyomatous stroma-further immunohistochemical and molecular genetic characteristics of unusual entity. Annals of diagnostic pathology. 2014; 18: 291–296. https://doi.org/10.1016/j.anndiagpath.2014.08.004
- Williamson SR, Cheng L, Eble JN, True LD, Gupta NS, et al. Renal cell carcinoma with angioleiomyoma-like stroma: Clinicopathological, immunohistochemical, and molecular features supporting classification as a distinct entity. Modern pathology: An official journal of the United States and Canadian Academy of Pathology, Inc. 2015; 28: 279–294. https://doi.org/10.1038/ modpathol.2014.105
- Hakimi AA, Tickoo SK, Jacobsen A, Sarungbam J, Sfakianos JP, et al. TCEB1-mutated renal cell carcinoma: A distinct genomic and morphological subtype. Modern pathology: An official journal of the United States and Canadian Academy of Pathology, Inc. 2015; 28: 845–853. https://doi.org/10.1038/modpathol.2015.6
- Parilla M, Alikhan M, Al-Kawaaz M, Patil S, Kadri S, et al. Genetic Underpinnings of Renal Cell Carcinoma With Leiomyomatous Stroma. The American journal of surgical pathology. 2019; 43: 1135–1144.
- Shah RB, Stohr BA, Tu ZJ, Gao Y, Przybycin CG, Nguyen J, et al. “Renal Cell Carcinoma With Leiomyomatous Stroma” Harbor Somatic Mutations of TSC1, TSC2, MTOR, and/or ELOC (TCEB1): Clinicopathologic and Molecular Characterization of 18 Sporadic Tumors Supports a Distinct Entity. Am J Surg Pathol. 2020; 44: 571-581.
- Gournay M, Dugay F, Belaud Rotureau MA, Peyronnet B, Mathieu R, et al. Renal cell carcinoma with leiomyomatous stroma in tuberous sclerosis complex: A distinct entity. Virchows Arch. 2021; 478: 793-799.
- Petersson F, Martinek P, Vanecek T, Pivovarcikova K, Peckova K, Ondic O, et al. Renal Cell Carcinoma With Leiomyomatous Stroma: A Group of Tumors With Indistinguishable Histopathologic Features, But 2 Distinct Genetic Profiles: Next-Generation Sequencing Analysis of 6 Cases Negative for Aberrations Related to the VHL gene. Applied immunohistochemistry & molecular morphology: AIMM. 2018; 26: 192–197. https://doi.org/10.1097/ PAI.0000000000000410
- Ferguson PM, Kench JG, Watson GF. Intranuclear inclusions are a distinguishing morphological feature of renal cell carcinoma with leiomyomatous stroma. Pathology. 2021; 53: 543-545.